Advanced Quetiapine Manufacturing: Overcoming Stability and Scalability Challenges in API Production
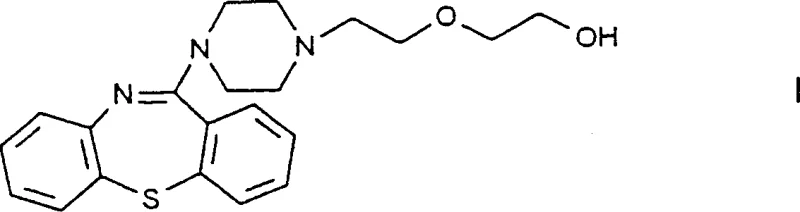
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-volume antipsychotic agents, and the synthesis of Quetiapine represents a critical area of process optimization. Patent CN1177839C introduces a transformative approach to producing 11-[4-[2-(2-hydroxyethoxy)ethyl]-1-piperazinyl]dibenzo[b,f]-1,4-thiazepine, commonly known as Quetiapine. This patent addresses significant historical bottlenecks in the production of this vital neuroleptic compound by redefining the intermediate chemistry. Unlike traditional methods that rely on unstable precursors, this innovation utilizes a novel haloethylpiperazinylthiazepine derivative that offers superior stability and ease of handling. The strategic shift in synthetic design not only enhances the chemical integrity of the process but also aligns perfectly with modern Good Manufacturing Practice (GMP) requirements for consistency and safety. For global procurement teams, understanding this technological leap is essential for securing a reliable supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
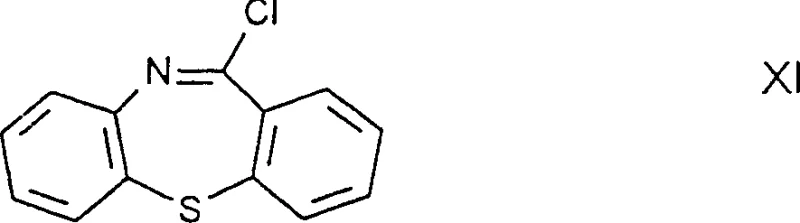
Historically, the industrial production of Quetiapine relied heavily on methodologies described in earlier patents such as EP240228, which utilized a gem-haloimine intermediate designated as Formula XI. This conventional pathway presented severe operational challenges, primarily due to the inherent instability of the gem-haloimine structure in the presence of moisture. When processed on a large industrial scale, these intermediates were prone to rapid hydrolysis in moist air, leading to significant degradation and the formation of hydrolysis byproducts that contaminated the final reaction mixture. Furthermore, the purification of the crude product obtained through this legacy method necessitated the use of silica gel column chromatography, a technique that is notoriously difficult to scale, expensive to operate, and inefficient for multi-ton production. The cumulative effect of these limitations resulted in a total yield of only approximately 62.6% calculated from the initial urethane derivative, rendering the process economically unattractive for high-volume manufacturing.
The Novel Approach
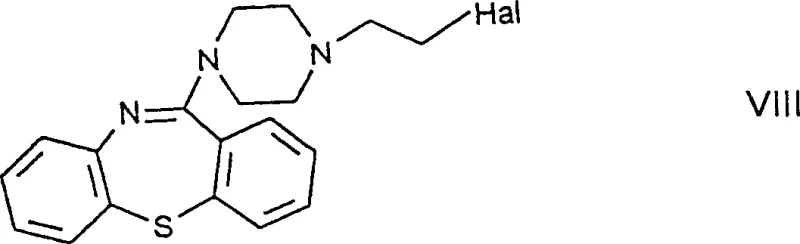
In stark contrast to the legacy techniques, the methodology disclosed in CN1177839C introduces a fundamentally different intermediate strategy centered around the haloethylpiperazinylthiazepine derivative, specifically Formula VIII. This new intermediate is chemically robust and does not suffer from the hydrolytic instability that plagued the previous gem-haloimine compounds. By reacting this stable haloethyl derivative directly with ethylene glycol, the process bypasses the need for complex and costly purification steps like column chromatography. Instead, the final product can be isolated and purified through standard crystallization techniques, which are far more amenable to industrial scale-up. This strategic modification allows for a total yield improvement to the range of 66-67%, while simultaneously simplifying the operational workflow. The ability to utilize stable intermediates means that raw materials can be stored and handled with greater flexibility, reducing the risk of batch failure due to environmental factors.
Mechanistic Insights into the Stabilized Cyclization and Substitution Pathway
The core chemical innovation lies in the construction of the dibenzothiazepine ring system coupled with the piperazine side chain in a manner that avoids labile functional groups. In the preferred embodiments, the synthesis involves the cyclization of a urethane derivative or an aminodiphenyl sulfide precursor using potent dehydrating and halogenating agents such as phosphorus oxychloride (POCl3) and phosphorus pentoxide (P2O5). This reaction conditions facilitate the formation of the rigid tricyclic thiazepine core while installing the chloroethyl group on the piperazine nitrogen. Unlike the gem-haloimine route where the halogen is attached to an imine carbon making it susceptible to nucleophilic attack by water, the halogen in Formula VIII is attached to a saturated ethyl chain. This structural difference is critical, as it renders the molecule inert to atmospheric moisture, thereby preserving the integrity of the intermediate throughout the manufacturing campaign. The subsequent step involves a classic Williamson ether synthesis where the chloroethyl group is displaced by the alkoxide of ethylene glycol.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions. In the old process, the hydrolysis of the gem-haloimine generated polar impurities that were difficult to separate without chromatography. In the new process, the primary byproducts are inorganic salts and excess reagents which can be easily removed via aqueous workup and phase separation. The use of ethylene glycol in excess acts both as a reagent and a solvent medium, driving the equilibrium towards the desired ether product. The reaction temperature is typically maintained around 100°C, ensuring complete conversion without inducing thermal degradation of the sensitive thiazepine ring. This controlled reactivity profile ensures that the impurity spectrum is predictable and manageable, allowing for the production of high-purity Quetiapine base that meets stringent pharmaceutical specifications without the need for extensive downstream processing.
How to Synthesize 11-[4-(2-Chloroethyl)piperazin-1-yl]-dibenzo[b,f]-1,4-thiazepine Efficiently
The synthesis of the key intermediate Formula VIII is the pivotal step that determines the success of the entire manufacturing campaign. The process begins with the reaction of a urethane derivative or an aminodiphenyl sulfide with a piperazine derivative, followed by a rigorous cyclization step. Detailed protocols indicate that mixing the precursor with phosphorus oxychloride and phosphorus pentoxide at reflux temperatures effectively closes the ring and installs the necessary chloroethyl functionality. This one-pot or two-step sequence is designed to maximize throughput while minimizing the handling of hazardous reagents. The resulting intermediate precipitates or can be extracted into organic solvents, ready for the final coupling reaction. For a comprehensive understanding of the specific molar ratios, temperature profiles, and workup procedures required to replicate this high-yielding transformation, operators should refer to the standardized synthesis guidelines provided below.
- Preparation of the stable intermediate 11-[4-(2-chloroethyl)piperazin-1-yl]-dibenzo[b,f]-1,4-thiazepine (Formula VIII) via cyclization using phosphorus oxychloride and phosphorus pentoxide.
- Reaction of the Formula VIII intermediate with ethylene glycol in the presence of a base such as sodium metal to form the Quetiapine base.
- Purification of the final product through crystallization and conversion to the hemifumarate salt, avoiding complex chromatographic separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to the process described in CN1177839C represents a significant opportunity for cost optimization and risk mitigation. The elimination of silica gel column chromatography is perhaps the most impactful economic driver, as chromatography is a low-throughput, high-solvent-consumption operation that drastically increases the cost of goods sold (COGS). By replacing this with crystallization, the process becomes inherently more scalable, allowing for larger batch sizes and reduced cycle times. Furthermore, the stability of the Formula VIII intermediate reduces the need for specialized storage conditions and minimizes the loss of valuable material due to degradation during warehousing or transport. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The removal of column chromatography significantly lowers solvent consumption and waste disposal costs, which are major expense drivers in API manufacturing. Additionally, the higher overall yield of 66-67% compared to the legacy 62.6% means that less raw material is required to produce the same amount of final product, directly improving material efficiency. The use of common reagents like ethylene glycol and phosphorus oxychloride, which are readily available in the global chemical market, further ensures that input costs remain stable and predictable. This streamlined approach reduces the number of unit operations, thereby lowering labor and energy consumption per kilogram of produced Quetiapine.
- Enhanced Supply Chain Reliability: The chemical stability of the new intermediates ensures that production campaigns are less vulnerable to interruptions caused by raw material spoilage. In the previous method, the sensitivity of the gem-haloimine to moisture required strict environmental controls, increasing the risk of batch rejection. The new robust intermediates can be stockpiled safely, providing a buffer against supply disruptions. This reliability is crucial for maintaining continuous production schedules and ensuring that downstream formulation partners receive their API shipments on time. The simplified purification process also reduces the lead time required for quality control testing, accelerating the release of finished batches.
- Scalability and Environmental Compliance: The shift from chromatographic purification to crystallization aligns with green chemistry principles by reducing the volume of organic solvents required for separation. This not only lowers the environmental footprint of the manufacturing process but also simplifies compliance with increasingly stringent environmental regulations regarding solvent emissions and waste treatment. The process is designed to be easily scaled from pilot plant quantities to multi-ton commercial production without the need for specialized equipment like large-scale chromatography columns. This scalability ensures that the manufacturing capacity can be expanded rapidly to meet growing global demand for antipsychotic medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of adopting this newer methodology. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on process efficiency.
Q: Why is the new Quetiapine synthesis method considered more economically viable than previous methods?
A: The new method described in CN1177839C eliminates the need for silica gel column chromatography, which is costly and difficult to scale. Additionally, it utilizes stable intermediates that do not hydrolyze in moist air, reducing waste and improving overall yield from approximately 62.6% to 66-67%.
Q: What are the critical stability advantages of the Formula VIII intermediate?
A: Unlike the gem-haloimine intermediates used in older processes (Formula XI), which are highly unstable and prone to hydrolysis in moist air, the Formula VIII haloethylpiperazinylthiazepine derivative is chemically stable. This stability ensures consistent quality during storage and handling, crucial for large-scale manufacturing.
Q: How does this process impact the purity profile of the final API?
A: By avoiding the hydrolysis side reactions associated with unstable gem-haloimines and replacing column chromatography with efficient crystallization steps, the process significantly reduces impurity levels. This results in high-purity Quetiapine suitable for pharmaceutical applications without extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of synthesizing heterocyclic APIs like Quetiapine requires a partner with deep technical expertise and proven scale-up capabilities. Our R&D team has extensively analyzed the pathways described in CN1177839C and possesses the know-how to implement these optimized routes effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Quetiapine intermediate or API meets the highest international standards. We understand that in the pharmaceutical sector, quality is non-negotiable, and our commitment to excellence is reflected in our robust quality management systems.
We invite you to collaborate with us to leverage these advanced manufacturing technologies for your product portfolio. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our optimized synthesis of Quetiapine can enhance your supply chain efficiency and drive value for your organization.
