Advanced Two-Step Asymmetric Synthesis of Florfenicol Intermediate for Efficient Commercial Scale-up and Supply
The global demand for broad-spectrum veterinary antibiotics continues to drive innovation in the synthesis of key precursors, with Patent CN110156645B representing a significant leap forward in the production of florfenicol intermediates. This patent discloses a highly efficient preparation method for (1R, 2R)-2-amino-1-(4-methylsulfonyl)phenyl)propane-1,3-diol, utilizing a sophisticated asymmetric addition reaction catalyzed by a novel copper salt complex. By shifting away from traditional resolution-based methodologies, this technology offers a streamlined pathway that addresses critical pain points regarding waste generation and operational complexity. For R&D directors and supply chain managers, understanding this shift is vital, as it promises not only higher purity profiles but also a more sustainable manufacturing footprint. The integration of a chiral ligand system allows for precise stereocontrol, ensuring that the final product meets the stringent optical purity requirements necessary for downstream pharmaceutical applications without the material losses associated with older techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical veterinary drug intermediate has relied on multi-step sequences that are inherently inefficient and environmentally burdensome. Traditional routes typically involve the condensation of p-methylsulfonylbenzaldehyde with glycine in the presence of copper sulfate, followed by esterification and a chiral resolution step using tartaric acid derivatives. This resolution process is particularly problematic because it inherently limits the maximum theoretical yield to 50%, as the unwanted enantiomer is discarded or requires costly recycling processes. Furthermore, the use of stoichiometric amounts of copper salts and the generation of significant wastewater during the resolution and hydrolysis steps create substantial environmental compliance challenges. The cumulative effect of these four or more reaction steps results in a complex operational workflow that increases the risk of impurity accumulation and drives up the overall cost of goods sold, making it less attractive for modern, high-volume manufacturing environments.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a direct and elegant two-step synthesis that bypasses the need for chiral resolution entirely. The process initiates with an asymmetric Henry reaction (nitroaldol addition) between p-methylsulfonylbenzaldehyde and nitroethanol, catalyzed by a chiral copper complex formed from Cu(OTf)2 and a specific ligand L. This single step establishes both the carbon-carbon bond and the requisite stereocenters with high fidelity, effectively compressing what was previously a multi-stage sequence into a single transformation. Following this, a straightforward catalytic hydrogenation reduces the nitro group to the primary amine, yielding the target intermediate with exceptional optical purity. This reduction in step count not only simplifies the process flow but also dramatically improves the overall mass balance of the production line.
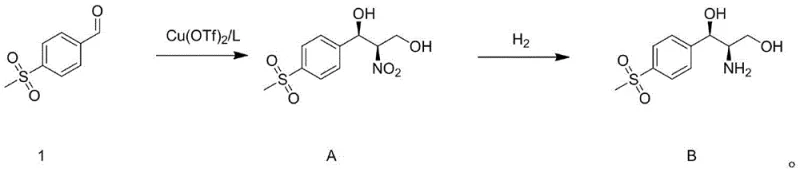
Mechanistic Insights into Cu(OTf)2-Catalyzed Asymmetric Henry Reaction
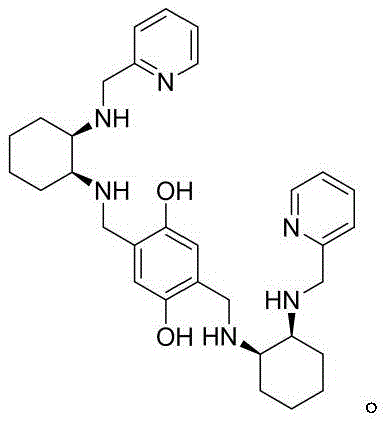
The success of this novel synthetic route hinges on the precise design and function of the chiral copper catalyst system. The catalyst is generated in situ by refluxing Copper(II) triflate (Cu(OTf)2) with a chiral ligand L, which features a rigid backbone derived from cyclohexanediamine and pyridine moieties. This specific ligand architecture creates a well-defined chiral pocket around the copper center, which acts as a Lewis acid to activate the aldehyde substrate. Simultaneously, the ligand environment directs the approach of the nitroethanol nucleophile, ensuring that the addition occurs exclusively from one face of the planar aldehyde. This stereochemical control is critical for achieving the high enantiomeric excess (ee) values reported, often exceeding 95%, which eliminates the need for downstream purification to remove the wrong enantiomer. The use of Cu(OTf)2 rather than simple copper salts enhances the Lewis acidity and solubility in organic solvents, further boosting the reaction rate and selectivity.
Following the asymmetric addition, the resulting nitro-diol intermediate undergoes a catalytic hydrogenation to reveal the final amino-diol structure. This step utilizes palladium on carbon (Pd/C) as a heterogeneous catalyst, which is a standard and robust choice for industrial reduction reactions. The mechanism involves the adsorption of hydrogen gas and the nitro compound onto the palladium surface, where the nitro group is sequentially reduced through nitroso and hydroxylamine intermediates to the primary amine. Crucially, the reaction conditions are mild, operating at moderate temperatures between 45-60°C and pressures of 50-60 psi, which minimizes the risk of side reactions such as over-reduction or dehalogenation (though no halogens are present here, the principle of mildness applies to preserving the sulfone group). The simplicity of filtering off the solid Pd/C catalyst after the reaction ensures that the final product stream is free from heavy metal contamination, a key requirement for pharmaceutical intermediates intended for veterinary use.
How to Synthesize Florfenicol Intermediate Efficiently
For technical teams looking to implement this synthesis, the process is designed to be operationally simple while maintaining rigorous control over critical parameters. The preparation begins with the formation of the active catalyst species, followed by the addition of the aldehyde and nitroalkane substrates in a polar protic solvent like methanol or ethanol. The reaction proceeds under stirring at controlled temperatures, after which the solvent is removed, and the crude nitro-intermediate is isolated. This intermediate is then subjected to hydrogenation in a standard pressure vessel. While the general workflow is straightforward, adherence to the specific molar ratios of catalyst components and reaction times is essential to replicate the high yields and purity described in the patent literature. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility.
- Prepare the chiral copper catalyst by refluxing Cu(OTf)2 with Ligand L in a toluene/alcohol mixture to form the active complex.
- Perform the asymmetric Henry reaction by mixing p-methylsulfonylbenzaldehyde and nitroethanol with the catalyst in methanol or ethanol at 20-80°C.
- Subject the resulting nitro-diols intermediate to catalytic hydrogenation using Pd/C in alcohol solvent at 50-60 psi to yield the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this two-step catalytic route offers transformative advantages over legacy manufacturing methods. The elimination of the chiral resolution step alone represents a massive gain in material efficiency, effectively doubling the theoretical yield from the starting aldehyde compared to resolution-based routes. This improvement in atom economy translates directly into a reduction in the volume of raw materials required per kilogram of finished product, thereby lowering the variable costs associated with production. Furthermore, the simplification of the process flow from four or more steps down to just two significantly reduces the operational overhead, including labor, energy consumption, and equipment occupancy time. These factors combine to create a more resilient and cost-effective supply chain capable of responding rapidly to fluctuations in market demand for veterinary antibiotics.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in waste and the elimination of expensive resolution agents. By avoiding the 50% material loss inherent in racemic resolution, the effective cost of the starting material per unit of product is significantly lowered. Additionally, the use of catalytic amounts of copper and ligand, rather than stoichiometric reagents, reduces the cost of goods related to chemicals. The simplified workup procedures, which avoid complex extractions and multiple crystallizations associated with resolution, further decrease utility costs and waste disposal fees. This holistic reduction in operational complexity allows for a much leaner manufacturing cost structure, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials such as p-methylsulfonylbenzaldehyde and nitroethanol ensures a robust supply chain foundation. Unlike specialized chiral pool starting materials that may suffer from supply constraints or price volatility, these commodity chemicals are produced at scale globally. The robustness of the catalytic system, which tolerates a range of reaction conditions without significant loss of performance, adds another layer of reliability, reducing the risk of batch failures. This stability allows supply chain managers to forecast production timelines with greater accuracy and maintain consistent inventory levels, mitigating the risk of stockouts that can disrupt the broader pharmaceutical manufacturing schedule.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations such as stirred tank reactors and filtration units that are common in fine chemical plants. The absence of cryogenic conditions or extreme pressures means that existing infrastructure can often be adapted for this synthesis without major capital expenditure. From an environmental standpoint, the reduction in wastewater generation, particularly from copper salts and resolution byproducts, aligns with increasingly stringent global environmental regulations. The green chemistry principles embedded in this route, such as high atom economy and catalytic reagent use, facilitate easier regulatory approval and reduce the environmental footprint of the manufacturing site, enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
To assist technical and procurement stakeholders in evaluating this technology, we have compiled answers to common questions regarding the synthesis and quality attributes of the intermediate. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is crucial for assessing the feasibility of integrating this intermediate into your specific supply chain or R&D pipeline. The following responses address key concerns regarding purity, scalability, and process robustness.
Q: How does the new copper-catalyzed route improve atom economy compared to traditional methods?
A: The new route eliminates the need for chiral resolution, which traditionally discards 50% of the material as the unwanted enantiomer, thereby theoretically doubling the atom utilization efficiency.
Q: What are the critical reaction conditions for maintaining high optical purity?
A: Maintaining the specific molar ratio of the copper salt to the chiral ligand and controlling the reaction temperature between 20-80°C are essential for achieving ee values exceeding 95%.
Q: Is the hydrogenation step scalable for industrial production?
A: Yes, the hydrogenation uses standard Pd/C catalysts under moderate pressure (50-60 psi) and temperature (45-60°C), making it highly suitable for large-scale reactor operations without specialized cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of veterinary pharmaceuticals, and we are uniquely positioned to support your needs with this advanced synthesis technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of florfenicol intermediate meets the highest standards of optical purity and chemical integrity. Our commitment to excellence ensures that our partners receive a product that is not only cost-effective but also fully compliant with global regulatory requirements for veterinary drug manufacturing.
We invite you to collaborate with us to leverage this innovative synthetic route for your production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, high-quality supply of this essential veterinary intermediate.
