Advanced Polymer-Supported Synthesis of Combretastatin A-4 for Scalable Pharmaceutical Production
Advanced Polymer-Supported Synthesis of Combretastatin A-4 for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for potent anticancer agents, and Combretastatin A-4 stands out as a premier candidate due to its unique vascular disrupting capabilities. Patent CN100343216C introduces a groundbreaking polymer-supported synthesis method that addresses critical bottlenecks in the production of this high-value molecule. Unlike traditional solution-phase techniques that struggle with isomer separation and purification complexity, this novel approach leverages solid-phase chemistry to streamline the manufacturing workflow. By anchoring the starting material, isovanillin, onto a trityl chloride resin, the process facilitates easier handling of intermediates and significantly enhances the selectivity for the biologically active Z-isomer. This technological advancement represents a paradigm shift for manufacturers aiming to secure a reliable API intermediate supplier status, offering a pathway to higher purity and reduced operational costs without compromising on the structural integrity of the final pharmacophore.
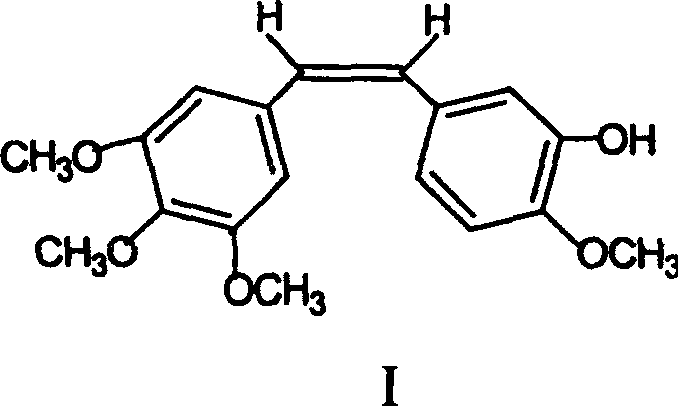
Furthermore, the clinical potential of Combretastatin A-4 has driven extensive research into its derivatives, such as the water-soluble prodrug Combretastatin A-4-P, to overcome solubility limitations. 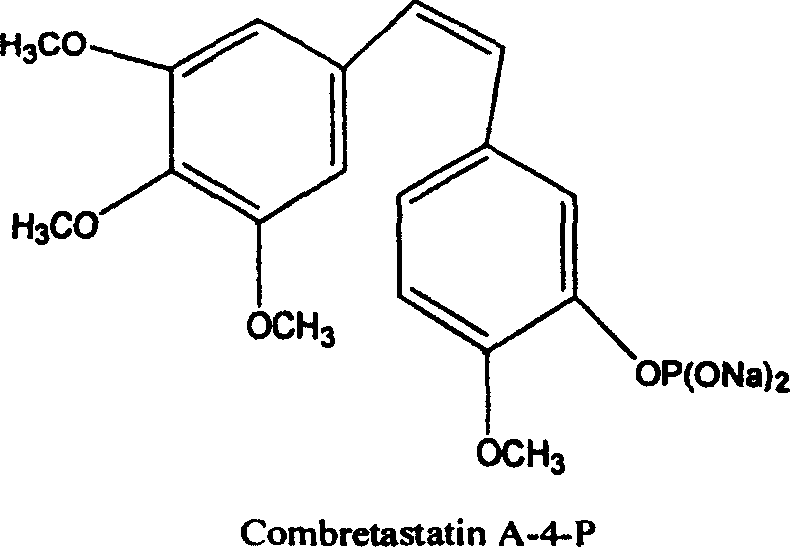 The ability to synthesize the parent compound efficiently is paramount for the subsequent production of these advanced therapeutic candidates. The polymer immobilization technique described in the patent not only simplifies the synthetic sequence but also aligns with modern green chemistry principles by reducing the reliance on hazardous solvents typically associated with extensive chromatographic purification. For R&D directors and procurement specialists, understanding this methodology is crucial for evaluating supply chain resilience and cost-effectiveness in the competitive oncology market.
The ability to synthesize the parent compound efficiently is paramount for the subsequent production of these advanced therapeutic candidates. The polymer immobilization technique described in the patent not only simplifies the synthetic sequence but also aligns with modern green chemistry principles by reducing the reliance on hazardous solvents typically associated with extensive chromatographic purification. For R&D directors and procurement specialists, understanding this methodology is crucial for evaluating supply chain resilience and cost-effectiveness in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Combretastatin A-4 has been plagued by significant technical challenges that hinder efficient commercial scale-up of complex stilbenes. Conventional solution-phase methods often rely on protecting groups like t-BuMe2SiCl or trityl chloride in homogeneous systems, which necessitate rigorous purification steps to remove triphenylphosphine oxide and other phosphorus-containing byproducts generated during the Wittig reaction. These traditional routes frequently suffer from low Z-type yields, producing a mixture of cis and trans isomers that requires energy-intensive and solvent-heavy column chromatography for separation. Moreover, the harsh reaction conditions and the difficulty in isolating the pure cis-isomer result in extended production cycles and increased waste generation. For supply chain heads, these inefficiencies translate into unpredictable lead times and higher raw material costs, making it difficult to maintain a consistent supply of high-purity API intermediates required for clinical and commercial demands.
The Novel Approach
In stark contrast, the polymer-supported synthesis method outlined in patent CN100343216C offers a transformative solution by integrating the benefits of solid-phase synthesis with classical organic transformations. By immobilizing isovanillin onto a polystyrene-based trityl chloride resin, the intermediate becomes insoluble, allowing for the removal of soluble impurities through simple filtration and washing protocols. This approach effectively eliminates the need for column chromatography, which is often the most costly and time-consuming step in fine chemical manufacturing. The novel route ensures high Z-type selectivity, directly addressing the critical quality attribute of the drug substance. Additionally, the final cleavage step using acid hydrolysis releases the product in high purity, ready for simple recrystallization. This streamlined process not only enhances cost reduction in pharmaceutical manufacturing but also significantly improves the overall throughput, making it an ideal strategy for establishing a reliable API intermediate supplier network capable of meeting global demand.
Mechanistic Insights into Polymer-Supported Wittig Reaction
The core of this innovative synthesis lies in the strategic application of the Wittig reaction on a solid support, which fundamentally alters the reaction kinetics and thermodynamic profile compared to solution-phase counterparts. The process begins with the nucleophilic substitution of the phenolic hydroxyl group of isovanillin onto the trityl chloride resin under alkaline conditions, forming a stable ether linkage that anchors the aromatic ring to the polymer matrix. Subsequently, the phosphonium salt, 3,4,5-trimethoxy benzyl triphenylphosphonium bromide, is deprotonated by a strong base such as n-butyllithium at cryogenic temperatures (-30°C to -20°C) to generate the reactive ylide species. When this ylide reacts with the resin-bound aldehyde, the steric constraints imposed by the polymer backbone appear to favor the formation of the Z-alkene geometry, which is essential for the biological activity of Combretastatin A-4. This mechanistic advantage ensures that the resulting stilbene backbone maintains the correct spatial configuration required for binding to the colchicine site on tubulin, thereby inhibiting microtubule assembly and inducing vascular collapse in tumor tissues.
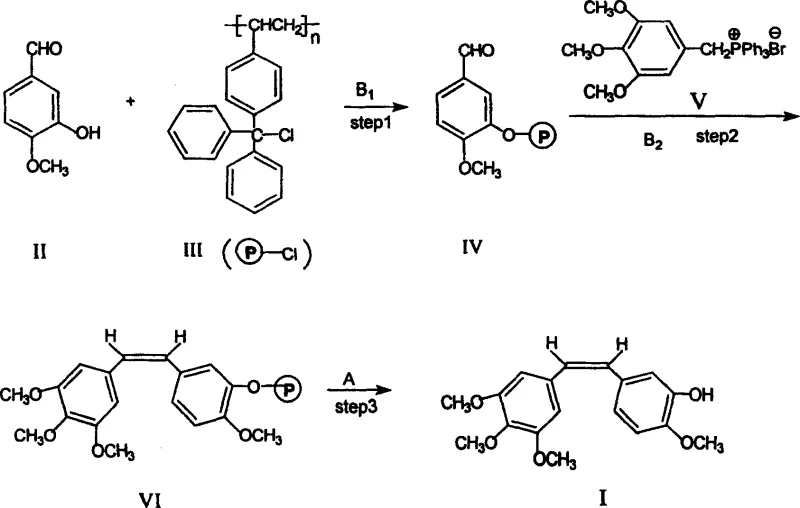
Following the carbon-carbon bond formation, the final step involves the acidic cleavage of the trityl ether linkage to release the free phenol. The use of concentrated hydrochloric acid or other mineral acids facilitates the hydrolysis of the trityl group, regenerating the 3-hydroxy functionality while simultaneously cleaving the molecule from the solid support. This step is critical for impurity control, as the triphenylmethanol byproduct derived from the resin remains in the aqueous or organic phase depending on the workup, while the desired product precipitates or can be extracted cleanly. The rigorous washing steps between each stage ensure that excess reagents and side products are removed before they can interfere with subsequent reactions, leading to a final product with exceptional purity profiles. For technical teams, this level of control over the reaction pathway minimizes the risk of genotoxic impurities and simplifies the validation process for regulatory filings, ensuring compliance with stringent international quality standards.
How to Synthesize Combretastatin A-4 Efficiently
The implementation of this polymer-supported route requires precise control over reaction parameters to maximize yield and stereochemical purity. The process is divided into three distinct operational stages: resin loading, Wittig coupling, and acidic cleavage. Each stage utilizes standard laboratory equipment such as three-necked flasks, dropping funnels, and filtration apparatus, making it highly adaptable for pilot plant operations. The initial protection step is conducted in tetrahydrofuran with triethylamine as a base, ensuring complete conversion of the starting material onto the resin. The subsequent Wittig reaction demands strict temperature control to maintain the stability of the ylide and prevent side reactions, while the final hydrolysis step utilizes mild heating to facilitate rapid cleavage without degrading the sensitive stilbene double bond. Detailed standardized synthesis steps are provided in the guide below to assist process chemists in replicating these results.
- Protect isovanillin using trityl chloride resin under alkaline conditions to form the polymer-bound intermediate.
- Perform a Wittig reaction with 3,4,5-trimethoxy benzyl triphenylphosphonium bromide using a strong base like n-butyllithium at low temperatures.
- Cleave the final product from the resin using acid hydrolysis, followed by filtration and recrystallization to obtain high-purity Combretastatin A-4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this polymer-supported technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of column chromatography represents a massive reduction in solvent consumption and waste disposal costs, which are major drivers of operational expenditure in fine chemical production. By replacing complex purification with simple filtration, the process drastically shortens the batch cycle time, allowing for faster turnover and improved responsiveness to market fluctuations. Furthermore, the high selectivity for the Z-isomer reduces the loss of valuable material associated with isomer separation, directly contributing to better atom economy and raw material utilization. These factors collectively enhance the economic viability of producing Combretastatin A-4, making it a more attractive asset for investment and long-term supply agreements.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the removal of chromatographic purification, which is notoriously expensive due to the high cost of silica gel and the large volumes of organic solvents required for elution. By utilizing a filter-and-wash protocol, manufacturers can significantly lower their utility and material costs, passing these savings on to customers or reinvesting them into capacity expansion. Additionally, the high yields reported in the patent examples suggest that less starting material is wasted, further optimizing the cost per kilogram of the final API intermediate. This efficiency is crucial for maintaining competitive pricing in the global pharmaceutical market while adhering to strict quality specifications.
- Enhanced Supply Chain Reliability: The simplicity of the unit operations involved in this synthesis route translates to greater robustness and reliability in the supply chain. Filtration and washing are scalable processes that are less prone to the variability and bottlenecks often encountered with chromatography columns, which can channel or clog at larger scales. This predictability ensures consistent delivery schedules and reduces the risk of stockouts, which is vital for clients relying on a steady flow of critical oncology intermediates. Moreover, the use of commercially available reagents like isovanillin and standard phosphonium salts ensures that raw material sourcing remains stable and unaffected by niche supply constraints.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with the industry's push towards greener manufacturing practices. The reduction in solvent waste lowers the burden on wastewater treatment facilities and minimizes the facility's carbon footprint. The process is inherently safer as well, as it avoids the handling of large quantities of fine silica dust and reduces the exposure of workers to potentially hazardous eluents. For companies aiming to meet ISO 14001 standards or other environmental certifications, adopting this cleaner synthesis route provides a clear pathway to compliance while demonstrating corporate responsibility to stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the polymer-supported synthesis of Combretastatin A-4. These insights are derived directly from the experimental data and claims presented in patent CN100343216C, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios. The answers highlight the specific advantages of the solid-phase approach over traditional methods, focusing on yield, purity, and operational simplicity.
Q: How does polymer support improve the Z-selectivity of Combretastatin A-4?
A: The polymer-supported method utilizes a trityl chloride resin which creates a specific steric environment during the Wittig reaction. This environment favors the formation of the cis-stilbene (Z-isomer), which is the biologically active conformation required for tubulin inhibition, significantly reducing the formation of the inactive E-isomer compared to traditional solution-phase methods.
Q: What are the purification advantages of this synthesis route?
A: Traditional synthesis often requires tedious column chromatography to separate the product from triphenylphosphine oxide and other byproducts. In this polymer-supported route, the intermediate remains bound to the solid support, allowing impurities to be removed via simple filtration and washing. The final cleavage step releases the pure product, drastically simplifying downstream processing.
Q: Is this method suitable for industrial scale-up?
A: Yes, the method is specifically designed for industrial applicability. By eliminating column chromatography and utilizing robust filtration steps for separation, the process reduces solvent consumption and processing time. The reported yields in the patent examples demonstrate high efficiency across multiple steps, making it a viable candidate for commercial scale-up of complex anticancer intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Combretastatin A-4 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We are committed to delivering Combretastatin A-4 with stringent purity specifications, utilizing advanced analytical techniques in our rigorous QC labs to verify Z-isomer content and impurity profiles. Our state-of-the-art facilities are equipped to handle polymer-supported chemistries safely and efficiently, guaranteeing a consistent supply of this valuable anticancer agent.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your project goals and accelerate your time to market.
