Optimizing Lorlatinib Production: A Technical Analysis of the TBTU-Mediated Cyclization Strategy
Optimizing Lorlatinib Production: A Technical Analysis of the TBTU-Mediated Cyclization Strategy
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with third-generation ALK inhibitors like Lorlatinib playing a pivotal role in treating non-small cell lung cancer (NSCLC). As demand for high-purity active pharmaceutical ingredients (APIs) surges, the efficiency of synthetic routes becomes a critical bottleneck for global supply chains. Patent CN112457293B introduces a transformative approach to the final cyclization step of Lorlatinib, utilizing O-benzotriazole-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU) in combination with 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). This technical insight report analyzes the mechanistic advantages of this novel condensation strategy, contrasting it with legacy methods to highlight its potential for cost reduction and scalable manufacturing. For procurement leaders and R&D directors, understanding these nuances is essential for securing a reliable API intermediate supplier capable of meeting stringent quality and volume requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Lorlatinib have historically struggled with suboptimal yields and inefficient reagent utilization, creating significant hurdles for commercial viability. Traditional protocols often rely on coupling agents like HATU (Hexafluorophosphate Azabenzotriazole Tetramethyl Uronium) paired with bases such as DIPEA or TEA. For instance, earlier iterations described in the background of the patent utilized HATU and DIPEA in DMF at low temperatures, resulting in dismal yields of merely 29%. Even when heating was applied to accelerate kinetics, yields only reached approximately 78%, accompanied by prolonged reaction times of 4 to 5 hours. Furthermore, alternative approaches employing COMU as a condensing agent failed to surpass 60% yield. These inefficiencies are not merely academic concerns; they translate directly into excessive raw material consumption, complex post-reaction workups due to high reagent loading, and difficult purification processes that compromise overall throughput. The accumulation of urea byproducts and the necessity for extensive chromatographic purification render these conventional routes economically unsustainable for large-scale production.
The Novel Approach
The innovation disclosed in CN112457293B fundamentally re-engineers the cyclization step by substituting traditional coupling systems with a specific TBTU and DBU combination. This strategic shift addresses the core inefficiencies of previous methods by optimizing the activation of the carboxylic acid moiety on the precursor molecule. 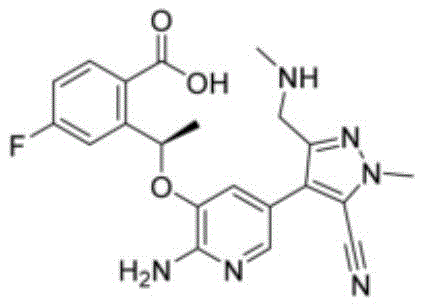 As illustrated by the structure of Compound A, the molecule possesses steric and electronic complexities that require precise activation to ensure successful macrocyclization without epimerization or side reactions. The new method operates under milder thermal conditions, typically between 40°C and 60°C, and achieves completion within a concise 2 to 3-hour window. Most critically, experimental data from the patent demonstrates that this approach consistently delivers yields exceeding 87%, with specific examples reaching 89.4% and purity levels above 97%. By drastically reducing the molar equivalents of the condensing agent and base required, this novel approach minimizes waste generation and simplifies the isolation of the final product, marking a substantial leap forward in process chemistry.
As illustrated by the structure of Compound A, the molecule possesses steric and electronic complexities that require precise activation to ensure successful macrocyclization without epimerization or side reactions. The new method operates under milder thermal conditions, typically between 40°C and 60°C, and achieves completion within a concise 2 to 3-hour window. Most critically, experimental data from the patent demonstrates that this approach consistently delivers yields exceeding 87%, with specific examples reaching 89.4% and purity levels above 97%. By drastically reducing the molar equivalents of the condensing agent and base required, this novel approach minimizes waste generation and simplifies the isolation of the final product, marking a substantial leap forward in process chemistry.
Mechanistic Insights into TBTU-Mediated Amide Bond Formation
The success of this synthetic route lies in the synergistic interaction between TBTU and DBU during the amide bond formation and subsequent cyclization. TBTU acts as a highly efficient uronium-type coupling reagent that activates the carboxylic acid group of the precursor to form an reactive O-acylisourea intermediate or an active ester species. Unlike phosphonium salts which can sometimes lead to racemization or slower activation kinetics, TBTU provides a balanced reactivity profile that is particularly well-suited for hindered substrates. The addition of DBU, a strong, non-nucleophilic amidine base, serves a dual purpose: it facilitates the deprotonation of the amine nucleophile to enhance its attack on the activated carbonyl, and it helps drive the equilibrium forward by scavenging the acidic byproducts generated during the coupling. This specific base selection is crucial, as comparative examples in the patent show that substituting DBU with weaker or more nucleophilic bases like DIPEA or TEA results in significantly lower yields and purity. The mechanism likely proceeds through a concerted pathway where the base promotes the intramolecular nucleophilic attack necessary for closing the macrocyclic ring, a step that is often the rate-determining factor in the synthesis of complex kinase inhibitors.
From an impurity control perspective, the TBTU/DBU system offers superior selectivity, which is paramount for meeting regulatory standards for oncology drugs. Conventional methods often suffer from the formation of N-acylurea byproducts or incomplete conversion, leading to difficult-to-remove impurities that persist through crystallization. The high purity observed in the new method (consistently >97% crude purity) suggests that the reaction pathway effectively suppresses these side reactions. The reduced loading of reagents (molar ratios of A:TBTU:DBU ranging from 1:0.5-1.5:0.1-1) means there is less residual coupling agent to remove during the workup. The quenching step using saturated ammonium chloride effectively neutralizes remaining active species, allowing for a clean extraction into organic solvents like ethyl acetate. This streamlined purification profile reduces the reliance on resource-intensive chromatography, enabling a more robust and reproducible manufacturing process that aligns with Quality by Design (QbD) principles essential for modern pharmaceutical production.
How to Synthesize Lorlatinib Efficiently
Implementing this optimized synthesis requires careful attention to stoichiometry and thermal management to maximize the benefits of the TBTU/DBU system. The process begins by dissolving the key precursor, Compound A, in a polar aprotic solvent such as DMF, although the patent notes that toluene and acetonitrile are also viable options depending on solubility profiles. The precise addition of TBTU and DBU follows, with the reaction mixture gently heated to maintain a temperature between 40°C and 60°C. Monitoring via TLC ensures the reaction is driven to completion without over-exposure to thermal stress. Following the reaction, a controlled quench with ammonium chloride allows for the safe decomposition of excess coupling reagents. The detailed standardized synthetic steps, including specific workup parameters and drying conditions, are outlined in the guide below to ensure reproducibility across different manufacturing scales.
- Dissolve the Compound A precursor in an organic solvent such as DMF or toluene.
- Add the condensing agent TBTU and the alkali promoter DBU, then heat the mixture to 40-60°C for 2-3 hours.
- Quench the reaction with ammonium chloride solution, extract with ethyl acetate, and purify the crude solid via washing and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this TBTU-mediated process represents a strategic opportunity to enhance supply security and reduce total cost of ownership. The primary economic driver here is the drastic reduction in reagent consumption. By lowering the molar equivalents of the expensive condensing agent and base, the direct material costs for the synthesis are significantly curtailed. Furthermore, the simplified workup procedure—eliminating the need for complex chromatographic separations often required by lower-purity legacy methods—reduces solvent usage and processing time. This efficiency translates into a leaner manufacturing footprint, allowing facilities to increase batch turnover rates without expanding infrastructure. The ability to achieve high yields consistently means less raw material is wasted on failed batches or reprocessing, providing a buffer against volatility in the pricing of key starting materials. Ultimately, this process optimization supports a more resilient supply chain capable of withstanding market fluctuations while maintaining competitive pricing structures for the final API.
- Cost Reduction in Manufacturing: The implementation of the TBTU/DBU system eliminates the need for excessive quantities of coupling reagents that characterize older HATU-based protocols. By operating with near-stoichiometric amounts of activators, the process inherently lowers the bill of materials for every kilogram of Lorlatinib produced. Additionally, the high crude purity achieved (>97%) significantly reduces the burden on downstream purification units, saving substantial amounts of solvents and energy otherwise spent on recrystallization or column chromatography. This holistic reduction in resource intensity drives down the variable cost per unit, offering a clear path to margin improvement for manufacturers.
- Enhanced Supply Chain Reliability: The robustness of this chemical route directly correlates to supply continuity. Conventional methods with low yields (e.g., 29%) are prone to supply shocks because they require significantly more starting material to produce the same output, straining upstream suppliers. In contrast, the high-efficiency nature of this new method ensures that production targets can be met with existing raw material inventories. The use of common, commercially available solvents like DMF and toluene further mitigates supply risk, as these are not subject to the same geopolitical or logistical constraints as specialized reagents. This reliability is critical for maintaining uninterrupted production schedules for life-saving oncology medications.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste issues, but this methodology is designed with green chemistry principles in mind. The reduction in reagent loading directly decreases the E-factor (mass of waste per mass of product), simplifying wastewater treatment and hazardous waste disposal. The milder reaction temperatures (40-60°C) also reduce the energy load on reactor heating systems compared to processes requiring reflux or cryogenic cooling. These factors make the technology highly scalable from pilot plants to multi-ton commercial reactors, ensuring that environmental compliance remains manageable even as production volumes ramp up to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in CN112457293B, offering clarity on why this method is becoming the preferred choice for advanced intermediate manufacturing. Understanding these details helps stakeholders make informed decisions about technology transfer and vendor qualification.
Q: Why is the TBTU/DBU system superior to HATU/DIPEA for Lorlatinib synthesis?
A: The TBTU/DBU system significantly improves yield (up to 89.4% vs 29-78%) and purity while reducing the molar equivalents of reagents required, simplifying downstream purification.
Q: What are the optimal reaction conditions for this cyclization?
A: The patent specifies heating the system to 40-60°C for 2-3 hours in solvents like DMF or toluene, which balances reaction kinetics with thermal stability.
Q: How does this method impact industrial scalability?
A: By eliminating the need for excessive reagents and harsh low-temperature conditions, the process reduces waste generation and operational complexity, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lorlatinib Supplier
As the global demand for third-generation ALK inhibitors continues to rise, partnering with a manufacturer that possesses deep technical expertise in complex cyclization chemistries is vital. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process knowledge to deliver high-purity Lorlatinib intermediates that meet the rigorous standards of international regulatory bodies. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your clinical and commercial needs seamlessly. We utilize stringent purity specifications and operate rigorous QC labs to guarantee that every batch conforms to the highest quality benchmarks, minimizing risk for your downstream formulation processes.
We invite you to engage with our technical procurement team to discuss how this optimized TBTU-mediated route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a secure and efficient supply of this critical oncology ingredient.
