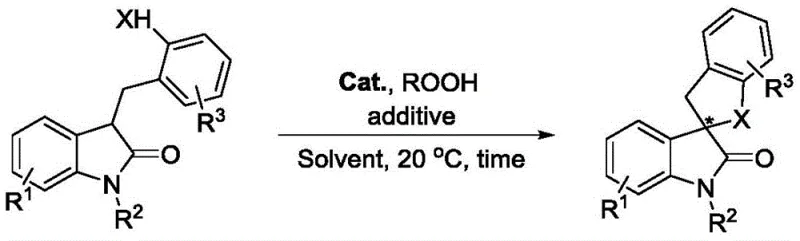
Advanced Organocatalytic Synthesis of Chiral Spiro Indolones for Scalable Pharmaceutical Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the demand for more efficient and environmentally benign methodologies to access complex chiral scaffolds. A significant breakthrough in this domain is documented in patent CN113980028A, which discloses a novel preparation method for chiral spiro indolone compounds. These structural motifs are ubiquitous in natural products and pharmaceutical agents, exhibiting potent biological activities ranging from anticancer to anti-HIV properties. The patented technology leverages an intramolecular carbonyl α-asymmetric oxidative coupling reaction, utilizing indolone derivatives bearing active hydrogen functional groups as substrates. By employing a cinchona alkaloid-derived quaternary ammonium iodide salt as a phase-transfer organocatalyst and a peroxide as the terminal oxidant, this method achieves high stereoselectivity under remarkably mild conditions. This represents a paradigm shift from traditional metal-catalyzed approaches, offering a robust pathway for the reliable chiral spiro indolone supplier to deliver high-purity intermediates essential for modern drug discovery pipelines.
The pursuit of efficient synthetic routes for chiral spiro indolones has historically been fraught with challenges related to reaction conditions and catalyst availability. Conventional methods often rely on Lewis acid-catalyzed 1,3-dipolar cycloadditions or nucleophilic phosphine-catalyzed cyclizations. While these strategies have yielded valuable results, they frequently suffer from limitations such as the requirement for stoichiometric amounts of expensive reagents, sensitivity to moisture and air, and the generation of toxic heavy metal waste streams that complicate downstream purification. Furthermore, achieving high enantioselectivity in these traditional frameworks often necessitates cryogenic temperatures or specialized ligands that are not commercially viable for large-scale operations. These factors collectively hinder the cost reduction in pharmaceutical intermediate manufacturing, creating bottlenecks for supply chains that require consistent, high-volume production of optically pure materials.
In stark contrast, the novel approach outlined in the patent data introduces a transformative strategy that circumvents these historical impediments. By utilizing a chiral quaternary ammonium iodide salt derived from cinchona alkaloids, the process eliminates the need for transition metals entirely, thereby simplifying the impurity profile and reducing the environmental footprint. The reaction proceeds efficiently at ambient temperature (20 °C) in common organic solvents like n-propyl acetate, demonstrating exceptional operational simplicity. The use of cumene hydroperoxide as a green oxidant further enhances the sustainability of the process. This methodology not only provides a widely applicable method for constructing chiral spiro indolone compounds but also ensures that the commercial scale-up of complex pharmaceutical intermediates is feasible without compromising on stereochemical integrity or safety standards.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Oxidative Coupling
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize process parameters. The catalytic cycle initiates with the interaction between the cinchona-derived quaternary ammonium iodide salt and the peroxide oxidant. Under the reaction conditions, the iodide anion is oxidized in situ to generate reactive hypoiodite or hypoiodous acid species. These electrophilic iodine species serve as the actual active agents that oxidize the enolizable carbonyl substrate, leading to the formation of a transient N-I intermediate. This activation step is pivotal as it renders the alpha-carbon susceptible to nucleophilic attack, setting the stage for the subsequent bond-forming event. The rigid chiral environment provided by the cinchona backbone ensures that this oxidation occurs with high facial selectivity, establishing the initial stereochemical bias required for the final product.
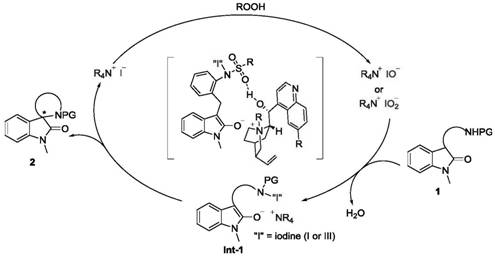
Following the formation of the N-I intermediate, the reaction proceeds through an intramolecular SN2-type displacement. The pendant nucleophile, which may be an amino, phenolic hydroxyl, or protected carboxyl group depending on the substrate design, attacks the activated alpha-carbon. This cyclization step is heavily influenced by steric induction and non-covalent interactions. Specifically, tight ion pairing between the bulky quaternary ammonium cation and the indolone enolate anion, along with potential hydrogen bonding between the catalyst's secondary alcohol moiety and the substrate's sulfonyl protecting group, creates a highly organized transition state. This precise spatial arrangement dictates the stereochemical outcome, resulting in the observed high enantiomeric ratios. Additionally, the reaction produces water as a byproduct, which can inhibit the catalyst or reverse the equilibrium; however, the strategic inclusion of molecular sieves mitigates this issue by sequestering water, thus driving the reaction to completion and maintaining high catalytic turnover.
How to Synthesize Chiral Spiro Indolone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocol parameters to maximize yield and enantioselectivity. The general procedure involves dissolving the indolone substrate in n-propyl acetate, followed by the addition of the chiral catalyst (typically 3 mol%) and activated 5Å molecular sieves. The mixture is cooled or maintained at 20 °C before the slow addition of cumene hydroperoxide (3 equivalents). The reaction is then stirred for approximately 12 hours. Upon completion, the mixture is quenched with aqueous sodium thiosulfate to reduce residual peroxides and iodine species, followed by extraction and purification via column chromatography. This standardized approach ensures reproducibility across different batches and scales. For detailed operational specifics regarding reagent grades and workup procedures, please refer to the structured guide below.
- Prepare the reaction mixture by adding the indolone-derived substrate, cinchona alkaloid-derived quaternary ammonium iodide catalyst (3 mol%), and 5Å molecular sieves to n-propyl acetate solvent.
- Introduce cumene hydroperoxide (3 equivalents) as the oxidant to the stirred mixture at a controlled temperature of 20°C.
- Maintain stirring for 12 hours to allow the intramolecular carbonyl alpha-asymmetric oxidative coupling to proceed to completion, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this organocatalytic methodology offers distinct strategic advantages over legacy synthetic routes. The elimination of precious metal catalysts removes a significant cost driver associated with both raw material acquisition and the rigorous purification steps required to meet residual metal specifications in pharmaceutical products. Furthermore, the use of commodity chemicals such as cumene hydroperoxide and n-propyl acetate ensures a stable and diversified supply base, reducing the risk of shortages that often plague specialized reagent markets. The mild reaction conditions also translate to lower energy consumption and reduced safety hazards, facilitating smoother regulatory approvals and faster time-to-market for new drug candidates relying on these chiral scaffolds.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the replacement of expensive transition metal catalysts with inexpensive, bio-derived cinchona alkaloid salts. This shift not only lowers the direct material cost but also simplifies the downstream processing workflow by eliminating the need for specialized scavengers to remove heavy metal traces. Consequently, the overall cost of goods sold (COGS) for the final API intermediate is significantly reduced, allowing for more competitive pricing structures in the global market without sacrificing quality or purity standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available starting materials and reagents. Unlike proprietary ligands or rare earth metals that may be subject to geopolitical supply constraints, the components of this catalytic system are industrial commodities with multiple global sources. This diversification minimizes the risk of production delays caused by single-source dependencies. Additionally, the robustness of the reaction at ambient temperature reduces the dependency on complex cryogenic infrastructure, ensuring that manufacturing can continue uninterrupted even during utility fluctuations or equipment maintenance scenarios.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating under mild thermal conditions that minimize the risk of thermal runaway events common in exothermic oxidations. This safety profile allows for larger batch sizes and more efficient reactor utilization. From an environmental standpoint, the metal-free nature of the catalysis aligns perfectly with green chemistry principles, reducing the burden of hazardous waste disposal. The simplified waste stream facilitates easier compliance with increasingly stringent environmental regulations, thereby avoiding potential fines and reputational damage associated with heavy metal pollution.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing clarity on reaction scope, catalyst loading, and purification strategies. Understanding these nuances is essential for process chemists aiming to adapt this methodology to specific substrate classes or manufacturing constraints.
Q: What are the advantages of this organocatalytic method over traditional Lewis acid catalysis?
A: Unlike traditional methods that often require harsh conditions or expensive transition metals, this novel approach utilizes a metal-free cinchona alkaloid catalyst under mild conditions (20°C), significantly reducing operational complexity and heavy metal contamination risks.
Q: Why are molecular sieves critical in this oxidative coupling reaction?
A: Water is a byproduct of the oxidative coupling reaction which can slow down the process or shift the equilibrium backwards. The addition of 5Å molecular sieves effectively absorbs generated water molecules, driving the reaction forward and improving both yield and efficiency.
Q: Is the catalyst system suitable for large-scale industrial production?
A: Yes, the catalyst is derived from readily available cinchona alkaloids and operates at ambient temperature without requiring cryogenic conditions or high pressure, making the process highly amenable to commercial scale-up and safe manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists is well-versed in the intricacies of organocatalytic transformations, including the oxidative coupling strategies described in patent CN113980028A. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from bench-scale discovery to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying enantiomeric excess and impurity profiles to meet the most demanding global regulatory standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, cost-effectiveness, and long-term reliability in the competitive pharmaceutical landscape.
