Scalable Enantioselective Synthesis of Non-Steroidal Antiestrogen Intermediates for Commercial Production
The pharmaceutical industry constantly seeks robust manufacturing pathways for complex active pharmaceutical ingredients (APIs), particularly non-steroidal antiestrogens which play a critical role in oncology therapeutics. Patent CN1146554C presents a groundbreaking advancement in this domain by disclosing a short, practical, and commercially viable process for the efficient enantioselective synthesis of compounds of Formula I and Formula XIV. These compounds, characterized by their specific 2H-benzopyran structures, represent high-value intermediates requiring stringent stereochemical control. The invention addresses the critical gap between laboratory-scale feasibility and industrial-scale reality, offering a methodology that prioritizes high chemical purity and enantiomeric excess without relying on expensive or impractical purification techniques. By leveraging novel dynamic resolution strategies and optimized reaction conditions, this technology provides a reliable foundation for the mass production of potent antiestrogenic agents.
Historically, the synthesis of these complex benzopyran derivatives has been plagued by significant inefficiencies that hindered large-scale adoption. Conventional methods, as documented in earlier literature such as J. Med. Chem. 1997, often resulted in mixtures of cis and trans isomers alongside E and Z chalcones, necessitating laborious chromatographic separations that are economically unfeasible for commercial manufacturing. Furthermore, traditional routes frequently employed hazardous chlorinated solvents like dichloromethane and dimethylformamide, posing severe environmental and safety challenges for supply chain management. The novel approach detailed in this patent fundamentally re-engineers the synthetic pathway to circumvent these bottlenecks. By utilizing specific hindered organic amine bases and optimizing solvent systems, the new method drives equilibrium reactions towards the desired trans-isomer with exceptional selectivity. This shift not only eliminates the need for column chromatography but also ensures that the final product is substantially free of undesirable geometric isomers, thereby streamlining the entire production workflow.
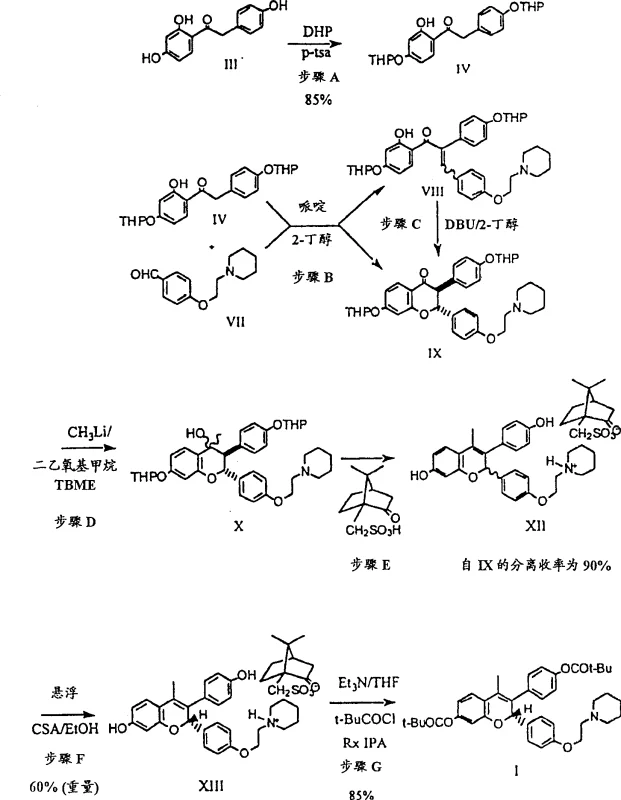
The mechanistic sophistication of this synthesis lies in its ability to manipulate chemical equilibria and stereochemistry simultaneously. A pivotal aspect of this technology is the dynamic resolution step, where a racemic mixture of diastereomeric salts is converted into a single, optically pure species. Unlike static resolution which theoretically caps yield at 50%, this process facilitates the in situ racemization of the unwanted diastereomer, allowing it to convert into the desired configuration under acidic catalysis. This mechanism ensures that the theoretical yield can exceed traditional limits while maintaining an optical purity of greater than 99% enantiomeric excess. Additionally, the initial cyclization steps are carefully controlled to prevent the formation of 1,4-addition byproducts during methylation, a common pitfall in organometallic chemistry. The use of specific ether solvents like tert-butyl methyl ether suppresses side reactions, ensuring that the carbon-carbon bond formation occurs exclusively at the carbonyl center.
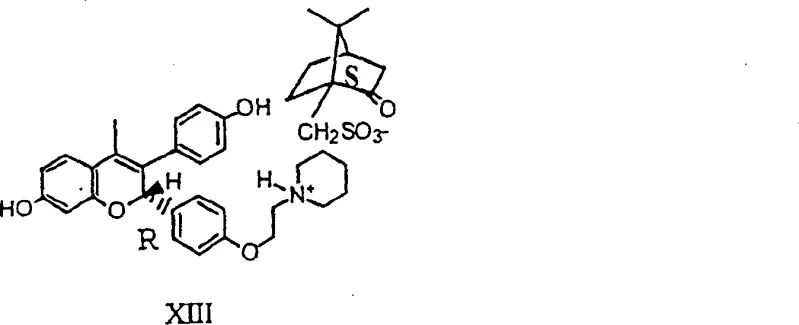
For research and development teams aiming to replicate or adapt this synthesis, understanding the precise sequence of operations is paramount to achieving the reported high yields and purity profiles. The process involves a series of interconnected transformations starting from protected phenolic precursors, moving through a critical Knoevenagel condensation, and culminating in a stereoselective resolution. Each step requires careful control of temperature, stoichiometry, and solvent composition to maintain the integrity of the sensitive intermediates. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for executing this complex synthesis in a controlled environment. Adherence to these parameters is essential for minimizing impurity formation and maximizing the efficiency of the dynamic resolution phase.
From a procurement and supply chain perspective, the adoption of this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The elimination of chromatographic purification represents a substantial reduction in manufacturing costs, as it removes the need for expensive silica gel, large volumes of elution solvents, and the associated waste disposal fees. Moreover, the substitution of hazardous chlorinated solvents with safer alternatives like ethanol and tert-butyl methyl ether simplifies regulatory compliance and reduces the risk profile associated with chemical handling and storage. This transition to greener chemistry not only aligns with modern environmental standards but also enhances supply chain reliability by utilizing readily available, commodity-grade reagents that are less susceptible to market volatility. The robustness of the crystallization steps further ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed purity specifications.
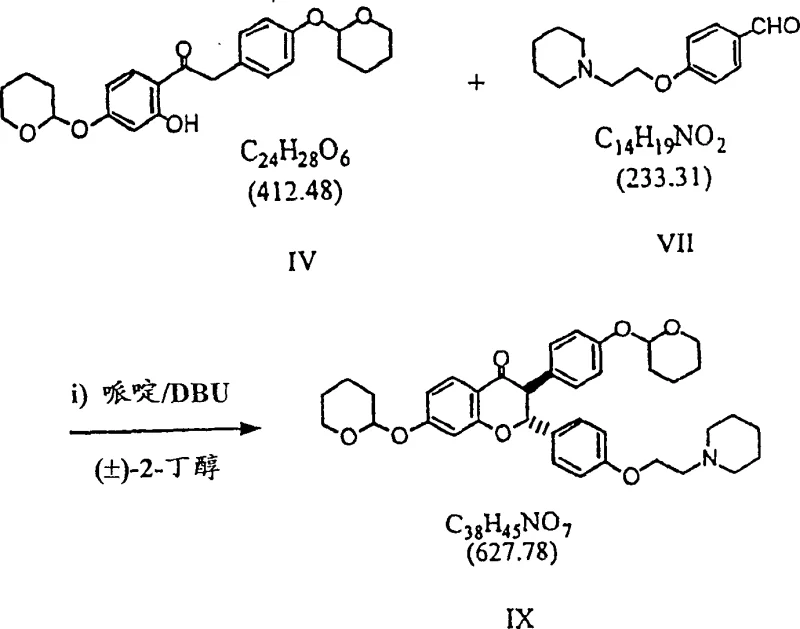
The scalability of this process is another critical factor for supply chain heads managing the transition from pilot plant to commercial production. The methodology is explicitly designed to avoid unit operations that do not translate well to large reactors, such as flash chromatography or complex distillations of heat-sensitive materials. Instead, the reliance on filtration and crystallization allows for the use of standard stainless steel equipment found in most multipurpose chemical manufacturing facilities. This compatibility significantly reduces the capital expenditure required for technology transfer and accelerates the time-to-market for new drug formulations. Furthermore, the high chemical purity achieved through crystallization minimizes the burden on downstream processing, ensuring that the final API meets stringent pharmacopeial standards with minimal additional refinement. This operational efficiency translates into a more agile supply chain capable of responding rapidly to fluctuating market demands.
Frequently asked questions regarding this technology often center on the specific improvements over legacy methods and the practical implications for quality control. Stakeholders are particularly interested in how the dynamic resolution mechanism functions to enhance yield beyond theoretical limits and what specific analytical methods are employed to verify the high enantiomeric excess. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers below address these core inquiries by drawing directly from the technical disclosures within the patent, providing clarity on the performance metrics and operational boundaries of this advanced synthetic route.
- Perform Knoevenagel condensation of protected acetophenone and aldehyde in 2-butanol with DBU to form trans-chromanone.
- Methylate the chromanone using methyllithium in TBME/DEM at low temperature to minimize 1,4-addition byproducts.
- Execute dynamic resolution using (S)-camphorsulfonic acid in ethanol to isolate the single S,S-diastereomeric salt.
Frequently Asked Questions (FAQ)
Q: How does this process improve upon prior art synthesis methods?
A: Unlike prior art which relied on costly chromatography and hazardous solvents like dichloromethane, this patent utilizes crystallization-driven purification and dynamic resolution, significantly enhancing chemical purity and operational safety.
Q: What is the optical purity achieved in the final product?
A: The described dynamic resolution method achieves an optical purity greater than 99% enantiomeric excess (de), surpassing the 92% de typically found in conventional resolution techniques.
Q: Can this synthesis be scaled for industrial manufacturing?
A: Yes, the process is specifically designed for industrial scale-up by avoiding impractical separation techniques and utilizing robust crystallization steps that ensure consistent quality from kilogram to tonne production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Formula I Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a supply chain for high-value pharmaceutical intermediates that combines technical excellence with commercial viability. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN1146554C are executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess required for antiestrogenic compounds. We understand that in the competitive landscape of oncology therapeutics, the reliability of your intermediate supply is non-negotiable, and we are committed to delivering products that meet the highest global standards.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities can align with your specific project requirements. Whether you are looking to optimize an existing supply chain or develop a new sourcing strategy for complex benzopyran derivatives, we offer a Customized Cost-Saving Analysis to demonstrate the tangible economic benefits of our manufacturing processes. We encourage you to request specific COA data and route feasibility assessments to validate our technical claims and explore the potential for long-term collaboration. By partnering with us, you gain access to a dedicated team focused on driving efficiency and quality in the production of critical pharmaceutical intermediates.
