Industrial Scale-Up of Optically Active Chrysanthemic Acid via Novel Resolution Technology
Industrial Scale-Up of Optically Active Chrysanthemic Acid via Novel Resolution Technology
The global demand for high-performance synthetic pyrethroid insecticides continues to drive the need for efficient, scalable production of their key acid components, specifically optically active chrysanthemic acid. Patent CN1373116A introduces a transformative methodology that addresses long-standing inefficiencies in the optical resolution of chrysanthemic acid derivatives. Unlike traditional methods that often struggle with low yields or require prohibitively expensive high-purity starting materials, this invention discloses a robust process capable of upgrading chrysanthemic acid with a trans-isomer ratio of not less than 70% and an initial optical purity as low as 2% e.e. to less than 10% e.e. This technological breakthrough allows manufacturers to utilize crude intermediates from asymmetric catalytic processes directly, bypassing costly purification steps and significantly enhancing the overall atom economy of the synthesis. For R&D directors and procurement strategists in the agrochemical sector, this represents a pivotal shift towards more sustainable and cost-effective manufacturing pathways for essential pest control intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of optically active chrysanthemic acid has been plagued by significant economic and technical bottlenecks. Conventional optical resolution techniques typically rely on reacting racemic or trans-rich chrysanthemic acid with optically active amines to form diastereomeric salts. However, these legacy methods often suffer from suboptimal yields because they generally require starting materials with relatively high optical purity to achieve effective separation, or they result in substantial loss of the desired isomer in the mother liquor. Furthermore, many traditional resolving agents are either too expensive for large-scale application or difficult to recover and recycle, leading to inflated production costs and increased waste generation. The inability to effectively process low-purity intermediates means that manufacturers must invest in additional upstream purification stages, complicating the supply chain and extending lead times for critical agrochemical intermediates needed for seasonal pesticide production cycles.
The Novel Approach
The methodology outlined in the patent data offers a sophisticated solution by leveraging specific classes of optically active organic amines defined by general formulas (A-1) and (A-2). These resolving agents, such as (S)-1-phenyl-2-(p-tolyl)ethylamine, exhibit superior selectivity even when interacting with substrates possessing minimal initial optical activity. By operating within a temperature range of -20 to 150°C and utilizing solvents like toluene or hexane, the process facilitates the selective crystallization of the desired (+)-trans-chrysanthemic acid diastereomeric salt. This approach effectively decouples the quality of the final product from the purity of the feedstock, allowing producers to feed crude acid streams directly into the resolution unit. The ability to recover and reuse the amine resolving agent further amplifies the economic benefits, creating a closed-loop system that minimizes raw material consumption and waste disposal costs.
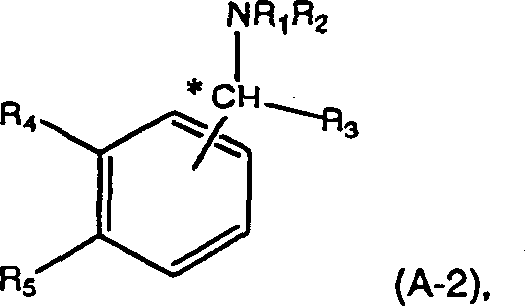
Mechanistic Insights into Asymmetric Copper-Catalyzed Synthesis and Resolution
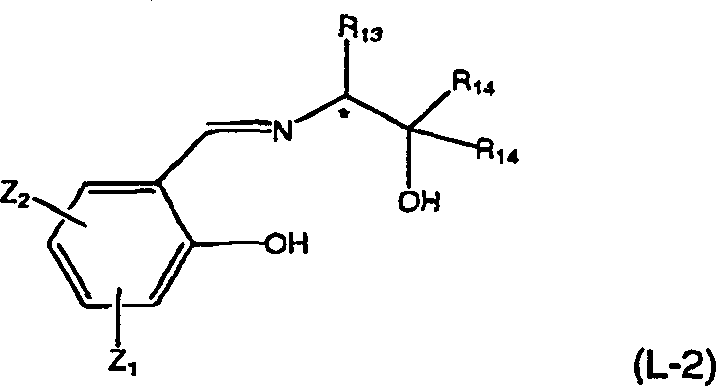
To fully appreciate the value chain integration offered by this technology, one must understand the upstream synthesis of the chrysanthemic acid substrate itself. The patent details the preparation of the requisite chrysanthemic acid esters via an asymmetric cyclopropanation reaction between 2,5-dimethyl-2,4-hexadiene and diazoacetates. This transformation is mediated by highly specialized asymmetric copper complexes formed in situ from copper salts and chiral ligands. The choice of ligand is critical; the patent highlights the efficacy of optically active salicylidene amino alcohol compounds (Formula L-2) and bisoxazoline derivatives. These ligands create a chiral environment around the copper center that directs the stereochemistry of the carbene insertion into the diene, yielding esters with moderate enantiomeric excess. This moderate purity is intentionally sufficient because the downstream resolution step is designed to amplify it, demonstrating a synergistic relationship between the catalytic synthesis and the classical resolution technique.
The resolution mechanism itself relies on the principle of diastereomeric salt formation, where the subtle differences in physical properties between the salt of the (+)-isomer and the (-)-isomer are exploited. When the crude chrysanthemic acid is mixed with the optically active amine in a solvent like toluene, the less soluble diastereomeric salt—containing the target (+)-trans-isomer—precipitates out of the solution upon cooling. The patent specifies that the amount of amine used is typically between 0.3 to 1.2 moles per mole of acid, a stoichiometry optimized to maximize yield while minimizing excess reagent. Following filtration, the salt is decomposed using mineral acids like sulfuric acid or bases like sodium hydroxide. This decomposition releases the free optically active acid into the organic phase while converting the amine into a water-soluble salt or free base, which can then be separated and regenerated. This precise control over solubility and phase separation ensures that the final product achieves optical purities exceeding 90% e.e., suitable for high-grade pyrethroid synthesis.
How to Synthesize Optically Active Chrysanthemic Acid Efficiently
The practical implementation of this patented process involves a sequence of carefully controlled unit operations designed to maximize yield and purity while ensuring operator safety and environmental compliance. The synthesis begins with the preparation of the reaction mixture, where the crude chrysanthemic acid and the specific optically active amine are dissolved in an aromatic or aliphatic hydrocarbon solvent under heated conditions to ensure complete homogeneity. Once the solution is clear, it is subjected to a controlled cooling profile, often allowing it to stand at ambient temperature or actively cooling it to induce nucleation and crystal growth of the target diastereomeric salt. The detailed standardized synthesis steps see the guide below for specific parameters regarding stirring rates, cooling gradients, and filtration techniques essential for reproducibility.
- Dissolve chrysanthemic acid (trans ratio ≥70%, e.e. 2-10%) and an optically active organic amine (0.3-1.2 mol equivalent) in a solvent like toluene under heating.
- Cool the solution to ambient temperature or lower to induce the crystallization of the diastereomeric salt containing the desired (+)-trans-chrysanthemic acid.
- Filter the crystals, decompose the salt with acid or base, and extract the final high-purity optically active chrysanthemic acid while recovering the amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology translates into tangible strategic advantages that extend beyond simple chemical yield improvements. The primary value driver is the drastic simplification of the upstream supply requirements; because the process tolerates low-purity starting materials, manufacturers are no longer beholden to suppliers of expensive, high-enantiomeric excess intermediates. This flexibility allows for the sourcing of cheaper, crude acid streams, effectively lowering the cost of goods sold (COGS) without compromising the quality of the final active ingredient. Furthermore, the integrated recovery system for the resolving amine means that the consumption of this potentially costly chiral auxiliary is minimized, contributing to substantial long-term operational savings and reducing the volatility associated with raw material pricing fluctuations in the fine chemical market.
- Cost Reduction in Manufacturing: The elimination of the need for high-purity starting materials fundamentally alters the cost structure of chrysanthemic acid production. By enabling the use of crude intermediates with optical purity as low as 2% e.e., the process removes the necessity for expensive pre-purification steps or high-cost asymmetric catalysts that demand near-perfect selectivity. Additionally, the capability to recover and recycle the optically active organic amine resolving agent significantly reduces the recurring expenditure on chiral reagents. This closed-loop reagent usage not only lowers direct material costs but also diminishes the environmental burden associated with waste disposal, aligning production practices with increasingly stringent global sustainability mandates and green chemistry principles.
- Enhanced Supply Chain Reliability: Dependence on a single source of high-purity intermediates creates a fragile supply chain vulnerable to disruptions. This technology diversifies the supply base by allowing manufacturers to qualify multiple suppliers of crude chrysanthemic acid, as the resolution step acts as a robust quality gate that standardizes the output regardless of input variability. The use of common, commodity solvents like toluene and standard mineral acids for workup further ensures that the process is not constrained by the availability of exotic reagents. This resilience is critical for maintaining continuous production schedules, especially during peak agricultural seasons when the demand for pyrethroid insecticides surges and supply chain bottlenecks can lead to significant revenue loss.
- Scalability and Environmental Compliance: The process conditions described, such as operation at ambient to moderately elevated temperatures and the use of standard filtration and extraction equipment, indicate a high degree of scalability from pilot plant to commercial tonnage. The ability to perform the resolution in common hydrocarbon solvents facilitates easy solvent recovery through distillation, minimizing volatile organic compound (VOC) emissions. Moreover, the efficient recovery of the amine resolving agent reduces the volume of hazardous chemical waste generated per kilogram of product. These factors collectively simplify the regulatory approval process for new manufacturing sites and ensure that production facilities can meet rigorous environmental, health, and safety (EHS) standards required by multinational agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process feasibility. Understanding these details is crucial for technical teams assessing the integration of this method into existing production lines or for procurement specialists negotiating supply contracts for chrysanthemic acid derivatives.
Q: What is the minimum optical purity required for the starting chrysanthemic acid in this process?
A: According to patent CN1373116A, the process is uniquely capable of resolving chrysanthemic acid with a very low initial optical purity, specifically ranging from 2% e.e. to less than 10% e.e., provided the trans-isomer ratio is not less than 70%.
Q: Can the optically active amine resolving agent be recycled?
A: Yes, the process design explicitly includes a recovery step where the optically active organic amine is extracted from the aqueous layer after salt decomposition, allowing it to be reused in subsequent batches to minimize raw material costs.
Q: What represents the key advantage of this resolution method over conventional techniques?
A: The primary advantage is the ability to efficiently upgrade low-purity intermediates into high-value (+)-trans-chrysanthemic acid (up to 95% e.e.) without requiring expensive, high-purity starting materials, thereby drastically improving the overall economic feasibility of pyrethroid production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chrysanthemic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of processes like CN1373116A are fully realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC capabilities to verify the optical purity and trans/cis ratios of every batch, guaranteeing that our optically active chrysanthemic acid meets the exacting standards required for next-generation pyrethroid formulations.
We invite you to collaborate with us to optimize your supply chain for agrochemical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your product quality while driving down overall production costs. Let us be your partner in delivering high-performance chemical solutions to the global market.
