Advanced Synthesis of 1,3,4-Oxadiazole Derivatives for Elastase Inhibitor Production
Advanced Synthesis of 1,3,4-Oxadiazole Derivatives for Elastase Inhibitor Production
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic intermediates, particularly those serving as critical building blocks for protease inhibitors. Patent CN1422261A introduces a groundbreaking methodology for the production of 1,3,4-oxadiazole derivatives, specifically targeting compounds useful as intermediates for serine protease inhibitors, such as elastase inhibitors. This technology addresses long-standing inefficiencies in heterocyclic chemistry by replacing cumbersome multi-step sequences with a streamlined, Lewis acid-catalyzed acylation protocol. The innovation lies not merely in the creation of new chemical entities but in the fundamental re-engineering of the synthetic route to enhance industrial viability. By shifting reaction conditions from extreme cryogenic temperatures to mild ambient ranges, this patent provides a blueprint for more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO98/24806 and WO00/55145, have historically relied on intricate synthetic sequences that pose significant challenges for industrial scale-up. Traditional routes often necessitate up to ten distinct reaction steps to arrive at the target 1,3,4-oxadiazole scaffold, inherently compounding yield losses and increasing waste generation. Furthermore, these legacy processes frequently demand rigorous cryogenic conditions, typically ranging from -78°C to -20°C, to control reactivity and prevent decomposition. Such low-temperature requirements impose severe constraints on manufacturing infrastructure, necessitating specialized cooling equipment and driving up operational expenditures. Additionally, the direct acylation of 1,3,4-oxadiazole rings with acid halides has been notoriously difficult to achieve with high selectivity, often resulting in poor yields and complex impurity profiles that complicate downstream purification efforts.
The Novel Approach
In stark contrast, the methodology described in CN1422261A utilizes a novel intermediate, represented by general formula (I), to bypass these historical bottlenecks. The core innovation involves a direct coupling reaction between a specific acyl chloride (Formula II) and a 1,3,4-oxadiazole derivative (Formula III) under mild thermal conditions. As illustrated in the reaction scheme below, this approach drastically reduces the step count and eliminates the need for energy-intensive cryogenic cooling.
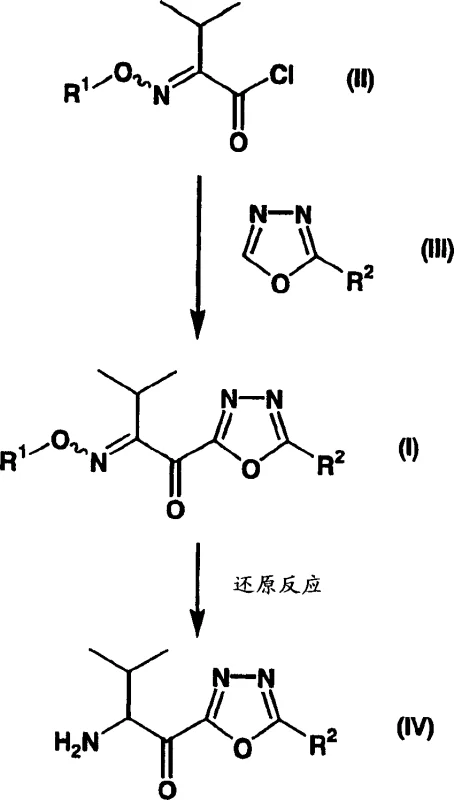
The new process operates effectively at temperatures between 0°C and 30°C, utilizing a sophisticated Lewis acid catalytic system to drive the reaction to completion with high efficiency. This shift allows for the use of standard reactor vessels and simplifies temperature control protocols, making the synthesis far more accessible for commercial production. By stabilizing the transition state through Lewis acid coordination, the method suppresses side reactions that typically plague heterocyclic acylations, thereby delivering the target 1,3,4-oxadiazole derivatives in substantially higher purity and yield compared to conventional techniques.
Mechanistic Insights into Lewis Acid-Catalyzed Acylation
The success of this synthetic route hinges on the precise activation of the electrophilic species within the reaction matrix. The mechanism involves the interaction of the acyl chloride (Formula II) with a Lewis acid system, typically comprising trimethylsilyl chloride (TMSCl) and an iodide source such as sodium iodide, in the presence of a tertiary amine base. This catalytic ensemble serves to enhance the electrophilicity of the carbonyl carbon, facilitating a nucleophilic attack by the nitrogen or carbon center of the 1,3,4-oxadiazole ring. The presence of the iodide ion is critical, as it likely generates a more reactive acyl iodide intermediate in situ, which is more susceptible to nucleophilic substitution than the corresponding chloride.
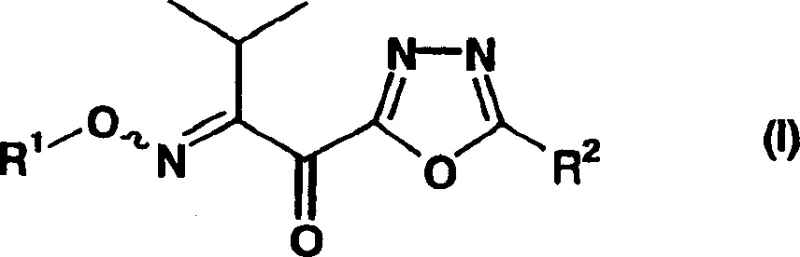
Furthermore, the steric environment of the 1,3,4-oxadiazole ring is carefully managed through the selection of substituents at the R2 position, which can range from tert-butyl groups to phenyl or cyclopropyl moieties. The reaction conditions are tuned to accommodate these variations without compromising the integrity of the heterocyclic core. Following the formation of the ketone intermediate (Formula I), the process proceeds to a reduction step to generate the final amine (Formula IV). This reduction, often achieved via catalytic hydrogenolysis using palladium-carbon or platinum-carbon catalysts, cleaves the protecting groups and reduces the imino functionality. The ability to carry out these transformations sequentially without isolating unstable intermediates contributes significantly to the overall robustness of the process, ensuring a clean impurity profile suitable for pharmaceutical applications.
How to Synthesize 1,3,4-Oxadiazole Derivatives Efficiently
The practical execution of this synthesis requires careful attention to reagent stoichiometry and solvent selection to maximize yield and purity. The process begins with the preparation of the activated acyl chloride, followed by the critical coupling step under Lewis acid catalysis. Detailed operational parameters, including specific molar ratios and workup procedures, are essential for reproducing the high efficiency reported in the patent literature. For R&D teams looking to implement this technology, understanding the nuances of the quenching and purification stages is vital to ensure the removal of residual metal catalysts and silane byproducts.
- Preparation of the acyl chloride intermediate (Formula II) from 3-methyl-2-(alkoxyimino)butanoic acid using thionyl chloride.
- Reaction of the acyl chloride with a 1,3,4-oxadiazole derivative (Formula III) in the presence of a Lewis acid (e.g., TMSCl/NaI) and tertiary amine at 0-30°C.
- Hydrogenolysis or catalytic reduction of the resulting ketone (Formula I) to yield the final amine intermediate (Formula IV).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers transformative benefits for procurement strategies and supply chain management within the pharmaceutical sector. The elimination of cryogenic reaction steps represents a direct reduction in capital expenditure and operational overhead, as facilities no longer require specialized low-temperature reactors or excessive energy consumption for cooling. This technological shift translates into a more resilient supply chain, less vulnerable to energy price fluctuations or equipment failures associated with extreme condition processing. Moreover, the reduced number of synthetic steps inherently lowers the consumption of raw materials and solvents, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers the cost of goods sold (COGS) by minimizing unit operations. By avoiding the use of expensive cryogenic infrastructure and reducing the total volume of solvents and reagents required per kilogram of product, manufacturers can achieve substantial cost savings. The high yield and selectivity of the Lewis acid-catalyzed step further reduce waste disposal costs and improve overall material efficiency, making the production of these elastase inhibitor intermediates economically superior to legacy methods.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as isobutyraldehyde and simple hydrazides, ensures a stable and secure supply of raw inputs. Unlike processes dependent on exotic or scarce reagents, this route utilizes commodity chemicals that are widely sourced, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that production can be maintained consistently across different manufacturing sites, ensuring reliable delivery schedules for downstream API producers and reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild operating conditions (0-30°C) and the use of standard organic solvents facilitate seamless scale-up from pilot plant to commercial tonnage production. This scalability is crucial for meeting the growing global demand for protease inhibitors without the need for prohibitive capital investment in new facilities. Additionally, the simplified workup and purification procedures reduce the generation of hazardous waste streams, aiding compliance with increasingly stringent environmental regulations and supporting sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this patented process is essential for stakeholders evaluating its potential integration into their supply chains. The following questions address common inquiries regarding the reaction mechanics, safety profile, and applicability of the technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of the CN1422261A synthesis method over prior art?
A: The primary advantage is the elimination of cryogenic reaction conditions (-78°C to -20°C) required by previous methods. This new process operates efficiently at 0-30°C, significantly reducing energy costs and equipment complexity while maintaining high yields.
Q: Which catalysts are essential for the acylation step in this novel route?
A: The process relies on a Lewis acid system, specifically utilizing trimethylsilyl chloride (TMSCl) in combination with an iodide salt (such as sodium iodide) and a tertiary amine like N-methylmorpholine or pyridine to activate the acyl chloride.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. By removing the need for complex low-temperature infrastructure and reducing the total number of synthetic steps compared to traditional routes, it offers superior operational feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4-Oxadiazole Derivatives Supplier
As the global demand for advanced protease inhibitors continues to rise, securing a dependable source of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in heterocyclic chemistry to deliver superior 1,3,4-oxadiazole derivatives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for drug substance manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall development costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your path to market.
