Revolutionizing Spiro Indolone Production via Green Aqueous Phase Catalysis for Commercial Scale
Revolutionizing Spiro Indolone Production via Green Aqueous Phase Catalysis for Commercial Scale
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of reducing waste disposal costs. In this context, the technology disclosed in patent CN109776381B represents a significant breakthrough in the synthesis of spirocyclic indolinone compounds, which are critical scaffolds in numerous bioactive molecules exhibiting anti-tuberculosis, anti-cancer, and anti-HIV activities. This proprietary methodology replaces traditional hazardous organic solvents with water, utilizing a dual-function quaternary ammonium iodide catalyst that serves as both a phase-transfer agent and a catalytic precursor for hypervalent iodine species. For R&D directors and procurement managers seeking a reliable spiro indolone supplier, this aqueous-phase approach offers a compelling value proposition by drastically simplifying the workup procedure and eliminating the need for expensive solvent recovery systems. The ability to conduct these oxidative coupling reactions under mild conditions (23-60°C) using inexpensive hydrogen peroxide as the terminal oxidant underscores the potential for substantial cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the high atom economy and excellent yields reported in the patent data suggest that this route is not merely a laboratory curiosity but a viable candidate for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirocyclic indolinone derivatives has relied heavily on oxidative coupling reactions performed in organic solvents such as dichloromethane, acetonitrile, or dimethylformamide. These conventional protocols often necessitate the use of stoichiometric amounts of heavy metal oxidants or expensive hypervalent iodine reagents, which generate significant quantities of toxic waste and complicate the purification process. From a supply chain perspective, the reliance on volatile organic compounds (VOCs) introduces severe safety hazards, including flammability and toxicity risks, which require specialized infrastructure and increase operational expenditures. Moreover, the solubility of polar intermediates in non-polar organic solvents can be inconsistent, leading to variable reaction rates and difficult-to-control impurity profiles. Previous attempts to utilize iodine salts in organic media have also shown limited substrate scope, often failing when applied to indolinone derivatives with diverse electronic properties. These technical bottlenecks result in prolonged lead times for high-purity spirocyclic indolinones and inflate the overall cost of goods sold (COGS), making it challenging for manufacturers to remain competitive in the global API market.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these challenges by employing water as the exclusive reaction medium, leveraging the unique amphiphilic properties of long-chain quaternary ammonium iodides like trimethyldodecylammonium iodide (TMDAI). In this system, the catalyst self-assembles into micelles within the aqueous phase, effectively solubilizing the hydrophobic organic substrates and creating a localized high-concentration environment that accelerates the reaction kinetics. This micellar catalysis allows for the efficient generation of reactive hypervalent iodine species in situ from cheap hydrogen peroxide, driving the intramolecular oxidative coupling to completion with exceptional efficiency. As illustrated in the reaction scheme below, the transformation proceeds smoothly to form the spirocyclic core without the need for any organic co-solvents, representing a true green chemistry advancement.
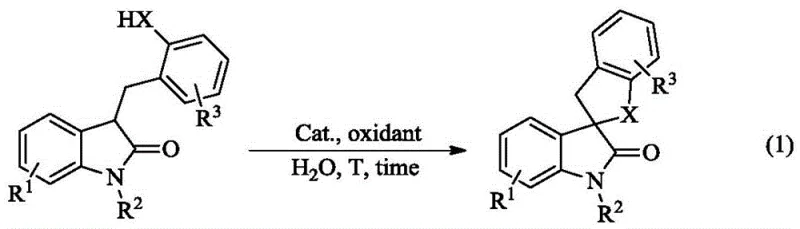
This novel approach not only enhances the environmental profile of the synthesis but also broadens the substrate scope significantly, accommodating various substituents on the aromatic rings without compromising yield. The simplicity of the protocol, which involves merely mixing the substrate, catalyst, and oxidant in water, reduces the operational complexity and minimizes the risk of human error during batch preparation. For procurement teams, this translates to a more robust supply chain where raw material availability is less constrained by solvent logistics, and the final product isolation is streamlined through simple extraction and crystallization steps.
Mechanistic Insights into Quaternary Ammonium Iodide-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this aqueous-phase transformation is crucial for R&D directors aiming to optimize the process for specific analogs. The reaction initiates with the oxidation of the iodide anion from the quaternary ammonium salt by hydrogen peroxide, generating reactive hypervalent iodine species such as hypoiodite (IO⁻) or periodate (IO₂⁻) within the micellar core. These electrophilic iodine species then interact with the electron-rich aromatic ring or the nitrogen center of the indolinone substrate, forming a transient N-I or O-I bonded intermediate. This activation step is critical as it increases the nucleophilicity of the adjacent carbon center, facilitating the subsequent intramolecular nucleophilic attack that closes the spirocyclic ring. The presence of the long alkyl chain on the ammonium cation is not merely incidental; it stabilizes the transition state within the hydrophobic pocket of the micelle, shielding the reactive intermediates from bulk water interference and preventing premature hydrolysis.
Furthermore, the versatility of this mechanism extends to different substrate classes, including 3-indolepropionic acid derivatives, as demonstrated by the generalized reaction pathway shown below. In these cases, the carboxylic acid moiety participates in the cyclization, leading to a distinct spiro-lactone fused indolinone structure. The ability of the catalyst system to adapt to different functional groups (phenolic hydroxyl, carboxyl, or protected amine) highlights the robustness of the catalytic cycle.
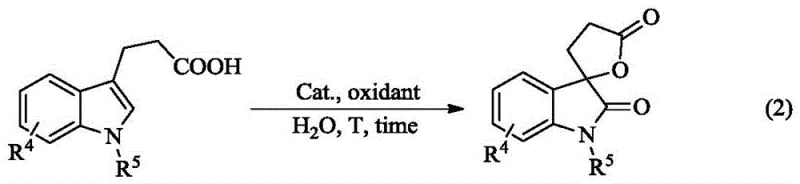
Impurity control is inherently superior in this system because the aqueous environment suppresses many side reactions common in organic solvents, such as radical polymerization or over-oxidation. The micellar confinement ensures that the reaction follows a specific trajectory towards the desired spiro-product, resulting in a cleaner crude reaction mixture. This high selectivity reduces the burden on downstream purification units, allowing for higher overall throughput and consistent quality across different batches. For technical teams, this means that scaling the reaction does not require extensive re-optimization of purification parameters, as the impurity profile remains stable regardless of the batch size.
How to Synthesize Spiro Indolone Compounds Efficiently
The practical implementation of this synthesis route is remarkably straightforward, requiring standard laboratory equipment and readily available reagents. The process begins with the suspension of the indolinone-derived substrate in water, followed by the addition of the TMDAI catalyst. Hydrogen peroxide is then introduced slowly to control the exotherm, although the reaction is generally mild enough to proceed safely at ambient or slightly elevated temperatures. Detailed standardized operating procedures regarding stoichiometry, addition rates, and specific workup protocols are essential for ensuring reproducibility at larger scales.
- Prepare the reaction mixture by combining the indolinone-derived substrate or 3-indolepropionic acid derivative with trimethyldodecylammonium iodide (TMDAI) catalyst in water.
- Add hydrogen peroxide (3 to 6 equivalents) as the oxidant to the aqueous suspension at temperatures ranging from 23°C to 60°C.
- Stir the reaction for 18 to 48 hours, then extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the transition to this aqueous-phase technology offers transformative benefits that extend beyond simple yield improvements. By eliminating the need for large volumes of organic solvents, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing through the removal of solvent purchase, storage, and disposal expenses. The use of water as a solvent also mitigates the regulatory burdens associated with VOC emissions, simplifying compliance with environmental protection laws and reducing the risk of production shutdowns due to regulatory non-compliance. Additionally, the catalyst system utilizes inexpensive, commodity-grade chemicals that are globally sourced, ensuring that the supply chain is not vulnerable to the geopolitical fluctuations often seen with specialized organometallic catalysts.
- Cost Reduction in Manufacturing: The economic impact of replacing organic solvents with water cannot be overstated, as it removes the entire lifecycle cost associated with solvent management. Traditional processes often incur hidden costs related to solvent recycling distillation columns and the treatment of halogenated waste streams, all of which are eradicated in this water-based protocol. Furthermore, the high conversion rates and minimal byproduct formation mean that raw material utilization is maximized, directly lowering the cost per kilogram of the active pharmaceutical ingredient. The catalyst loading is low, and the potential for recycling the aqueous catalyst solution further enhances the economic viability of the process for long-term production campaigns.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step synthetic routes involving sensitive reagents often leads to supply disruptions; however, this method relies on robust, shelf-stable starting materials. The starting indolinone derivatives and 3-indolepropionic acids are commercially available in bulk quantities from multiple vendors, reducing the risk of single-source dependency. The mild reaction conditions (23-60°C) also mean that the process can be run in standard glass-lined or stainless steel reactors without the need for cryogenic cooling or high-pressure equipment, making it accessible to a wider network of contract manufacturing organizations (CMOs). This flexibility ensures reducing lead time for high-purity spirocyclic indolinones, allowing pharmaceutical companies to respond faster to market demands.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale is notoriously difficult when heat transfer and mixing become limiting factors, but the aqueous nature of this reaction simplifies thermal management significantly. Water has a high heat capacity, which helps dissipate the heat generated during the oxidation step, preventing runaway reactions and ensuring safe operation at the 100 MT scale. The absence of toxic organic waste streams aligns perfectly with modern green chemistry principles, facilitating easier permitting for new manufacturing facilities. This environmental compatibility is increasingly becoming a prerequisite for partnerships with major multinational pharma companies who have strict sustainability mandates for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and relevance for industry professionals.
Q: What is the primary advantage of using quaternary ammonium iodide in this synthesis?
A: Quaternary ammonium iodide acts as both a catalyst and a surfactant. Its long hydrophobic chain forms micelles in water, significantly increasing the solubility of organic substrates and facilitating the reaction in an aqueous environment without organic co-solvents.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to the use of water as a solvent, which simplifies heat management and safety protocols compared to volatile organic solvents. The catalyst system is robust, and product isolation is straightforward.
Q: What are the typical reaction conditions for high yield?
A: Optimal conditions typically involve using 10 mol% TMDAI catalyst and 3 to 6 equivalents of hydrogen peroxide. Reaction temperatures range from room temperature (23°C) to 60°C, with reaction times between 24 to 48 hours depending on the substrate reactivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of novel synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. While the patent technology provides the blueprint, translating it into a commercial reality demands rigorous process engineering and quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of spiro indolone intermediate meets the exacting standards required for downstream API synthesis. Our facility is equipped to handle the specific requirements of aqueous-phase chemistry, including specialized wastewater treatment systems that align with the green nature of this process.
We invite procurement leaders and R&D heads to engage with us for a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging this advanced aqueous synthesis route, we can help you optimize your supply chain and reduce overall production costs without compromising on quality. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Let us collaborate to bring your next-generation therapeutic candidates to market faster and more sustainably.
