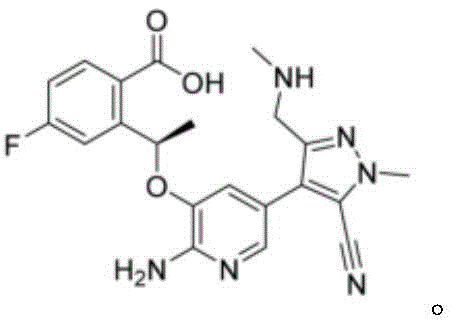
Optimizing Lorlatinib Production: A Technical Analysis of the TBTU-DBU Cyclization Strategy
The pharmaceutical industry continuously seeks robust synthetic routes for third-generation ALK inhibitors, specifically focusing on the production of Lorlatinib. Patent CN112480072A introduces a significant technological advancement in this domain by detailing a preparation method that utilizes a specific combination of O-benzotriazole-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU) and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). This novel approach addresses critical bottlenecks found in earlier methodologies, offering a pathway that balances high chemical efficiency with economic viability. For R&D directors and procurement strategists, understanding the nuances of this cyclization reaction is paramount, as it directly influences the cost of goods sold (COGS) and the reliability of the supply chain for this high-value anticancer active pharmaceutical ingredient. The patent explicitly outlines a process where Compound A undergoes intramolecular condensation under mild thermal conditions, resulting in superior yields compared to traditional coupling agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Lorlatinib have historically relied on coupling agents such as HATU or COMU, often in conjunction with bases like DIPEA or TEA. These conventional routes suffer from significant inefficiencies, primarily characterized by excessive reagent loading and suboptimal reaction yields. For instance, existing literature and comparative data indicate that using HATU with DIPEA can result in yields as low as 29% under certain low-temperature conditions, or require extended reaction times of 4 to 5 hours at elevated temperatures to reach approximately 78% yield. Furthermore, the use of COMU has been associated with yields dropping below 60%. Beyond the yield deficit, these methods necessitate large quantities of expensive condensing agents and promoters, which not only drives up raw material costs but also complicates the post-reaction workup. The accumulation of urea byproducts and salt residues from these coupling reagents creates a complex impurity profile, demanding rigorous and costly purification steps to meet the stringent quality standards required for oncology drugs.
The Novel Approach
In stark contrast, the methodology disclosed in CN112480072A leverages the synergistic effect of TBTU and DBU to overcome these historical limitations. By optimizing the molar ratios of the condensing agent and the base promoter, the inventors have achieved a reaction system that operates efficiently at moderate temperatures between 40°C and 60°C. This novel approach drastically reduces the reaction time to merely 2 to 3 hours while simultaneously boosting the isolated yield to ranges between 85.8% and 89.4%. The strategic selection of DBU as a non-nucleophilic base facilitates a cleaner cyclization process, minimizing side reactions that typically plague amide bond formations. Additionally, the flexibility in solvent choice, allowing for the use of DMF, DMSO, acetonitrile, toluene, or xylene, provides process chemists with the ability to tailor the reaction medium for optimal solubility and downstream processing. This adaptability is crucial for industrial scale-up, where solvent recovery and environmental impact are key considerations for a reliable API intermediate supplier.
Mechanistic Insights into TBTU-DBU Mediated Cyclization
The core of this synthetic breakthrough lies in the mechanistic efficiency of the TBTU-DBU system. TBTU acts as a potent uronium-type coupling reagent that activates the carboxylic acid moiety of Compound A, forming a highly reactive O-acylisourea intermediate. This activated species is particularly susceptible to nucleophilic attack by the proximal amine group within the same molecule, driving the intramolecular cyclization forward. The presence of DBU is critical in this mechanism; as a strong, sterically hindered organic base, it effectively scavenges the protons generated during the activation and cyclization steps without participating in unwanted nucleophilic side reactions. This ensures that the reaction equilibrium is shifted decisively towards the formation of the macrocyclic lactam structure characteristic of Lorlatinib. The mild thermal conditions (40-60°C) are sufficient to overcome the activation energy barrier for this cyclization without inducing thermal degradation of the sensitive functional groups present in the molecule, such as the fluorophenyl and pyrazole rings.
From an impurity control perspective, this mechanism offers distinct advantages over HATU-based systems. The reduced tendency for racemization and the minimization of N-acylurea byproducts result in a cleaner crude reaction mixture. The patent data supports this, showing purity levels consistently above 97% even before extensive chromatographic purification. The simplified impurity profile means that the final crystallization or washing steps (such as the n-hexane wash described in the examples) are far more effective at removing residual starting materials and reagents. For quality control teams, this translates to a more robust process with lower batch-to-batch variability. The ability to quench the reaction simply with ammonium chloride solution further streamlines the workflow, avoiding the complex aqueous workups often required to remove phosphorus-containing byproducts from other coupling reagents. This mechanistic clarity provides a solid foundation for scaling the process from laboratory grams to commercial metric tons.
How to Synthesize Lorlatinib Efficiently
The operational simplicity of this patented route makes it highly attractive for technology transfer and commercial manufacturing. The process begins with the dissolution of the key intermediate, Compound A, in a suitable organic solvent, with DMF being the preferred choice for optimal kinetics, although toluene serves as a viable green alternative. Following the addition of TBTU and DBU in controlled molar ratios, the system is heated to maintain a steady reaction temperature, ensuring complete conversion as monitored by TLC. The detailed standardized synthesis steps, including specific quenching and extraction protocols, are outlined in the structured guide below to assist process engineers in replicating these high-yield results.
- Dissolve Compound A in an organic solvent such as DMF or Toluene and add the condensing agent TBTU along with the base promoter DBU.
- Heat the reaction mixture to a temperature range of 40-60°C and maintain stirring for 2 to 3 hours to ensure complete cyclization.
- Quench the reaction with saturated ammonium chloride solution, extract the organic phase, and purify the crude solid via washing and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this TBTU-DBU mediated synthesis represents a strategic opportunity to optimize the cost structure and reliability of Lorlatinib supply. The primary driver of value here is the substantial reduction in raw material intensity. By achieving higher yields with lower loading of coupling reagents, the overall material cost per kilogram of API is significantly decreased. This efficiency gain is compounded by the simplified downstream processing; fewer purification steps mean reduced solvent consumption, lower energy usage for distillation, and shorter cycle times. These factors collectively contribute to a more competitive pricing model for the final API, allowing pharmaceutical companies to better manage their budget allocations for oncology portfolios while maintaining healthy margins.
- Cost Reduction in Manufacturing: The elimination of excessive coupling agents and the move towards higher yielding reactions directly lowers the variable cost of production. Unlike previous methods that required stoichiometric excesses of expensive reagents like HATU to drive the reaction, this optimized protocol operates efficiently with near-stoichiometric amounts of TBTU. This reduction in reagent load not only saves on direct material costs but also reduces the volume of chemical waste generated, thereby lowering waste disposal fees. Furthermore, the ability to use cost-effective solvents like toluene instead of solely relying on high-boiling polar aprotic solvents can lead to additional savings in solvent recovery operations. The cumulative effect of these efficiencies creates a leaner manufacturing process that is economically superior to legacy synthetic routes.
- Enhanced Supply Chain Reliability: Diversifying the solvent system to include widely available industrial solvents like toluene and xylene mitigates the risk of supply disruptions associated with specialized reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, ensures consistent production output even when facing minor variations in raw material specifications. This resilience is critical for maintaining continuous supply to downstream drug product manufacturers. Moreover, the high purity of the crude product reduces the dependency on scarce chromatography resins or specialized purification media, further stabilizing the supply chain against external market fluctuations. A reliable lorlatinib intermediate supplier can leverage this robustness to guarantee tighter delivery schedules and improved service levels.
- Scalability and Environmental Compliance: The simplified workup procedure, involving straightforward extraction and crystallization, is inherently easier to scale than complex multi-step purifications. This scalability ensures that the transition from pilot plant to full commercial production can be executed with minimal technical risk. From an environmental standpoint, the reduction in chemical waste and the potential for solvent recycling align with increasingly strict global environmental regulations. The process generates fewer hazardous byproducts, simplifying the permitting process for manufacturing facilities and reducing the long-term environmental liability. This alignment with green chemistry principles not only enhances corporate sustainability profiles but also future-proofs the manufacturing asset against tightening regulatory frameworks regarding chemical emissions and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using TBTU and DBU over HATU for Lorlatinib synthesis?
A: The use of TBTU and DBU significantly reduces reagent consumption while achieving yields exceeding 85%, compared to lower yields and higher waste generation associated with HATU-based protocols.
Q: Can this synthesis method be scaled using solvents other than DMF?
A: Yes, the patent demonstrates successful scalability using alternative solvents such as Toluene, which offers potential advantages in solvent recovery and environmental compliance without compromising yield.
Q: What purity levels can be expected from this cyclization process?
A: Experimental data indicates that the process consistently delivers product purity above 97%, minimizing the need for extensive downstream purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lorlatinib Supplier
The technical potential of the TBTU-DBU cyclization route for Lorlatinib is immense, offering a clear path to cost-effective and high-quality production. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Lorlatinib intermediate adheres to the highest international standards. We understand the critical nature of oncology supply chains and are committed to delivering consistency and reliability in every shipment.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the potential economic advantages of switching to this newer methodology. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to secure a sustainable and efficient supply of this vital anticancer medicine.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
