Advanced Synthesis of Triazolinethione Derivatives for High-Efficiency Fungicide Manufacturing
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of heterocyclic intermediates, particularly within the triazole class. Patent CN1286818C introduces a transformative methodology for preparing triazolinethione derivatives of Formula (I), which serve as critical precursors for microbiocidally active compounds. Unlike conventional routes that rely on hazardous elemental sulfur or complex reagents, this novel approach utilizes a streamlined thiocyanate-mediated cyclization strategy. This technical breakthrough not only enhances reaction yields but also simplifies the purification process, addressing key pain points for reliable agrochemical intermediate suppliers seeking to optimize their production portfolios. The core innovation lies in the sequential transformation of chlorohydrin or epoxide precursors into hydrazine derivatives, followed by a highly selective ring closure.
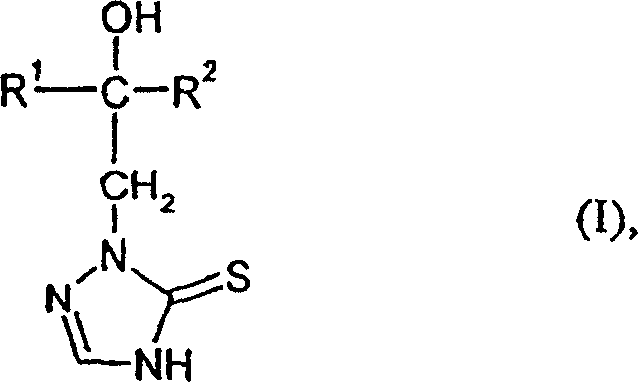
This structural versatility allows for the incorporation of diverse substituents, including halogenated aryl and cycloalkyl groups, which are essential for tuning the biological activity of the final fungicidal agents. By shifting away from legacy chemistries, manufacturers can achieve cost reduction in fungicide manufacturing while maintaining stringent quality standards required by regulatory bodies. The following analysis details the mechanistic advantages and commercial implications of adopting this patented synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triazoline-thione compounds has been plagued by significant operational challenges that hinder efficient commercial production. Traditional methods often involve reacting triazole derivatives with highly basic conditions followed by hydrolysis using Salmons-Saxl reagents, or alternatively, direct reaction with elemental sulfur at elevated temperatures. These legacy processes suffer from inherently low yields, often struggling to exceed theoretical expectations due to side reactions and decomposition. Furthermore, the requirement for high-temperature sulfurization poses severe safety risks and necessitates specialized equipment to handle corrosive byproducts and noxious sulfur vapors. The difficulty in maintaining precise reaction conditions on a technical scale frequently leads to batch-to-batch variability, complicating the supply chain for high-purity OLED material or agrochemical precursors. Additionally, the isolation of the desired product from complex reaction mixtures containing unreacted sulfur and polymeric impurities requires extensive downstream processing, driving up operational expenditures and waste generation.
The Novel Approach
In stark contrast, the method disclosed in CN1286818C offers a robust alternative that circumvents these historical bottlenecks through a mild, three-step cascade. The process initiates with the nucleophilic substitution of a 1-chloro-2-hydroxyethane derivative or an oxirane with hydrazine hydrate, generating a stable hydrazine intermediate under controlled temperatures. This is followed by a condensation reaction with a carbonyl compound and a thiocyanate salt, which facilitates the formation of the triazolidinethione ring with high regioselectivity. The final step involves a dehydration reaction using formic acid to aromatize the ring, yielding the target triazolinethione derivative. This route eliminates the need for elemental sulfur and harsh basic conditions, operating instead within a moderate temperature range of 80°C to 150°C. The use of common reagents like potassium thiocyanate and acetone ensures raw material availability, while the cleaner reaction profile allows for straightforward product isolation via simple recrystallization, significantly enhancing overall process efficiency.
Mechanistic Insights into Thiocyanate-Mediated Cyclization
The core of this synthetic advancement lies in the efficient construction of the 1,2,4-triazole-5-thione ring system via a thiocyanate-mediated cyclization mechanism. The reaction begins with the formation of a hydrazonoyl intermediate through the condensation of the hydrazine derivative with the carbonyl component. Subsequently, the nucleophilic attack of the thiocyanate anion on the electrophilic carbon of the hydrazone initiates the ring closure. This step is critical, as the thiocyanate group serves as both the sulfur source and a component of the heterocyclic ring, ensuring atom economy. The presence of an acid catalyst, such as hydrochloric acid or tosic acid, protonates the intermediate species, lowering the activation energy for cyclization and driving the equilibrium towards the triazolidinethione product. This mechanism avoids the radical pathways associated with elemental sulfur, thereby minimizing the formation of polysulfide impurities that are difficult to remove.
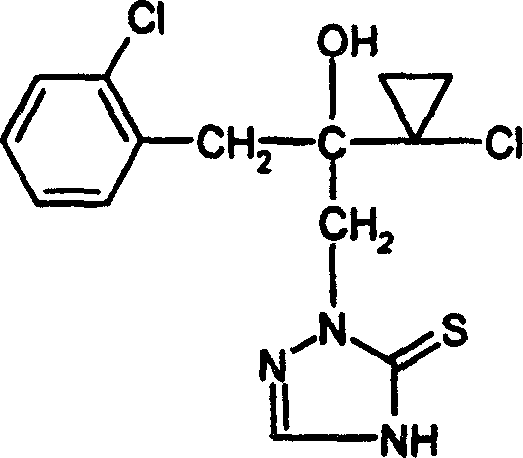
Following the cyclization, the treatment with formic acid acts as a dehydrating agent, facilitating the elimination of water to establish the double bond character within the triazole ring, thus converting the triazolidinethione into the fully aromatic triazolinethione. This dehydration step is highly selective, preserving sensitive functional groups such as the chlorohydrin moiety and the cyclopropyl ring, which might otherwise be susceptible to degradation under harsher conditions. The ability to perform this transformation with such chemoselectivity is paramount for R&D teams focusing on commercial scale-up of complex polymer additives or pharmaceutical intermediates where structural integrity is non-negotiable. The result is a clean reaction profile that yields colorless crystalline solids, as evidenced by the sharp melting points reported in the patent examples, indicating a high degree of crystallinity and purity.
How to Synthesize 2-(1-chloro-cyclopropyl)-1-(2-chloro-phenyl)-3-(4,5-dihydro-1,2,4-triazole-5-thioketones-1-yl)-propan-2-ol Efficiently
The synthesis of specific high-value intermediates, such as the chloro-phenyl substituted triazolinethione described in Example 1, demonstrates the practical applicability of this patented method. The process leverages the reactivity of 2-(1-chloro-cyclopropyl)-3-(2-chloro-phenyl)-2-hydroxyl-propyl-1-hydrazine, reacting it first with acetone and potassium thiocyanate to form the cyclic precursor. This intermediate is then subjected to reflux with formic acid to achieve the final dehydration. The detailed standardized synthesis steps for this specific transformation are outlined below to guide process development teams in replicating these results.
- React 1-chloro-2-hydroxyethane or oxirane derivatives with hydrazine hydrate to form the hydrazine intermediate.
- Cyclize the hydrazine derivative with a carbonyl compound and potassium/sodium thiocyanate in the presence of acid.
- Dehydrate the resulting triazolidinethione using formic acid to obtain the final triazolinethione product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this thiocyanate-based synthesis route presents compelling economic and logistical benefits that extend beyond mere chemical yield. By replacing expensive and hazardous reagents like Salmons-Saxl or elemental sulfur with commodity chemicals such as thiocyanates and formic acid, the direct material costs are significantly optimized. The elimination of heavy metal catalysts or complex organometallic reagents further reduces the burden on waste treatment facilities, aligning with increasingly strict environmental regulations. Moreover, the milder reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint for the manufacturing site. These factors collectively enhance the sustainability profile of the supply chain, making the final product more attractive to downstream customers who prioritize green chemistry principles.
- Cost Reduction in Manufacturing: The substitution of high-cost specialty reagents with bulk commodity chemicals drastically lowers the bill of materials. The avoidance of expensive purification steps required to remove sulfur residues or heavy metals translates directly into improved gross margins. Furthermore, the higher reaction yields observed in this method mean that less raw material is wasted per kilogram of finished product, maximizing resource utilization and reducing the overall cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as acetone, hydrazine hydrate, and potassium thiocyanate mitigates the risk of supply disruptions often associated with niche reagents. Since these commodities are produced at a global scale, procurement teams can secure long-term contracts with multiple vendors, ensuring business continuity. The robustness of the reaction also means that production schedules are less likely to be impacted by batch failures or extended reaction times, leading to more predictable lead times for customers.
- Scalability and Environmental Compliance: The process operates effectively at atmospheric pressure and moderate temperatures, removing the need for high-pressure reactors or specialized corrosion-resistant infrastructure. This simplicity facilitates rapid scale-up from pilot plant to commercial production volumes. Additionally, the aqueous workup and solvent recovery steps are straightforward, minimizing the generation of hazardous waste streams. This ease of compliance with environmental standards reduces regulatory risk and accelerates the time-to-market for new fungicidal formulations derived from these intermediates.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the process parameters and product specifications. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process engineers.
Q: What are the primary advantages of the thiocyanate cyclization method over traditional sulfurization?
A: The thiocyanate method avoids harsh high-temperature sulfur reactions and expensive Salmons-Saxl reagents, offering significantly higher yields (up to 76% vs 20% in comparative examples) and milder reaction conditions suitable for large-scale production.
Q: Can this process be scaled for commercial agrochemical manufacturing?
A: Yes, the process utilizes readily available raw materials like acetone and thiocyanates, operates at moderate temperatures (80-150°C), and avoids difficult isolation steps, making it highly amenable to commercial scale-up.
Q: What is the purity profile of the resulting triazolinethione derivatives?
A: The method allows for easy purification via recrystallization from solvents like toluene, typically yielding colorless crystalline solids with high purity suitable for downstream fungicide synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinethione Derivatives Supplier
The technological advancements detailed in CN1286818C represent a significant leap forward in the production of fungicidal intermediates, offering a pathway to higher purity and greater efficiency. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of triazolinethione derivative meets the exacting standards required for modern agrochemical applications.
We invite you to collaborate with us to leverage this superior synthetic route for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
