Advanced Florfenicol Manufacturing: A Breakthrough in Sulfuryl Fluoride Mediated Synthesis
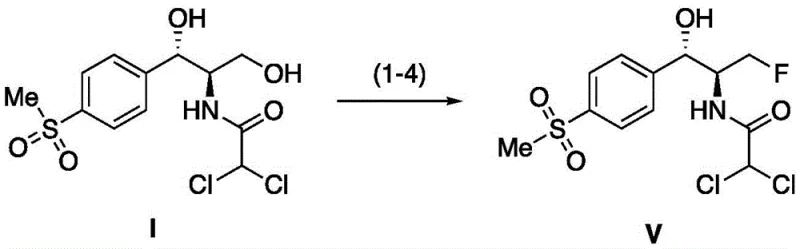
The pharmaceutical and veterinary industries are constantly seeking more efficient and environmentally benign pathways for the production of critical antibiotics. Patent CN111153838A, published on May 15, 2020, introduces a transformative synthetic method for Florfenicol, a broad-spectrum amino alcohol antibiotic widely used in aquaculture and poultry. This innovation addresses long-standing challenges in the dehydroxyfluorination step, a crucial transformation in converting Thiamphenicol to Florfenicol. Traditionally, this step has relied on reagents that pose significant safety hazards or generate complex waste streams. The disclosed method utilizes sulfuryl fluoride (SO2F2) as a mediating reagent, replacing the historically common Ishikawa reagent or DAST. This shift not only simplifies the reaction pathway but also drastically alters the waste profile, substituting difficult-to-treat organic fluorinated amides with sulfuric acid, a byproduct manageable through mature industrial neutralization processes.
This technological leap is particularly relevant for manufacturers aiming to optimize their veterinary drug intermediate supply chains. By leveraging a gaseous reagent that can be easily introduced and removed, the process offers enhanced control over reaction kinetics and selectivity. The patent outlines a robust four-step sequence—cyclization, rearrangement, dehydroxyfluorination, and hydrolysis—that can be executed sequentially or even telescoped into a one-pot synthesis. For R&D directors and process chemists, this represents a significant opportunity to refine impurity profiles and improve overall process mass intensity (PMI). The ability to achieve medium to high yields while utilizing commercially available starting materials like Thiamphenicol positions this method as a viable candidate for immediate scale-up in commercial facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the fluorine atom in Florfenicol synthesis has been a bottleneck due to the limitations of available fluorinating agents. Early patents, such as US 4361557A, utilized DAST (diethylaminosulfur trifluoride) for the dehydroxyfluorination of primary alcohols containing an oxazoline ring. However, DAST is notorious for its thermal instability and poor selectivity; it often indiscriminately fluorinates secondary alcohol structures within the substrate, leading to a complex mixture of unwanted by-products that are difficult to separate. This lack of chemoselectivity necessitates extensive purification steps, driving up costs and reducing overall throughput. Furthermore, the handling of DAST requires stringent safety protocols due to its potential for explosive decomposition, posing a risk to plant personnel and equipment integrity.
In more recent years, the industry has shifted towards using Ishikawa's reagent (N,N-diethyl-α,α-difluoro(m-methoxybenzyl)amine) to mitigate some of these selectivity issues. While effective, this reagent introduces its own set of logistical and environmental burdens. Upon reaction, Ishikawa's reagent decomposes to form large quantities of N,N-diethyl-2,3,3,3-tetrafluoropropanamide. This fluorinated amide byproduct is chemically stable and difficult to recover or recycle, creating a significant waste disposal challenge. The accumulation of such persistent organic pollutants increases the environmental footprint of the manufacturing process and complicates regulatory compliance regarding effluent treatment. Consequently, there is a pressing need for a dehydroxyfluorination strategy that balances high selectivity with environmental sustainability and operational safety.
The Novel Approach
The methodology described in CN111153838A offers a compelling alternative by employing sulfuryl fluoride (SO2F2) as the primary dehydroxyfluorination mediator. Unlike liquid reagents that leave behind heavy organic residues, SO2F2 is a gas at room temperature, allowing for precise stoichiometric control and easy removal of excess reagent simply by venting or purging with inert gas. The core innovation lies in the activation of the hydroxyl group via sulfuryl fluoride to form a reactive intermediate, which subsequently undergoes nucleophilic substitution by fluoride ions provided by a source like triethylamine trihydrofluoride. This mechanism ensures high regioselectivity for the primary alcohol, preserving the integrity of the secondary hydroxyl group essential for biological activity.
Moreover, the byproduct of the sulfuryl fluoride reaction is sulfuric acid (or its salts upon neutralization), which stands in stark contrast to the complex fluorinated amides generated by Ishikawa's reagent. Sulfuric acid is a commodity chemical with well-established neutralization and disposal protocols, significantly lowering the barrier for wastewater treatment. The process is versatile, accommodating various organic solvents such as dichloromethane, 1,2-dichloroethane, and tetrahydrofuran, and operates effectively at mild temperatures ranging from 0°C to 50°C. This flexibility allows manufacturers to adapt the process to existing infrastructure without requiring exotic cryogenic conditions or high-pressure reactors beyond standard gas-handling capabilities. The result is a streamlined, safer, and more cost-effective route to high-purity Florfenicol.
Mechanistic Insights into Sulfuryl Fluoride Mediated Fluorination
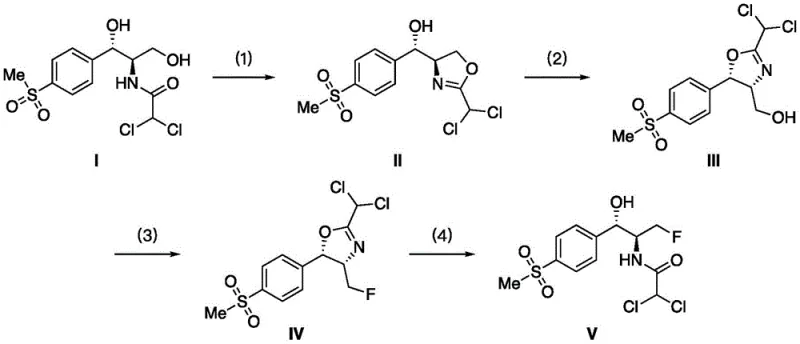
The reaction mechanism proceeds through a carefully orchestrated sequence of four distinct chemical transformations, each optimized to maximize yield and minimize side reactions. The process initiates with the cyclization of Thiamphenicol (Compound I) under a sulfuryl fluoride atmosphere in the presence of an acid-binding agent such as triethylamine or potassium carbonate. In this step, the sulfuryl fluoride activates the amide or hydroxyl functionality to facilitate the formation of the oxazoline ring, yielding Compound II. This cyclization is critical as it protects the stereochemistry of the molecule and sets the stage for the subsequent fluorination. The reaction is typically conducted at temperatures between 0°C and 50°C, with the partial pressure of SO2F2 maintained between 0.1 and 0.5 MPa to ensure sufficient reagent concentration in the liquid phase.
Following cyclization, the intermediate undergoes a rearrangement reaction mediated by triethylamine trihydrofluoride (Et3N·3HF). This step converts Compound II into Compound III, preparing the molecular scaffold for the definitive fluorine insertion. The use of Et3N·3HF is strategic; it serves as a mild yet effective source of nucleophilic fluoride ions while maintaining a basic environment that prevents acid-catalyzed degradation of the sensitive oxazoline ring. The rearrangement is typically heated to temperatures between 40°C and 100°C to overcome the activation energy barrier, ensuring complete conversion of the intermediate. Subsequent dehydroxyfluorination of Compound III reintroduces the sulfuryl fluoride atmosphere, activating the primary hydroxyl group for displacement by fluoride. Finally, the protective oxazoline ring is opened via hydrolysis using dilute hydrochloric acid, releasing the free amine and yielding the final Florfenicol product (Compound V).
How to Synthesize Florfenicol Efficiently
The synthesis of Florfenicol via this novel pathway is designed for operational simplicity and scalability. The patent details a procedure where Thiamphenicol is first dissolved in an organic solvent like dichloromethane and treated with an acid scavenger. Sulfuryl fluoride gas is then introduced to the system to drive the initial cyclization. Once this step is complete, the atmosphere can be swapped for nitrogen, and the fluorinating agent (Et3N·3HF) is added for the rearrangement phase. Crucially, the process allows for the re-introduction of sulfuryl fluoride for the final fluorination step before a concluding acidic workup. This modularity enables chemists to monitor each stage via TLC or HPLC, ensuring that intermediates are fully consumed before proceeding, which is vital for maintaining high purity standards in the final API. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles, please refer to the standardized synthesis guide below.
- Cyclization of Thiamphenicol (Compound I) under sulfuryl fluoride atmosphere with an acid-binding agent to form Compound II.
- Rearrangement of Compound II using triethylamine trihydrofluoride to generate Compound III.
- Dehydroxyfluorination of Compound III under sulfuryl fluoride atmosphere to produce Compound IV.
- Hydrolysis of Compound IV using dilute hydrochloric acid to yield final Florfenicol (Compound V).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfuryl fluoride-based methodology presents a clear value proposition centered on cost efficiency and risk mitigation. The primary driver for cost reduction is the replacement of expensive, specialized fluorinating reagents like Ishikawa's reagent with sulfuryl fluoride, which is produced on a massive industrial scale for other applications (such as fumigation and semiconductor etching) and is therefore significantly more affordable. Furthermore, the elimination of complex fluorinated amide byproducts removes the need for specialized waste incineration or hazardous waste disposal contracts, leading to substantial savings in environmental compliance costs. The simplified waste stream, consisting largely of inorganic salts and sulfuric acid, can often be treated on-site, reducing the logistical burden of transporting hazardous chemical waste off-site.
Supply chain reliability is another critical benefit derived from this process innovation. The reliance on commodity chemicals—sulfuryl fluoride, triethylamine, and common organic solvents—means that manufacturers are less vulnerable to the supply disruptions that often plague niche fine chemical reagents. The potential for a one-pot synthesis, as demonstrated in the patent examples, further enhances supply security by reducing the number of unit operations and intermediate isolation steps. Fewer processing steps translate to shorter cycle times and reduced exposure to potential contamination or yield loss during transfers between reactors. This streamlined approach facilitates faster batch turnover, allowing suppliers to respond more agilely to fluctuations in market demand for veterinary antibiotics. Additionally, the improved safety profile of using a gas rather than thermally unstable liquids reduces insurance premiums and operational downtime associated with safety incidents.
- Cost Reduction in Manufacturing: The substitution of high-cost Ishikawa reagent with inexpensive sulfuryl fluoride gas directly lowers the raw material cost per kilogram of Florfenicol produced. Additionally, the generation of sulfuric acid as a byproduct instead of persistent organic fluorinated waste eliminates the need for expensive hazardous waste disposal services, resulting in significant downstream cost savings. The ability to recycle solvents like dichloromethane or 1,2-dichloroethane further contributes to the economic viability of the process.
- Enhanced Supply Chain Reliability: By utilizing globally available commodity gases and solvents, the manufacturing process becomes resilient to the supply chain bottlenecks often associated with custom-synthesized fine chemical reagents. The simplified workflow, potentially executable as a one-pot reaction, reduces the dependency on multiple processing vessels and lengthy purification sequences, thereby shortening the overall lead time from raw material intake to finished goods inventory.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of gas-liquid reactions which can be efficiently managed in standard stirred-tank reactors equipped with gas spargers. The environmental profile is markedly improved as the avoidance of difficult-to-degrade fluorinated amides simplifies effluent treatment, ensuring compliance with increasingly stringent global environmental regulations regarding persistent organic pollutants and fluorine-containing waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfuryl fluoride-mediated synthesis. These insights are derived directly from the experimental data and claims presented in patent CN111153838A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer.
Q: What are the advantages of using sulfuryl fluoride over Ishikawa reagent?
A: Sulfuryl fluoride avoids the formation of difficult-to-recycle N,N-diethyl-2,3,3,3-tetrafluoropropanamide byproducts associated with Ishikawa reagent. Instead, it produces sulfuric acid, which is easier to treat using mature industrial processes.
Q: Can this synthesis be performed as a one-pot reaction?
A: Yes, the patent describes a one-pot method where cyclization, rearrangement, dehydroxyfluorination, and ring-opening occur continuously without intermediate separation, significantly simplifying the operational workflow.
Q: What are the typical reaction conditions for the dehydroxyfluorination step?
A: The dehydroxyfluorination is conducted under a sulfuryl fluoride atmosphere (0.1 to 0.5 MPa partial pressure) at temperatures ranging from 0°C to 50°C in solvents like dichloromethane or 1,2-dichloroethane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global veterinary pharmaceutical market. Our team of expert process chemists has extensively analyzed the sulfuryl fluoride-mediated pathway described in CN111153838A and possesses the technical capability to implement this advanced route at commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with the necessary gas-handling infrastructure and corrosion-resistant reactors required to safely manage sulfuryl fluoride chemistry, guaranteeing consistent quality and supply continuity.
We are committed to delivering stringent purity specifications for all our veterinary intermediates and APIs, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. Whether you are looking to optimize an existing supply chain or develop a new source for high-quality Florfenicol, we invite you to engage with our technical sales team. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can meet your strategic sourcing goals.
