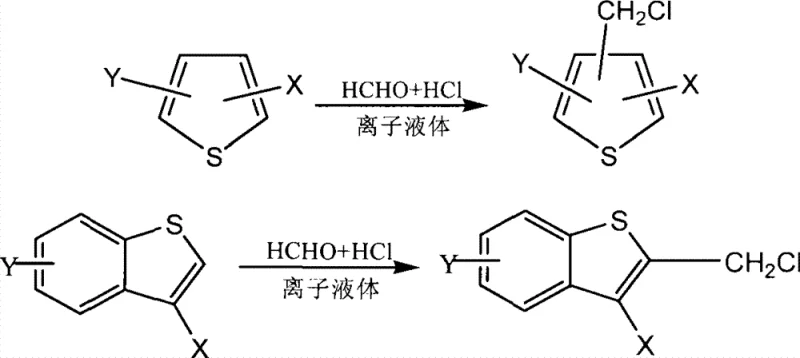
Advanced Ionic Liquid Catalysis for Scalable Thiophene Chloromethylation in Pharmaceutical Manufacturing
The chemical landscape of fine organic synthesis is continually evolving, driven by the urgent need for more sustainable and efficient manufacturing protocols that align with modern environmental standards. Patent CN101381362B introduces a groundbreaking methodology for the preparation of thiophene derivative chloromethylation products, utilizing ionic liquids as a novel catalytic system. This technology represents a significant departure from traditional acid-catalyzed processes, offering a pathway to high-purity intermediates essential for the pharmaceutical and agrochemical industries. The core innovation lies in the substitution of corrosive protonic acids with stable, recyclable ionic liquids, which not only enhances reaction efficiency but also drastically simplifies the downstream purification workflow. For R&D directors and process chemists, this patent data suggests a robust route for introducing chloromethyl groups onto thiophene rings, a critical transformation for building complex molecular architectures found in active pharmaceutical ingredients. The reported yields, ranging from 70% to 92%, indicate a highly reliable chemical process that minimizes raw material loss and maximizes output per batch.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the Blanc chloromethylation of aromatic and heteroaromatic compounds has relied heavily on the use of strong protonic acids such as sulfuric acid, hydrochloric acid, or phosphoric acid, often in conjunction with hazardous reagents like chloromethyl ether. These conventional methods present substantial challenges for industrial scale-up, primarily due to the severe corrosion issues they impose on reactor equipment and the complex waste streams they generate. The use of excess strong acids necessitates rigorous neutralization steps, often involving large volumes of base, which leads to significant salt waste and environmental burden. Furthermore, the separation of the desired chloromethylated product from the acidic reaction mixture can be cumbersome, often requiring multiple extraction and washing cycles that reduce overall process efficiency. In many cases, the harsh reaction conditions required by these traditional catalysts can lead to side reactions, such as polymerization or over-chlorination, which compromise the purity of the final intermediate and complicate the impurity profile. For procurement and supply chain managers, these inefficiencies translate into higher operational costs and longer lead times, making the conventional approach less attractive for large-scale commercial production.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent leverages the unique physicochemical properties of ionic liquids to facilitate the chloromethylation reaction under much milder and controlled conditions. The ionic liquids employed, such as various imidazolium salts, act as both catalysts and phase-transfer agents, enabling a homogeneous reaction environment that enhances the contact between reactants. This innovation allows the reaction to proceed at moderate temperatures, typically between 20°C and 60°C, significantly reducing energy consumption compared to high-temperature reflux methods. A key advantage of this system is the biphasic separation that occurs post-reaction, where the product resides in the organic phase while the ionic liquid catalyst remains in the aqueous phase. This automatic zoning eliminates the need for complex quenching procedures and allows for the direct recovery of the catalyst for reuse. The simplicity of the workup, involving merely extraction and vacuum distillation, ensures that high-quality products are obtained with minimal processing steps, thereby streamlining the manufacturing workflow and reducing the potential for human error or contamination during handling.
Mechanistic Insights into Ionic Liquid-Catalyzed Chloromethylation
The mechanistic pathway of this ionic liquid-catalyzed chloromethylation involves the activation of formaldehyde and hydrochloric acid to generate the electrophilic chloromethyl species in situ. The ionic liquid plays a crucial role in stabilizing the transition states and facilitating the electrophilic substitution on the thiophene ring. Unlike traditional Lewis acids that might coordinate too strongly or degrade, the ionic liquid provides a stable ionic environment that promotes the formation of the reactive intermediate without decomposing the sensitive thiophene substrate. The catalytic cycle is sustained by the ability of the ionic liquid to solubilize the inorganic reagents while maintaining immiscibility with the organic product, effectively driving the equilibrium towards product formation. This mechanism ensures high selectivity for the mono-chloromethylated product, minimizing the formation of di-chloromethylated byproducts which are common in less controlled acidic environments. The precise control over the reaction kinetics afforded by the ionic liquid medium allows for fine-tuning of the substitution pattern, which is critical for synthesizing specific isomers required in drug development.
Furthermore, the impurity control mechanism inherent in this process is superior to conventional acid catalysis due to the mildness of the reaction conditions. The absence of strong oxidizing acids reduces the risk of ring oxidation or sulfonation, which are common side reactions in thiophene chemistry. The ionic liquid medium also suppresses polymerization of the formaldehyde reagent, ensuring that the electrophile is available for the desired substitution rather than being consumed in side reactions. This results in a cleaner crude reaction mixture, which simplifies the purification process and reduces the load on downstream distillation columns. For quality control teams, this means a more consistent impurity profile across different batches, facilitating easier regulatory approval for pharmaceutical intermediates. The ability to recycle the ionic liquid without significant loss of catalytic activity further ensures that the impurity profile remains stable over multiple production cycles, providing long-term reliability for commercial supply chains.
How to Synthesize Thiophene Chloromethylation Products Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible method for producing high-quality chloromethyl thiophene derivatives suitable for industrial application. The process begins with the precise weighing of the thiophene substrate, paraformaldehyde, and the selected ionic liquid catalyst, which are then charged into a reaction vessel equipped with stirring and reflux capabilities. Concentrated hydrochloric acid is added to initiate the reaction, and the mixture is heated to the specified temperature while maintaining continuous agitation to ensure homogeneity. The reaction progress is monitored over a period of 4 to 10 hours, after which the mixture is allowed to cool and separate into distinct organic and aqueous layers. The detailed standardized synthesis steps see the guide below for exact parameters regarding specific substrates.
- Prepare the reaction mixture by adding ionic liquid, thiophene derivative, paraformaldehyde, and hydrochloric acid to a flask equipped with stirring and reflux.
- Heat the mixture to the designated temperature between 20°C and 60°C while maintaining continuous magnetic agitation for 4 to 10 hours.
- Separate the organic phase from the ionic liquid catalyst and purify the crude product via vacuum distillation to obtain high-purity chloromethylated thiophene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ionic liquid-catalyzed process offers substantial strategic advantages for procurement and supply chain management teams looking to optimize their manufacturing costs and reliability. The elimination of corrosive strong acids reduces the requirement for specialized corrosion-resistant equipment, allowing for the use of standard glass-lined or stainless steel reactors which are more readily available and cost-effective. The recyclability of the ionic liquid catalyst means that the consumption of catalytic materials is drastically reduced over time, leading to significant long-term savings on raw material costs. Additionally, the simplified workup procedure reduces the consumption of solvents and washing agents, further lowering the variable costs associated with each production batch. These efficiencies collectively contribute to a more competitive pricing structure for the final intermediate, making it an attractive option for cost-sensitive pharmaceutical and agrochemical projects.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily driven by the reduction in waste treatment costs and the extended lifespan of the catalyst. By avoiding the generation of large volumes of acidic waste, facilities can save significantly on neutralization chemicals and waste disposal fees, which are often a major component of operational expenditure. The ability to reuse the ionic liquid multiple times without complex regeneration steps means that the effective cost per kilogram of catalyst is negligible after the initial investment. Moreover, the high yields reported in the patent data ensure that raw material utilization is maximized, reducing the cost of goods sold by minimizing the amount of starting material required to produce a fixed quantity of product. This lean manufacturing approach aligns perfectly with the goals of procurement managers seeking to reduce overall production costs without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly enhanced by the robustness of this chemical process, which is less susceptible to variations in raw material quality or environmental conditions. The mild reaction conditions reduce the risk of thermal runaways or equipment failures, ensuring consistent production schedules and on-time delivery to customers. The availability of the ionic liquid catalysts and common reagents like paraformaldehyde and hydrochloric acid ensures that the supply chain is not dependent on scarce or geopolitically sensitive materials. This stability allows supply chain heads to plan inventory levels more accurately and reduce the need for safety stock, thereby freeing up working capital. The simplified logistics of handling non-corrosive catalysts also reduces transportation risks and regulatory burdens associated with hazardous chemical shipments.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the homogeneous nature of the reaction and the ease of heat management at moderate temperatures. The environmental profile of the process is significantly improved, as the ionic liquids are non-volatile and do not contribute to atmospheric emissions, helping facilities meet stringent environmental regulations. The reduction in solvent usage and waste generation supports sustainability goals and can facilitate easier permitting for new production lines. For companies aiming to enhance their green chemistry credentials, adopting this technology demonstrates a commitment to environmentally responsible manufacturing practices. The scalability ensures that demand surges can be met without the need for extensive process re-engineering, providing a flexible manufacturing base for growing markets.
Frequently Asked Questions (FAQ)
The following questions and answers address common technical and commercial inquiries regarding the implementation of this ionic liquid-catalyzed chloromethylation technology. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and benefits for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What catalyst system is utilized in this chloromethylation process?
A: The process utilizes environmentally friendly ionic liquids, such as imidazolium salts, which act as efficient phase-transfer catalysts and can be recycled after simple dehydration.
Q: What are the typical yields for this thiophene derivative synthesis?
A: According to patent data, the reaction achieves high conversion rates with isolated yields ranging consistently from 70% to 92% across various substituted thiophene substrates.
Q: Is the ionic liquid catalyst reusable for industrial scale-up?
A: Yes, the ionic liquid catalyst remains in the aqueous phase after reaction and can be recovered and reused multiple times after simple water removal, significantly reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiophene Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of ionic liquid catalysis and can effectively translate the methodologies described in patent CN101381362B into robust commercial processes. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of thiophene derivative meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and consistency makes us an ideal partner for companies seeking a reliable source of high-performance chemical intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impact of switching to this greener, more efficient technology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you optimize your supply chain and achieve your production goals with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
