Advanced Manufacturing of Combretastatin Intermediates via Optimized Wittig Condensation and Iron Reduction
Introduction to Next-Generation Combretastatin Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for potent vascular disrupting agents, specifically combretastatin A-4 and its analogs, which exhibit remarkable anti-tumor activity. Patent CN1646476A introduces a transformative methodology for preparing these critical 1,2-stilbene derivatives, addressing long-standing inefficiencies in yield and purity that have plagued earlier production methods. By leveraging a refined Wittig condensation strategy coupled with a novel iron-based reduction protocol, this technology offers a pathway to high-purity intermediates essential for downstream drug development. The innovation lies not only in the chemical transformation but in the strategic replacement of hazardous reagents with more economical and environmentally benign alternatives, setting a new standard for scalable API intermediate production.
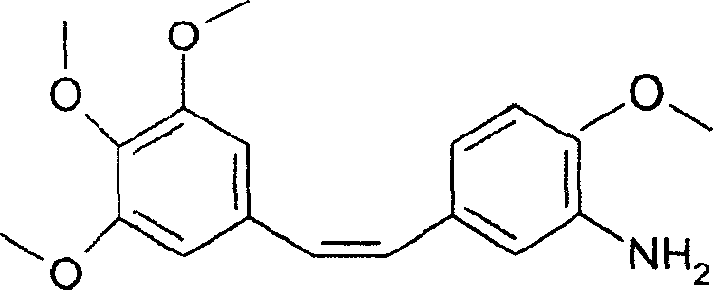
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of combretastatin derivatives relied heavily on zinc-mediated reduction of nitro-stilbene precursors, a process fraught with significant technical and economic drawbacks. As documented in prior art such as US Patent 5,525,632, the use of zinc in acetic acid often resulted in incomplete reactions and the formation of persistent azo-compound impurities due to the coupling of amino and nitroso intermediates. Furthermore, the yields were notoriously low, typically ranging between 21% and 33% based on the aldehyde starting material, necessitating large volumes of raw materials to produce modest quantities of product. The reliance on zinc also introduced substantial heavy metal waste streams, complicating environmental compliance and increasing the cost of waste treatment, while alternative catalytic hydrogenation methods risked saturating the critical ethylene double bond, destroying the biological activity of the molecule.
The Novel Approach
The methodology disclosed in CN1646476A fundamentally re-engineers this synthetic landscape by introducing two distinct, superior pathways that circumvent these historical bottlenecks. The first improvement involves substituting zinc with iron powder in a hydrochloric acid medium, which dramatically enhances the reduction yield to approximately 60% while effectively suppressing the formation of azo-byproducts. Even more revolutionary is the second approach, which utilizes pre-reduced amino-precursors—either 3-amino-4-methoxybenzaldehyde or the corresponding phosphonium salt—to perform a direct Wittig condensation. This eliminates the reduction step entirely, thereby removing the handling of potentially carcinogenic nitro-intermediates and streamlining the process into fewer operational units. This dual-pathway flexibility allows manufacturers to select the optimal route based on raw material availability and specific purity requirements.
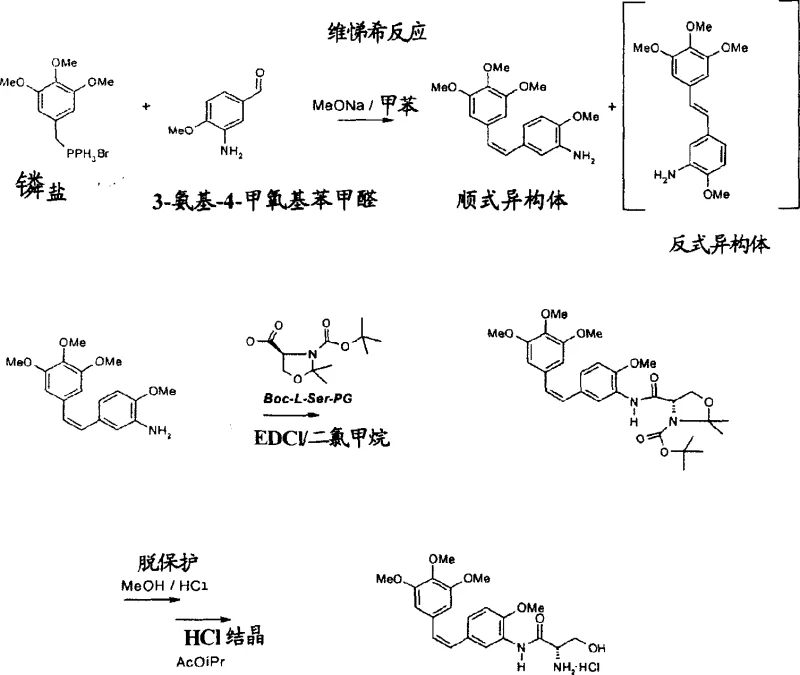
Mechanistic Insights into Optimized Wittig Condensation and Iron Reduction
The core of this technological advancement rests on the precise control of stereoselectivity during the olefin formation and the chemoselectivity during the functional group transformation. In the Wittig reaction phase, the use of sodium methoxide in toluene or THF at controlled low temperatures (0-10°C) favors the kinetic formation of the Z-isomer over the thermodynamically stable E-isomer, achieving Z/E ratios as high as 75/25. This stereocontrol is critical because the cis-configuration is requisite for the biological binding affinity of combretastatins to tubulin. Following the condensation, the novel iron reduction mechanism operates under mild acidic conditions where iron acts as a single-electron donor, selectively reducing the nitro group to an amine without affecting the sensitive stilbene double bond. This contrasts sharply with palladium or platinum-catalyzed hydrogenation, which often leads to over-reduction and loss of potency, ensuring that the structural integrity of the pharmacophore is maintained throughout the synthesis.
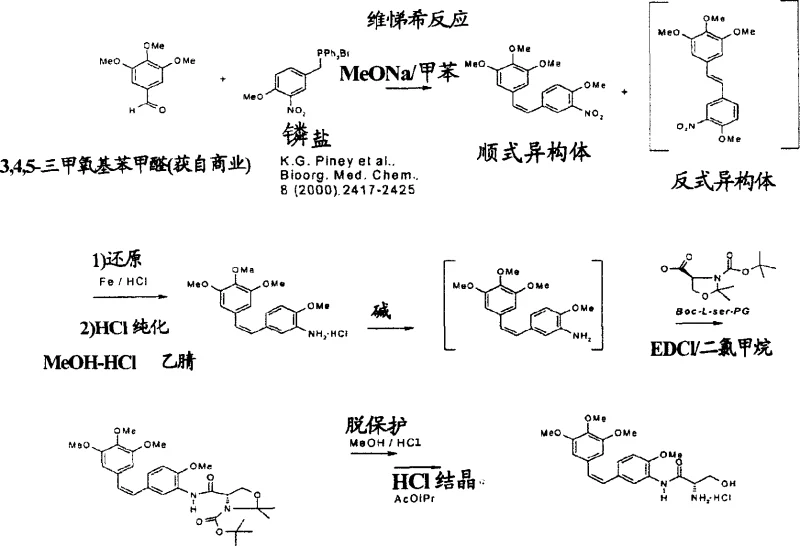
Furthermore, the purification mechanism leverages the differential solubility of the geometric isomers in specific solvent systems to achieve exceptional purity levels without resorting to costly preparative chromatography. By converting the crude amine mixture into its hydrochloride salt and utilizing solvent mixtures like methanol and acetonitrile, the process induces selective crystallization of the desired Z-isomer while keeping the E-isomer in solution. This crystallization-driven purification is a hallmark of robust industrial chemistry, as it replaces complex separation techniques with simple unit operations that are easily scalable. The result is a final product with purity exceeding 97% as determined by HPLC, meeting the stringent specifications required for pharmaceutical grade intermediates. This level of control over impurity profiles ensures that downstream coupling reactions with amino acids, such as L-serine, proceed with high efficiency and minimal side reactions.
How to Synthesize Combretastatin Intermediates Efficiently
The synthesis of these high-value intermediates follows a logical progression of olefination, reduction, and coupling steps that have been optimized for industrial feasibility. The process begins with the preparation of the phosphonium salt or the selection of the appropriate aldehyde partner, followed by the base-mediated condensation to establish the stilbene framework. Subsequent steps involve the careful management of the reduction environment to ensure complete conversion of the nitro group while preserving the alkene geometry. For a comprehensive understanding of the specific reagent quantities, temperature profiles, and workup procedures detailed in the patent examples, please refer to the standardized synthesis guide below.
- Perform Wittig condensation between trimethoxybenzyl phosphonium salts and methoxy-nitrobenzaldehyde (or amino-analogs) using sodium methoxide in toluene or THF to generate the stilbene backbone.
- If using nitro-precursors, conduct selective reduction of the nitro group to an amine using iron powder and hydrochloric acid in ethanol/water, avoiding double bond saturation.
- Purify the resulting Z-isomer rich mixture via crystallization of the hydrochloride salt, followed by coupling with protected L-serine to form the final amide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The shift from zinc to iron reduction represents a significant cost optimization opportunity, as iron is substantially less expensive than zinc and does not require the extensive waste remediation protocols associated with heavy metal disposal. Additionally, the ability to utilize direct amino-precursors removes an entire processing step, reducing energy consumption, solvent usage, and labor hours, which collectively contribute to a lower cost of goods sold. The simplified process flow also minimizes the risk of batch failures due to incomplete reactions or difficult purifications, ensuring a more predictable and reliable supply of critical intermediates for global drug development programs.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts like palladium or platinum, which were previously considered for reduction steps, drastically lowers the raw material costs associated with the synthesis. By relying on abundant and inexpensive iron powder and avoiding the need for complex chromatographic separations through effective crystallization strategies, the overall manufacturing expense is significantly reduced. This economic efficiency is further amplified by the higher yields achieved, meaning less starting material is wasted per kilogram of final product, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as 3,4,5-trimethoxybenzaldehyde and various methoxy-substituted benzyl derivatives, are commodity chemicals with established global supply chains, reducing the risk of procurement bottlenecks. The robustness of the iron reduction method ensures consistent batch-to-batch quality, which is essential for maintaining regulatory compliance and avoiding supply disruptions caused by out-of-specification products. Furthermore, the flexibility to switch between the nitro-reduction route and the direct amino-condensation route provides a strategic buffer against fluctuations in the availability of specific starting materials.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like toluene, ethanol, and dichloromethane that are easily handled in large-scale reactor systems. The replacement of zinc with iron significantly reduces the environmental footprint of the manufacturing process by minimizing toxic heavy metal waste, aligning with modern green chemistry principles and easing the burden on wastewater treatment facilities. This environmental compatibility facilitates smoother regulatory approvals and supports the sustainability goals of forward-thinking pharmaceutical companies seeking to reduce their ecological impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and process descriptions found within the patent documentation, providing clarity on yield expectations, impurity profiles, and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: How does the iron reduction method improve upon traditional zinc reduction for combretastatin synthesis?
A: The patented iron reduction method significantly increases yield from approximately 21-33% (with zinc) to around 60%, while eliminating the formation of difficult-to-remove azo-compound impurities and reducing heavy metal waste.
Q: What is the advantage of using amino-precursors in the Wittig reaction?
A: Using amino-precursors allows for a direct one-step condensation, bypassing the hazardous nitro-reduction stage entirely. This reduces the generation of CMR (Carcinogenic, Mutagenic, Reprotoxic) substances and simplifies the overall process safety profile.
Q: Can this process achieve high Z-isomer selectivity?
A: Yes, the optimized reaction conditions, particularly using sodium methoxide in toluene at controlled temperatures, favor the formation of the Z-isomer with ratios up to 75/25 (Z/E), which can be further enriched to >97% purity through selective crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Combretastatin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1646476A can be translated into reliable, large-scale manufacturing outcomes. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of combretastatin intermediate meets the exacting standards required by global regulatory authorities.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your project economics. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey from clinical development to commercial success with our proven expertise in complex organic synthesis.
