Advanced Aryloxy Bifunctional Synthesis of Spiro Indolone for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those with potential biological activity such as spiro indolones. A significant breakthrough in this domain is detailed in patent CN111057062A, which discloses a novel preparation method for synthesizing spiro indolone through aryloxy bifunctional synthesis based on furan and indole derivatives. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a greener, phosphotungstic acid-catalyzed system that leverages the inherent low aromaticity of indole and furan rings. By controlling reaction conditions and regioselectivity, this method enables the direct construction of stereospecific spiro indoline-3,3'-oxoindole frameworks via intramolecular C3 and C2 arylation. For R&D directors and procurement specialists, this innovation offers a compelling value proposition by simplifying synthetic routes while enhancing the purity profile of the final active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiro indole compounds has relied heavily on oxidation and lactonization reactions of indole propionic acid or series Michael ring expansion conversions of hydroxy indolone. These conventional methodologies suffer from significant drawbacks that hinder their efficiency in a commercial setting. Primarily, they often necessitate the use of expensive transition metal catalysts which not only drive up raw material costs but also introduce complex downstream purification challenges to remove trace metal impurities. Furthermore, these traditional routes are frequently plagued by harsh reaction conditions, multi-step sequences, and generally low yields, which collectively erode the overall process economics. Another critical issue is the lack of regioselectivity in methods like the Michael ring expansion, leading to mixtures of isomers that require energy-intensive separation techniques. The reliance on complex raw material synthesis in existing green methods further limits their scalability, creating bottlenecks for supply chain managers aiming for consistent high-volume production.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes simple and easily synthesized indole derivatives, specifically N-(o-bromophenyl)-3-indole carboxamide, as the key starting materials. This method ingeniously employs phosphotungstic acid as a recyclable catalyst to facilitate a Heck reaction that simultaneously dearomatizes the furan and indole rings. This bifunctional synthesis strategy allows for the direct construction of the spiro framework without the need for pre-functionalized complex precursors. The process is designed to be highly selective, generating stereospecific spiro indoline-3,3'-oxoindole and 5,11-dihydro-6H-indolo[3,2-c]quinoline-6-one derivatives by precisely tuning reaction parameters. By eliminating transition metals and streamlining the step count, this approach drastically simplifies the post-reaction treatment workflow. The ability to recycle the phosphotungstic acid catalyst further enhances the economic viability, making it a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
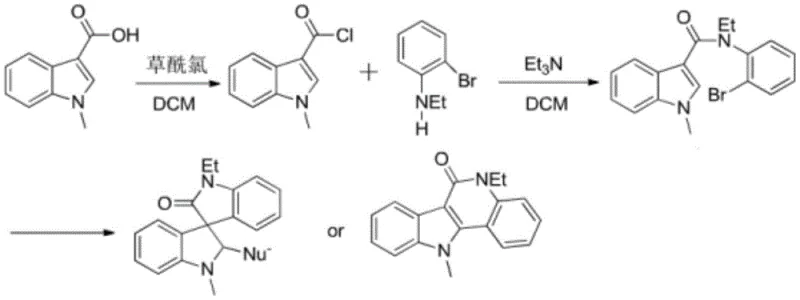
Mechanistic Insights into Phosphotungstic Acid-Catalyzed Heck Cyclization
The core of this technological advancement lies in the unique mechanistic pathway driven by the phosphotungstic acid catalyst during the intramolecular Heck reaction. Unlike traditional palladium-catalyzed cycles that require rigorous exclusion of oxygen and moisture, this system operates robustly under the specified thermal conditions. The mechanism initiates with the activation of the aryl bromide moiety on the N-(o-bromophenyl) group, facilitating an oxidative addition-like step that is stabilized by the polyoxometalate structure of the catalyst. This activation lowers the energy barrier for the subsequent migratory insertion into the electron-rich indole double bond. The catalyst effectively promotes the dearomatization of the indole ring, a thermodynamically challenging step, by stabilizing the cationic intermediates formed during the cyclization process. This precise control over the electronic environment ensures that the reaction proceeds through the desired C3 or C2 arylation pathway, minimizing side reactions such as polymerization or non-specific halogenation.
Furthermore, the impurity control mechanism is intrinsically linked to the high selectivity of this catalytic system. In conventional methods, competing reaction pathways often lead to regioisomers that are structurally similar to the target molecule, making purification difficult. However, the steric and electronic properties of the phosphotungstic acid catalyst create a specific coordination sphere that favors the formation of the spiro cyclic transition state over linear or alternative cyclic products. The reaction conditions, specifically the temperature control around 60 +/- 3 °C during the pre-reaction phase and the subsequent pressurized heating, are optimized to suppress the formation of by-products. This results in a crude product with a significantly cleaner impurity profile, reducing the burden on downstream purification units like column chromatography or crystallization. For quality assurance teams, this means a more consistent product batch-to-batch, ensuring that stringent purity specifications for API intermediates are met with greater reliability.
How to Synthesize Spiro Indolone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this chemistry from the laboratory bench to pilot plant operations. The process begins with a pre-reaction step where indole-3-carboxylic acid is condensed with o-bromoaniline in the presence of the catalyst, establishing the necessary amide linkage. This is followed by a controlled synthesis phase in a pressurized kettle, where the cyclization occurs under specific thermal and pressure gradients. The detailed operational parameters, including the use of anhydrous conditions and specific solvent systems like dichloromethane and DMF, are critical for maximizing yield. The final isolation involves a sophisticated distillation setup that separates the high-boiling spiro indolone product from residual solvents and lower boiling impurities. For a comprehensive understanding of the exact stoichiometric ratios and equipment specifications required for replication, please refer to the standardized synthesis steps provided below.
- Pre-react indole-3-carboxylic acid with o-bromoaniline and phosphotungstic acid catalyst at 60°C to form the amide precursor.
- Transfer materials to a synthesis kettle, pressurize to 0.20 Mpa, and heat under controlled conditions to initiate cyclization.
- Purify the crude product via distillation under vacuum (-0.1 Mpa) controlling kettle temperature at 145°C and tower top temperature below 195°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process addresses several critical pain points that typically affect the procurement of complex heterocyclic intermediates. The shift away from precious transition metals to a recyclable phosphotungstic acid system fundamentally alters the cost structure of the manufacturing process. By removing the need for expensive palladium or rhodium catalysts and the associated scavenging resins required to meet residual metal limits, the overall variable cost of goods sold is significantly reduced. Additionally, the use of readily available starting materials like indole-3-carboxylic acid and o-bromoaniline ensures a stable supply base, mitigating the risks associated with sourcing exotic or custom-synthesized reagents. The streamlined workflow, which combines dearomatization and spiro-cycle formation in fewer steps, also translates to reduced processing time and lower utility consumption per kilogram of product. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts is a primary driver for cost optimization in this process. Traditional methods often incur high expenses not only for the catalyst itself but also for the specialized ligands and the extensive purification steps needed to remove metal traces to ppm levels. By utilizing phosphotungstic acid, which can be recovered and reused, the process removes these substantial cost centers. Furthermore, the simplified post-reaction treatment reduces the consumption of silica gel and solvents typically used in extensive column chromatography, leading to substantial savings in waste disposal and material costs. The higher reaction yield reported in the patent further amplifies these savings by maximizing the output from each batch of raw materials, thereby improving the overall atom economy of the synthesis.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex catalysts or unstable intermediates. This method mitigates such risks by relying on commodity chemicals that are widely available in the global market. The robustness of the reaction conditions, which tolerate standard industrial heating and pressure parameters, ensures that production can be maintained consistently without frequent interruptions due to sensitive reaction failures. The ability to scale the process using standard stainless steel reactors, as indicated by the distillation and pressure specifications in the patent, means that manufacturing can be easily transferred between different facilities if necessary. This flexibility is crucial for maintaining long-term supply agreements and meeting the fluctuating demands of the pharmaceutical market without compromising on delivery timelines.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the green chemistry attributes of this process offer a distinct competitive advantage. The absence of heavy metals simplifies wastewater treatment and reduces the hazardous waste footprint of the manufacturing facility. The patent describes a continuous distillation process with specific temperature controls (e.g., kettle at 145 °C, tower top ≤ 195 °C) that is inherently scalable from pilot to commercial tonnage. This design facilitates the commercial scale-up of complex pharmaceutical intermediates while adhering to strict environmental, health, and safety (EHS) standards. The recyclability of the catalyst aligns with circular economy principles, further enhancing the sustainability profile of the product and appealing to eco-conscious stakeholders in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects. The answers provided reflect the practical implications of the reaction mechanism and process design on real-world manufacturing scenarios.
Q: What are the advantages of using phosphotungstic acid over transition metals?
A: Phosphotungstic acid eliminates the need for expensive transition metal catalysts, simplifying post-reaction treatment and allowing for catalyst recycling, which significantly reduces production costs and environmental impact.
Q: How does this method improve regioselectivity compared to traditional Michael reactions?
A: By utilizing an intramolecular Heck reaction mechanism, this process achieves precise C3 and C2 arylation control, avoiding the regioselectivity issues common in series Michael ring expansion conversions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent outlines specific industrial parameters including pressure control (0.20 Mpa) and continuous distillation protocols, demonstrating high feasibility for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Indolone Supplier
The technological potential of this aryloxy bifunctional synthesis route is immense, offering a pathway to high-value spiro indolone derivatives that are critical for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex patent methodologies into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and impurity profile of every batch. Our commitment to quality ensures that the spiro indolone intermediates we supply meet the exacting standards required for global pharmaceutical applications.
We invite potential partners to engage with us to explore how this innovative synthesis route can be tailored to your specific project needs. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener methodology. We encourage you to contact our technical procurement team to request specific COA data for similar heterocyclic compounds and to discuss route feasibility assessments for your target molecules. Together, we can accelerate the development of life-saving medicines by ensuring a reliable and cost-effective supply of these critical building blocks.
