Advanced Metalloporphyrin Catalysis for Efficient Carboxylic Acid Ester Production
The chemical industry is constantly seeking more sustainable and efficient pathways for synthesizing essential building blocks, and patent CN101148406A presents a significant breakthrough in the field of oxidative esterification. This intellectual property details a novel method for preparing carboxylic acid esters directly from aldehydes, alcohols, and oxidizing agents, utilizing metalloporphyrin complexes as highly effective catalysts. Unlike traditional methods that rely on harsh acidic conditions, this innovation leverages the unique electronic properties of metalloporphyrins to facilitate oxidation under remarkably mild temperatures and pressures. For R&D directors and process engineers, this represents a paradigm shift towards greener chemistry, offering a route that minimizes waste generation while maximizing atom economy. The technology is particularly relevant for the production of high-value intermediates used in pharmaceuticals, agrochemicals, and advanced polymer materials, where purity and process control are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of carboxylic acid esters has predominantly relied on the Fischer esterification process, which involves the reaction of carboxylic acids with alcohols in the presence of strong inorganic acid catalysts like sulfuric acid. This conventional approach suffers from several critical drawbacks that impact both operational safety and economic efficiency on a commercial scale. The use of corrosive acids necessitates the use of specialized, expensive reactor materials to prevent equipment degradation, leading to higher capital expenditure and maintenance costs. Furthermore, the reaction often requires high temperatures to drive equilibrium, which can result in side reactions, product degradation, and the formation of difficult-to-remove impurities. From an environmental perspective, the disposal of spent acid catalysts and the neutralization of acidic wastewater pose significant regulatory challenges and increase the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology described in patent CN101148406A introduces a catalytic oxidative esterification pathway that bypasses the need for pre-formed carboxylic acids and corrosive mineral acids entirely. By employing metalloporphyrins, the reaction proceeds directly from aldehydes and alcohols using molecular oxygen as the terminal oxidant, which is both abundant and environmentally benign. This novel approach operates under mild conditions, typically around 80°C and low oxygen pressures, drastically reducing energy consumption and thermal stress on the reactants. The flexibility of the catalyst system allows it to be adapted for both intermittent batch processes and continuous flow manufacturing, providing supply chain managers with versatile production options. This shift not only simplifies the downstream purification process due to higher selectivity but also aligns with modern green chemistry principles by eliminating hazardous reagents.
Mechanistic Insights into Metalloporphyrin-Catalyzed Oxidation
The core of this technological advancement lies in the sophisticated coordination chemistry of the metalloporphyrin catalyst, which acts as a biomimetic model for heme-containing enzymes like cytochrome P450. The central metal ion within the porphyrin ring, which can be selected from a wide range of transition metals including Iron, Manganese, Cobalt, and others, serves as the active site for oxygen activation. During the catalytic cycle, the metal center facilitates the transfer of oxygen atoms to the aldehyde substrate, generating a reactive intermediate that subsequently undergoes nucleophilic attack by the alcohol. This mechanism ensures high conversion rates and exceptional selectivity towards the desired ester product, minimizing the formation of over-oxidized byproducts like carboxylic acids. The robustness of the porphyrin ligand framework protects the metal center from deactivation, allowing the catalyst to remain effective even at very low loading levels measured in parts per million.
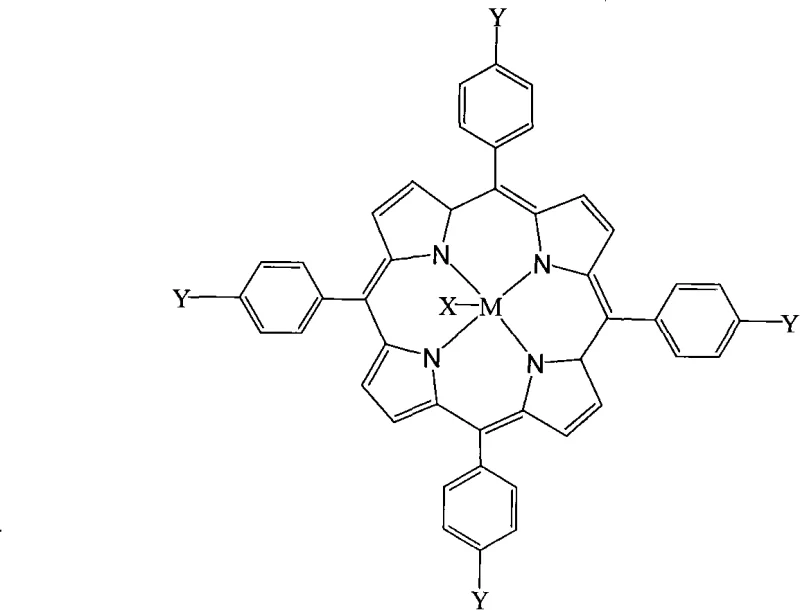
Furthermore, the structural tunability of the metalloporphyrin complex allows for precise optimization of the electronic and steric environment around the active site. As illustrated in the catalyst structure, the substituents denoted by X and Y can be varied among halogens, alkoxyl groups, nitro groups, hydroxyl groups, or alkyl chains to fine-tune the catalyst's performance for specific substrates. This modularity is crucial for R&D teams aiming to suppress specific impurity profiles or enhance reaction rates for sterically hindered aldehydes. The ability to control the impurity spectrum at the molecular level translates directly to reduced purification costs and higher final product quality. Understanding these mechanistic nuances enables process chemists to design robust manufacturing protocols that consistently meet stringent purity specifications required by regulated industries.
How to Synthesize Carboxylic Acid Esters Efficiently
Implementing this oxidative esterification protocol requires careful attention to catalyst preparation and reaction parameter control to achieve the reported high yields and selectivity. The process begins with the dissolution of the metalloporphyrin catalyst in the alcohol solvent, ensuring a homogeneous distribution before the introduction of the aldehyde substrate. Operational simplicity is a key feature, as the catalyst can be added directly to the reaction solution without complex activation steps. For detailed standard operating procedures and specific stoichiometric ratios tailored to your specific aldehyde-alcohol pair, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by weighing specific amounts of metalloporphyrin catalyst and dissolving it in the chosen alcohol solvent within a reactor.
- Add the aldehyde substrate to the reactor and heat the mixture to approximately 80°C while stirring continuously to ensure homogeneity.
- Introduce oxygen gas into the system to maintain a pressure between 0-0.5 MPa and allow the reaction to proceed for about 2 hours before analysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metalloporphyrin-catalyzed process offers substantial strategic advantages beyond mere technical performance. The elimination of corrosive inorganic acids significantly extends the lifespan of reactor vessels and piping, leading to drastic reductions in long-term capital maintenance and replacement costs. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which simplifies the downstream separation and purification train, thereby reducing solvent consumption and waste disposal expenses. These factors combine to create a more cost-effective manufacturing landscape that enhances overall profit margins without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the removal of expensive and hazardous acid catalysts and the associated neutralization waste streams. By operating under mild conditions, the process consumes significantly less energy for heating and cooling compared to traditional high-temperature esterification methods. The low loading requirement of the metalloporphyrin catalyst, often in the ppm range, ensures that catalyst costs do not become a prohibitive factor, even when using precious metal variants. Consequently, the total cost of ownership for the manufacturing process is optimized through reduced raw material waste and lower utility consumption.
- Enhanced Supply Chain Reliability: The versatility of the catalyst system supports both batch and continuous production modes, providing supply chain planners with the flexibility to scale output according to market demand fluctuations. The use of molecular oxygen as an oxidant eliminates the dependency on stoichiometric chemical oxidants that may have volatile supply chains or storage hazards. Furthermore, the mild reaction conditions reduce the risk of thermal runaway incidents, ensuring safer operations and minimizing the potential for unplanned production shutdowns due to safety violations or equipment failure.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the robust nature of the metalloporphyrin catalyst and the simplicity of the reaction engineering. The process generates minimal hazardous waste, aligning with increasingly strict global environmental regulations and reducing the burden on wastewater treatment facilities. This environmental compatibility not only lowers compliance costs but also enhances the corporate sustainability profile, which is becoming a critical factor in supplier selection for multinational corporations committed to green sourcing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative esterification technology. These insights are derived directly from the experimental data and claims presented in the patent literature to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or new facility designs.
Q: What are the advantages of using metalloporphyrin catalysts over traditional acid catalysts?
A: Metalloporphyrin catalysts enable the reaction to proceed under much milder conditions compared to traditional inorganic acid catalysis, significantly reducing equipment corrosion and environmental pollution while improving selectivity.
Q: Can this oxidative esterification process be scaled for continuous production?
A: Yes, the patent data explicitly demonstrates that the method is suitable for both batch and continuous production modes, allowing for flexible manufacturing strategies depending on volume requirements.
Q: Which metals are effective in the metalloporphyrin catalyst structure?
A: The catalyst structure is highly versatile, supporting transition metals such as Iron (Fe), Manganese (Mn), Cobalt (Co), Nickel (Ni), Palladium (Pd), Platinum (Pt), Copper (Cu), Zinc (Zn), Molybdenum (Mo), Silver (Ag), and Cadmium (Cd).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like metalloporphyrin-mediated oxidation in driving the next generation of chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carboxylic acid esters or fine chemical intermediates we produce adheres to the highest international quality standards.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can be customized for your specific product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your supply chain with reliable, high-quality chemical solutions.
