Scalable Cyclization Technology for High-Purity Substituted Benzothiazole Derivatives
Scalable Cyclization Technology for High-Purity Substituted Benzothiazole Derivatives
The pharmaceutical industry continuously seeks robust, scalable methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical intermediates for central nervous system therapeutics. Patent CN1738808A introduces a transformative cyclization process for the preparation of amino-substituted benzothiazole derivatives of formula (I), which function as potent adenosine receptor ligands. This technology addresses long-standing challenges in heterocyclic synthesis by replacing costly transition metal catalysis with a highly efficient oxidative cyclization system utilizing dimethyl sulfoxide (DMSO) and hydrobromic acid (HBr). The ability to generate these pharmacologically active cores with yields reaching up to 90% represents a significant leap forward in process chemistry, offering a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates.
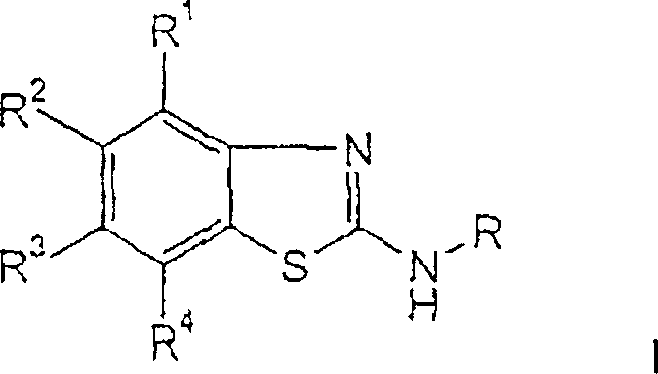
These benzothiazole derivatives are not merely academic curiosities; they are pivotal building blocks for drugs targeting Alzheimer's disease, Parkinson's disease, and various psychiatric disorders. The structural versatility allowed by the substituents R1 through R4 enables fine-tuning of pharmacokinetic properties, making this scaffold highly desirable for medicinal chemistry programs. By securing a supply chain based on this improved cyclization method, manufacturers can ensure the consistent availability of high-purity pharmaceutical intermediates required for late-stage clinical trials and eventual market launch.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing benzothiazole derivatives have been plagued by significant economic and technical inefficiencies that hinder large-scale manufacturing. For instance, earlier approaches described in WO01/97786 relied heavily on palladium-catalyzed coupling reactions involving expensive ligands such as BDCP and reagents like Pd(OAc)2. These transition metal systems not only inflate the raw material costs drastically but also introduce severe purification bottlenecks, often necessitating chromatographic separation to remove trace metal contaminants to meet stringent regulatory standards for active pharmaceutical ingredients. Furthermore, alternative halogen-mediated cyclizations using bromine in chloroform or acetic acid frequently suffered from low yields due to competitive side reactions on the electron-rich amino-substituted benzene ring, leading to unpredictable impurity profiles that complicate downstream processing.
The Novel Approach
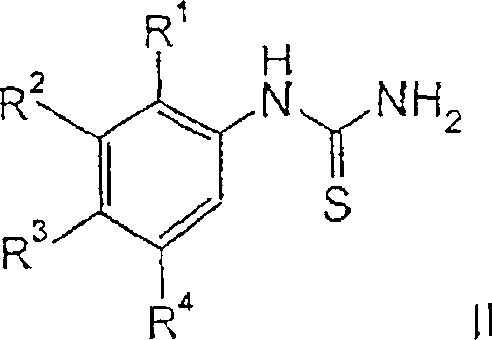
The innovative process disclosed in CN1738808A circumvents these historical obstacles by employing a sulfoxide-mediated oxidative cyclization that operates under mild yet effective conditions. By treating thiourea precursors of formula II or III with a combination of DMSO and HBr in a suitable solvent like ethyl acetate or acetic acid, the reaction achieves rapid ring closure with minimal by-product formation. This novel approach eliminates the dependency on precious metals entirely, thereby simplifying the waste stream and reducing the environmental footprint of the manufacturing process. The strategic selection of solvents, particularly ethyl acetate, exploits the solubility characteristics of the intermediate HBr salts to drive the reaction equilibrium forward and facilitate isolation through simple filtration, marking a substantial improvement in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into DMSO/HBr Mediated Oxidative Cyclization
The mechanistic elegance of this transformation lies in the dual role of the reagent system, where DMSO acts as a soft oxidant to activate the sulfur atom of the thiourea moiety while HBr serves as both a proton source and a nucleophilic promoter. Upon mixing, the sulfur atom of the thiourea is activated by the sulfoxide, generating a reactive sulfonium-like species that is highly susceptible to intramolecular nucleophilic attack by the ortho-position of the aromatic ring. The presence of HBr ensures the protonation of the leaving group and stabilizes the developing positive charge during the cyclization event, effectively lowering the activation energy barrier for ring closure. This mechanism avoids the radical pathways often associated with halogen-based oxidants, thereby suppressing the formation of poly-halogenated impurities that typically degrade the quality of the final product.
Furthermore, the choice of solvent plays a critical role in controlling the reaction trajectory and impurity profile. When the reaction is conducted in ethyl acetate, the resulting benzothiazole hydrobromide salt exhibits extremely low solubility, causing it to precipitate out of the solution almost immediately upon formation. This precipitation effect serves as a thermodynamic sink that drives the reaction to completion according to Le Chatelier's principle while simultaneously protecting the product from further degradation or dimerization reactions that might occur if it remained in solution at elevated temperatures. This phenomenon is particularly advantageous when dealing with substrates containing basic amine functionalities, as the acidic environment ensures these groups remain protonated and unreactive towards electrophilic side reactions, ensuring high chemoselectivity throughout the synthesis.
How to Synthesize Substituted Benzothiazole Derivatives Efficiently
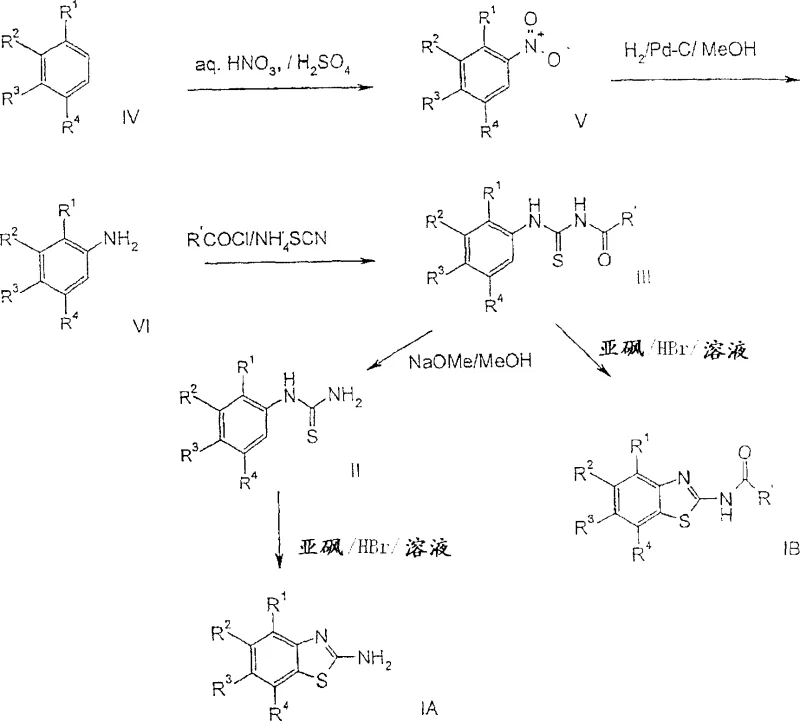
The practical implementation of this cyclization technology involves a straightforward sequence of operations that can be easily adapted for multi-kilogram production scales without requiring specialized equipment. The process begins with the suspension of the thiourea precursor in ethyl acetate, followed by the controlled addition of hydrobromic acid and dimethyl sulfoxide under reflux conditions. Detailed operational parameters regarding temperature gradients, addition rates, and workup procedures are critical for maximizing yield and purity, and the standardized synthesis steps see the guide below for comprehensive technical instructions.
- Preparation of Thiourea Precursor: React substituted aniline with benzoyl isothiocyanate or ammonium thiocyanate/benzoyl chloride to form the N-benzoyl thiourea intermediate.
- Cyclization Reaction: Suspend the thiourea in ethyl acetate or acetic acid, add HBr solution, and treat with DMSO under reflux conditions to induce ring closure.
- Isolation and Purification: Cool the reaction mixture, filter the precipitated HBr salt, and basify with aqueous ammonia to isolate the free base benzothiazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMSO/HBr cyclization protocol offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of palladium catalysts and exotic ligands removes a major source of supply chain volatility and cost fluctuation, as these precious metals are subject to significant market price swings and geopolitical sourcing risks. By shifting to commodity chemicals like DMSO, HBr, and ethyl acetate, manufacturers can lock in stable pricing structures and ensure long-term supply continuity, which is essential for maintaining uninterrupted production schedules for critical drug substances.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated scavenging resins required for metal removal leads to a drastic simplification of the bill of materials. Additionally, the high crude purity achieved through precipitation allows for the bypassing of resource-intensive chromatographic purification steps, significantly lowering solvent consumption and labor costs per kilogram of output. This streamlined workflow translates directly into improved gross margins for the final active pharmaceutical ingredient, providing a competitive edge in price-sensitive generic markets.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk-grade reagents ensures that production is not bottlenecked by the lead times of specialized fine chemicals. Since DMSO and hydrobromic acid are produced globally in massive quantities for various industrial applications, the risk of supply disruption is minimized. This robustness allows for more accurate demand forecasting and inventory planning, enabling the supply chain team to respond agilely to sudden increases in market demand without the fear of raw material shortages.
- Scalability and Environmental Compliance: The exothermic nature of the reaction is manageable through controlled addition rates, and the use of ethyl acetate, a Class 3 solvent with low toxicity, aligns well with modern green chemistry initiatives and regulatory guidelines regarding residual solvents. The simplified workup procedure generates less hazardous waste compared to heavy metal-catalyzed processes, reducing the burden on wastewater treatment facilities and lowering the overall environmental compliance costs associated with manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this cyclization technology, derived directly from the experimental data and embodiments provided in the patent literature. Understanding these nuances is vital for process engineers aiming to transfer this laboratory-scale success into a validated commercial manufacturing process.
Q: What are the primary advantages of the DMSO/HBr cyclization method over palladium-catalyzed routes?
A: The DMSO/HBr method eliminates the need for expensive palladium catalysts and specialized ligands, significantly reducing raw material costs and removing the requirement for chromatographic purification steps often needed to remove metal residues.
Q: How does this process handle impurities during large-scale production?
A: By utilizing ethyl acetate as a solvent, the process leverages the low solubility of the product HBr salt to precipitate it directly from the reaction mixture, effectively separating it from soluble by-products and preventing competitive dimerization reactions.
Q: Can this cyclization method be applied to substrates with basic amine substituents?
A: Yes, the protocol is effective for substrates containing basic units attached to the aromatic ring, provided that sufficient equivalents of HBr (typically 2 equivalents) are used to ensure complete conversion and salt formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust process chemistry in the successful development of CNS therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and compliant with cGMP standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzothiazole intermediate meets the exacting requirements of our global partners.
We invite you to collaborate with us to leverage this advanced cyclization technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your drug development timeline while optimizing your overall cost structure.
