Revolutionizing Ester Synthesis: Stable Metal-Organic Catalysts for Green Carbonylation
Revolutionizing Ester Synthesis: Stable Metal-Organic Catalysts for Green Carbonylation
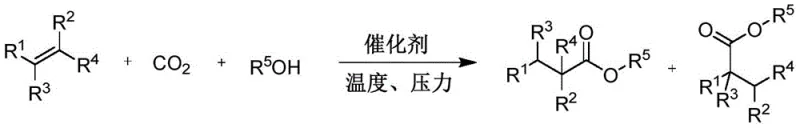
The chemical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by the urgent need to reduce carbon footprints and eliminate hazardous reagents. A groundbreaking development in this arena is detailed in patent CN111068789A, which introduces a novel class of metal-organic catalysts designed for the carbonylation of olefins using carbon dioxide (CO2). This technology represents a significant leap forward for the production of high-value carboxylic acid esters, which serve as critical building blocks in the pharmaceutical and fine chemical sectors. By utilizing CO2 as a simple C1 source and organic alcohols as hydrogen donors, this invention effectively transforms a greenhouse gas into valuable chemical feedstocks. The core innovation lies in the development of structurally stable (P, N, carbene) sulfonate ligands that coordinate with metals such as Ruthenium, Iridium, or Cobalt. This approach not only addresses the environmental challenges associated with traditional carbonylation methods but also offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates. For R&D directors and procurement managers alike, understanding the mechanistic depth and operational safety of this patent is essential for future-proofing supply chains against regulatory pressures and raw material volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of carboxylic acid esters from olefins has relied heavily on the hydroesterification reaction using carbon monoxide (CO) as the carbonyl source. While effective, this conventional methodology is fraught with significant operational and safety drawbacks that hinder modern sustainable manufacturing goals. The primary concern is the extreme toxicity of carbon monoxide, which necessitates rigorous safety protocols, specialized containment infrastructure, and continuous monitoring systems to protect personnel and the environment. Furthermore, traditional processes often operate under harsh conditions involving high temperatures and pressures, which can lead to energy inefficiencies and increased operational costs. From a catalytic perspective, many existing methods depend on noble metals such as Palladium (Pd) or Rhodium (Rh) paired with expensive and air-sensitive phosphine ligands. These catalysts are not only costly to procure but also prone to deactivation, leading to lower turnover numbers and difficulties in product separation. The reliance on such sensitive catalytic systems often results in narrow substrate scopes and poor selectivity, generating complex impurity profiles that require extensive downstream purification, thereby inflating the overall cost of goods sold (COGS) for the final API intermediate.
The Novel Approach
In stark contrast to the hazardous status quo, the technology disclosed in patent CN111068789A offers a transformative solution by replacing toxic CO with benign and abundant CO2. This novel approach leverages a unique class of (P, N, carbene) sulfonate ligands that impart exceptional structural stability to the metal center, allowing the reaction to proceed efficiently under relatively milder conditions compared to traditional high-pressure carbonylation. The use of organic alcohols as hydrogen donors simplifies the reaction stoichiometry and eliminates the need for external hydrogen gas, further enhancing process safety. Crucially, this catalyst system demonstrates broad substrate tolerance, accommodating various substituted styrenes and cyclic olefins with high conversion rates and selectivity. The stability of the sulfonate ligand framework ensures that the catalyst remains active over extended periods, reducing the frequency of catalyst replenishment and minimizing metal contamination in the final product. This shift from toxic CO to CO2 not only aligns with green chemistry principles but also drastically simplifies the regulatory compliance landscape for manufacturing facilities, making it an attractive option for cost reduction in fine chemical manufacturing.
Mechanistic Insights into (P, N, Carbene) Sulfonate Ligand Catalysis
The efficacy of this catalytic system is rooted in the sophisticated design of the ligand architecture, which features a sulfonate group capable of strong coordination with the metal center. As illustrated in the catalyst structures, the ligands (designated as M, Y, and Z types in the patent) create a rigid chelating environment that stabilizes the active metal species against decomposition. The sulfonate moiety acts as a robust anchor, preventing ligand dissociation under the thermal stress of the reaction conditions (100-150°C). This stability is paramount for maintaining high turnover frequencies (TOF) and ensuring consistent product quality batch after batch. The electronic properties of the ligands can be finely tuned by modifying the substituents on the phosphine or carbene rings, such as introducing electron-withdrawing groups like trifluoromethyl (-CF3) or electron-donating alkyl groups. This tunability allows chemists to optimize the electron density at the metal center, facilitating the difficult activation of the thermodynamically stable CO2 molecule. The mechanism likely involves the insertion of CO2 into a metal-alkyl bond followed by alcoholysis, a pathway that is significantly accelerated by the specific geometric constraints imposed by the (P, N, carbene) framework.
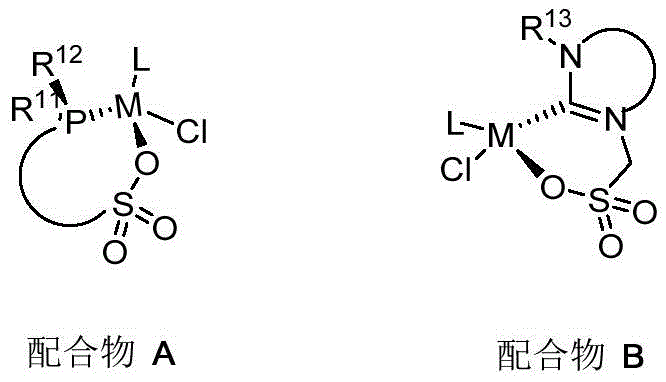
Furthermore, the choice of metal precursor plays a critical role in the catalytic cycle. The patent highlights the effectiveness of Ruthenium (Ru) and Iridium (Ir) complexes, such as [Ru(p-cymene)Cl2]2 and [Ir(C5Me5)Cl2]2. These metals are known for their ability to facilitate hydride transfer and CO2 insertion steps efficiently. The formation of the active catalyst species involves the deprotonation of the sulfonic acid ligand by a base like potassium tert-butoxide, generating an anionic sulfonate that coordinates with the metal cation. This ion-pairing interaction is crucial for solubility in organic solvents like toluene or THF and ensures homogeneous catalysis. Impurity control is inherently built into this mechanism; the high stability of the catalyst minimizes the formation of metal nanoparticles or decomposed ligand byproducts that often plague less robust systems. Consequently, the resulting carboxylic acid esters exhibit high purity, reducing the burden on downstream purification units and ensuring that the final pharmaceutical intermediates meet stringent regulatory specifications for residual metal content.
How to Synthesize High-Purity Carboxylic Acid Esters Efficiently
The synthesis of these valuable esters using the patented catalyst system is a straightforward yet highly controlled process that balances reaction kinetics with safety. The procedure begins with the in-situ or ex-situ preparation of the metal-organic catalyst, where the sulfonic acid ligand is treated with a base and the metal precursor in a degassed solvent. Once the active catalyst is formed, it is introduced to a reaction vessel containing the olefin substrate and the desired alcohol. The system is then pressurized with CO2, typically ranging from 20 to 60 bar, and heated to temperatures between 100°C and 150°C. The reaction time can vary from 12 to 36 hours depending on the specific substrate and desired conversion levels. Acid additives, such as p-toluenesulfonic acid (TsOH), are often employed to promote the reaction rate and improve selectivity towards the linear ester product over branched isomers. This detailed synthetic route allows for the precise manipulation of reaction parameters to maximize yield while minimizing byproduct formation. For a comprehensive understanding of the specific molar ratios, solvent choices, and workup procedures, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the catalyst by reacting sulfonic acid ligands (such as Dppbsa) with metal precursors like [Ru(p-cymene)Cl2]2 or [Ir(C5Me5)Cl2]2 in the presence of a base.
- Charge a reaction vessel with the olefin substrate, organic alcohol (hydrogen donor), the prepared catalyst, and an acid additive like p-toluenesulfonic acid.
- Pressurize the system with CO2 (20-60 bar) and heat to 100-150°C for 12-36 hours to achieve high conversion and selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CO2-based carbonylation technology offers profound strategic advantages that extend beyond mere technical performance. The most immediate benefit is the drastic simplification of raw material sourcing and handling. By eliminating the need for toxic carbon monoxide gas, facilities can avoid the substantial costs associated with specialized gas storage, leak detection systems, and emergency response protocols. This reduction in safety infrastructure translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX). Moreover, the catalyst system utilizes base metals or more abundant noble metals stabilized by easily synthesized ligands, reducing dependency on volatile markets for exotic phosphine ligands. The robustness of the catalyst also implies longer catalyst life and lower consumption rates, which significantly reduces the cost per kilogram of the final product. From a supply chain reliability perspective, the use of CO2 as a feedstock ensures a virtually unlimited and geographically independent supply of the C1 source, insulating production from the logistical disruptions that often affect specialized gas deliveries.
- Cost Reduction in Manufacturing: The transition to this novel catalyst system eliminates the need for expensive and sensitive phosphine ligands that are traditionally required for noble metal catalysis. By utilizing stable sulfonate ligands that can be synthesized from commodity chemicals, the raw material costs are significantly lowered. Additionally, the enhanced stability of the catalyst reduces the frequency of replacement and minimizes product loss due to catalyst decomposition, leading to substantial cost savings over the lifecycle of the manufacturing process. The ability to operate without toxic CO also removes the financial burden of stringent safety compliance and waste treatment associated with hazardous gases.
- Enhanced Supply Chain Reliability: Relying on CO2 as a primary feedstock offers unparalleled supply security, as it is a widely available byproduct of various industrial processes. This abundance ensures that production schedules are not disrupted by shortages of specialized reagents. Furthermore, the catalyst's tolerance to a wide range of substrates means that the same catalytic system can be used to produce a diverse portfolio of ester intermediates, allowing for flexible manufacturing lines that can quickly adapt to market demand changes without requiring extensive retooling or new catalyst qualification.
- Scalability and Environmental Compliance: The homogeneous nature of the catalysis combined with the stability of the ligand system facilitates seamless scale-up from laboratory benchtop to multi-ton commercial production. The process generates minimal hazardous waste, as the primary byproduct is water or unreacted alcohol, which can be easily recycled. This aligns perfectly with increasingly strict environmental regulations regarding greenhouse gas emissions and chemical waste disposal. Adopting this technology positions the supply chain as a leader in sustainability, potentially unlocking green financing opportunities and preferential treatment from environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CO2 fixation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing workflows. The responses cover aspects ranging from catalyst preparation to substrate scope, ensuring that stakeholders have a comprehensive understanding of the operational requirements.
Q: What are the primary advantages of using CO2 over CO in hydroesterification?
A: Using CO2 eliminates the severe safety hazards and toxicity associated with carbon monoxide (CO) gas. Furthermore, CO2 is an abundant, inexpensive C1 source that contributes to green chemistry goals by fixing greenhouse gases into valuable chemical products.
Q: Which metals are effective in this novel catalyst system?
A: The patent demonstrates high efficacy with Ruthenium (Ru) and Iridium (Ir) complexes stabilized by (P, N, carbene) sulfonate ligands. These metals offer a balance of activity and stability superior to traditional nickel or iron systems for this specific transformation.
Q: Does this process require expensive phosphine ligands?
A: No, the invention utilizes structurally stable sulfonic acid-based ligands which are synthesized from readily available starting materials. This avoids the need for highly sensitive or prohibitively expensive phosphine ligands often required in noble metal catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in patent CN111068789A for the production of high-purity carboxylic acid esters. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of CO2 carbonylation reactions safely and effectively. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pharmaceutical intermediate meets the highest global standards. Our commitment to quality ensures that the complex organometallic residues are minimized, delivering products that are ready for the next step in your synthesis pipeline without extensive purification.
We invite you to collaborate with us to leverage this advanced catalytic chemistry for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, safer process. We encourage you to contact our technical procurement team to request specific COA data for our ester intermediates and to discuss route feasibility assessments tailored to your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge technology and a supply chain partner dedicated to driving innovation and efficiency in the pharmaceutical industry.
