Advanced Synthesis of Isoxazole-Arabinose Triazole Derivatives for Oncology and Antiviral Applications
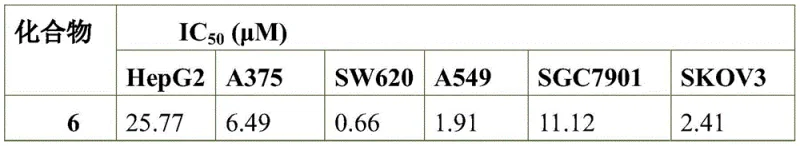
The pharmaceutical landscape is continuously evolving towards multifunctional small molecules that combine distinct pharmacophores to enhance therapeutic efficacy. Patent CN113429445A introduces a sophisticated chemical entity, specifically an isoxazole derivative containing an arabinose triazole structure, which represents a significant advancement in the design of dual-action therapeutic agents. This novel compound integrates the biological potential of 7-azaindole, known for its kinase inhibitory properties, with the metabolic stability of the isoxazole ring and the solubility-enhancing characteristics of the arabinose sugar moiety. The synthesis described in this patent offers a robust pathway for creating complex heterocyclic systems that demonstrate remarkable potency against various tumor cell lines, including SW620 and A549, as well as exhibiting strong in vitro anti-Hepatitis B virus activity. For research and development teams seeking to expand their oncology or antiviral pipelines, understanding the synthetic accessibility and structural nuances of this molecule is critical for future drug discovery programs.
As a reliable pharmaceutical intermediate supplier, recognizing the strategic value of such multi-target ligands is essential. The integration of a glycosylated triazole unit not only improves the physicochemical properties, such as water solubility and bioavailability, but also introduces specific interactions with biological receptors through hydrogen bonding. This patent provides a foundational blueprint for manufacturing high-purity oncology intermediates that can serve as lead compounds or advanced building blocks for next-generation medicines. The detailed methodology ensures that the stereochemistry of the arabinose component is preserved, which is often a challenging aspect in carbohydrate chemistry, thereby guaranteeing the biological fidelity of the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex glycosylated heterocycles has been fraught with challenges related to regioselectivity, harsh reaction conditions, and the use of toxic heavy metal catalysts. Conventional routes for constructing isoxazole rings often rely on stoichiometric amounts of hazardous reagents or require extreme temperatures that can degrade sensitive sugar protecting groups. Furthermore, introducing a triazole ring typically involves copper-catalyzed azide-alkyne cycloaddition (CuAAC), which necessitates rigorous purification steps to remove residual copper traces that are unacceptable in pharmaceutical grade materials. These legacy methods often result in lower overall yields, increased waste generation, and significant difficulties in scaling up due to safety concerns associated with azide handling and exothermic reactions. Additionally, maintaining the stereochemical integrity of the sugar moiety during aggressive cyclization steps is frequently problematic, leading to mixtures of anomers that complicate downstream purification and reduce the potency of the final active pharmaceutical ingredient.
The Novel Approach
The methodology outlined in patent CN113429445A presents a transformative solution by employing a Chloramine-T mediated 1,3-dipolar cycloaddition strategy. This approach elegantly bypasses the need for transition metal catalysts, utilizing the oxidative power of Chloramine-T to generate nitrile oxides in situ from oxime precursors under relatively mild reflux conditions in ethanol. This green chemistry aspect significantly simplifies the workup procedure, as the removal of metal residues is rendered unnecessary, directly addressing the purity concerns of R&D directors. The reaction sequence is designed to be telescoped efficiently, starting from readily available 3-nitro-1-benzenesulfonyl-7-azaindole and a pre-functionalized arabinose triazole salicylaldoxime. By conducting the cyclization in a protic solvent like ethanol, the process ensures better solubility of the polar intermediates and facilitates the subsequent recrystallization steps. This novel route not only enhances the atom economy of the synthesis but also provides a safer operational profile, making it highly attractive for cost reduction in API manufacturing where safety and environmental compliance are paramount.
Mechanistic Insights into Chloramine-T Mediated Cycloaddition
The core of this synthetic innovation lies in the generation and trapping of a nitrile oxide dipole. In the presence of Chloramine-T (N-chloro-p-toluenesulfonamide sodium salt), the oxime functionality of the arabinose-triazole precursor undergoes oxidative dehydrogenation to form the reactive nitrile oxide species. This transient intermediate then participates in a concerted [3+2] cycloaddition with the electron-deficient double bond system present in the activated 7-azaindole derivative. The regioselectivity of this addition is governed by the electronic properties of the dipole and dipolarophile, ensuring the formation of the desired isoxazoline skeleton fused to the pyrrolo-pyridine core. The use of Chloramine-T is particularly advantageous as it acts as a mild oxidant that tolerates the sensitive acetyl protecting groups on the arabinose ring, preventing premature deprotection or degradation of the sugar moiety. This mechanistic pathway avoids the formation of radical species that could lead to polymerization or side reactions, thereby ensuring a clean reaction profile with minimal byproduct formation.

Following the construction of the isoxazole core, the final step involves the removal of the acetyl protecting groups to reveal the free hydroxyl groups of the arabinose sugar. This is achieved using sodium methoxide in methanol under nitrogen protection at controlled temperatures. The base-catalyzed transesterification proceeds smoothly to yield the final triol product without affecting the sensitive isoxazole or triazole rings. This deprotection strategy is critical for restoring the hydrophilicity of the molecule, which is essential for its biological activity and solubility profile. The entire process demonstrates excellent control over impurity profiles, as the mild conditions prevent the epimerization of the sugar chiral centers. For quality control teams, this means a more consistent impurity spectrum that is easier to characterize and control, aligning with stringent regulatory requirements for new drug substances.
How to Synthesize Isoxazole-Arabinose Triazole Derivative Efficiently
The synthesis protocol described in the patent offers a reproducible and scalable method for producing this high-value intermediate. The process begins with the preparation of the key oxime intermediate, followed by the crucial cycloaddition step and final deprotection. Each stage is optimized for yield and purity, utilizing standard laboratory equipment and commercially available reagents. The following guide outlines the critical operational parameters derived from the patent examples, ensuring that technical teams can replicate the success of the original inventors while adapting the process for larger scale production needs. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in pilot plant settings.
- Preparation of Acetyl Arabinose Triazole Salicylaldoxime via condensation with hydroxylamine hydrochloride.
- 1,3-Dipolar cycloaddition using Chloramine-T in ethanol to construct the isoxazole core fused to the 7-azaindole scaffold.
- Deprotection of acetyl groups using sodium methoxide in methanol to yield the final triol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of expensive and supply-constrained transition metal catalysts, such as palladium or copper, directly translates to reduced raw material costs and simplified vendor management. By relying on commodity chemicals like Chloramine-T, ethanol, and sodium methoxide, the manufacturing process becomes less susceptible to geopolitical supply disruptions that often affect specialized catalytic reagents. Furthermore, the use of common solvents simplifies solvent recovery and recycling protocols, contributing to a lower overall environmental footprint and reduced waste disposal costs. This alignment with green chemistry principles not only lowers operational expenditures but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical sourcing strategies.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts, which are subject to volatile market pricing and require costly removal steps to meet ppm-level specifications. By utilizing Chloramine-T as a stoichiometric oxidant, the cost of goods sold (COGS) is significantly optimized. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, reducing the burden on purification resources and increasing the overall throughput of the manufacturing line. The ability to perform recrystallization directly from the reaction mixture further streamlines the isolation process, saving both time and energy compared to complex chromatographic separations often required in traditional routes.
- Enhanced Supply Chain Reliability: The starting materials, including 7-azaindole derivatives and salicylaldehyde precursors, are widely available from multiple global suppliers, ensuring a robust and redundant supply base. This diversity mitigates the risk of single-source dependency, a critical factor for maintaining continuous production schedules. The synthetic steps operate under atmospheric pressure and moderate temperatures, reducing the need for specialized high-pressure reactors or cryogenic cooling infrastructure. This operational simplicity allows for flexible manufacturing across different sites, enhancing the resilience of the supply network against localized disruptions or facility maintenance downtimes.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot production without significant re-optimization. The absence of heavy metals simplifies the environmental permitting process and reduces the cost of wastewater treatment, as the effluent does not require specialized heavy metal scavenging. The solid byproducts, primarily sulfonamide derivatives, are easier to handle and dispose of compared to toxic metal sludge. This ease of scale-up ensures that commercial demands can be met rapidly, reducing lead time for high-purity pharmaceutical intermediates and accelerating the time-to-market for downstream drug products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this isoxazole derivative. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific development pipeline.
Q: What is the primary biological activity of this isoxazole derivative?
A: The compound exhibits potent antitumor activity against cell lines such as SW620 (colon cancer) and A549 (lung cancer), alongside significant anti-Hepatitis B virus (HBV) replication inhibition.
Q: How does the Chloramine-T method improve synthesis efficiency?
A: Chloramine-T serves as a mild oxidant to generate nitrile oxides in situ from oximes, facilitating 1,3-dipolar cycloaddition under reflux conditions without requiring expensive transition metal catalysts.
Q: Is this intermediate suitable for large-scale pharmaceutical production?
A: Yes, the process utilizes common solvents like ethanol and methanol, avoids cryogenic conditions, and employs straightforward workup procedures like filtration and column chromatography, ensuring scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazole Derivative Supplier
The technical sophistication of the isoxazole-arabinose triazole derivative underscores the need for a manufacturing partner with deep expertise in complex heterocyclic chemistry. NINGBO INNO PHARMCHEM stands ready to support your development goals with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including strict moisture control for the sodium methoxide step and efficient solvent recovery systems for the ethanol reflux process. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the highest standards of quality, fully compliant with international regulatory guidelines for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative chemistry for your next breakthrough therapy. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can enhance your project's economic viability. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your journey from bench to bedside with confidence and reliability.
