Advanced Synthetic Route for Rosuvastatin Calcium: Enhancing Commercial Scalability and Purity
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with HMG-CoA reductase inhibitors remaining a cornerstone in the management of hypercholesterolemia and atherosclerosis. Patent CN1742000A discloses a novel and highly efficient method for the preparation of Rosuvastatin Calcium, a potent statin known for its superior efficacy compared to earlier generations of lipid-lowering agents. This technical insight report analyzes the synthetic breakthroughs presented in the patent, focusing on the optimization of reaction pathways that directly address the pain points of traditional manufacturing. By shifting away from the cumbersome multi-step side chain generation of prior art, this new route offers a streamlined approach that enhances both chemical yield and operational feasibility. For R&D directors and procurement specialists, understanding the nuances of this condensation and reduction strategy is critical for securing a reliable supply of high-purity API intermediates. The following analysis details how this specific intellectual property translates into tangible commercial advantages, including reduced material costs and improved supply chain continuity for global pharmaceutical manufacturers seeking to optimize their statin production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN1742000A, the industry standard for synthesizing Rosuvastatin was heavily reliant on the methodologies disclosed in US Patent 5,260,440. This conventional approach is characterized by a significant bottleneck: the generation of the phosphorane side chain, which necessitates no fewer than eight distinct synthetic steps. Each of these steps introduces potential points of failure, yield loss, and accumulation of impurities, thereby complicating the purification process and driving up the overall cost of goods sold. Furthermore, the reagents required for this extended sequence are often expensive and specialized, creating a barrier to entry for cost-effective mass production. The time-consuming nature of these eight steps also impacts the lead time for batch completion, making the supply chain less responsive to market fluctuations. For procurement managers, the reliance on such a complex pathway意味着 higher vulnerability to raw material price volatility and logistical delays, rendering the conventional method less attractive for large-scale commercial operations where margin compression is a constant concern.
The Novel Approach
In stark contrast to the legacy methods, the process disclosed in CN1742000A introduces a paradigm shift by utilizing a direct condensation reaction between a specific phosphorane derivative and a pyrimidine aldehyde. This novel approach effectively bypasses the need for the protracted eight-step side chain synthesis, consolidating the construction of the molecular framework into a more direct and atom-economical sequence. The use of 1-cyano-2S-2-[(tert-butyldimethylsilyl)oxy]-4-oxo-5-triphenylphosphoranylidene pentane as a key starting material allows for a more efficient coupling with the pyrimidine core. This reduction in step count not only accelerates the overall production timeline but also significantly lowers the consumption of solvents and reagents. From a commercial perspective, this simplification translates to a drastic reduction in operational overhead and waste generation. The method is explicitly designed to be operated on a commercial scale, offering a robust alternative that aligns with the modern industry's demand for leaner, greener, and more cost-efficient manufacturing processes without compromising the structural integrity of the final active pharmaceutical ingredient.
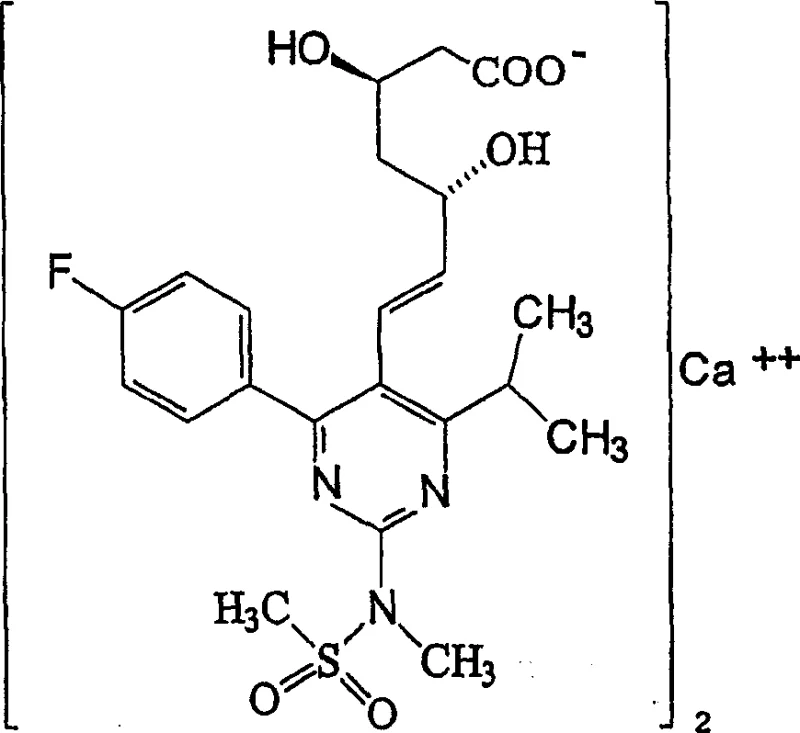
Mechanistic Insights into Condensation and Stereoselective Reduction
The core of this synthetic innovation lies in the precise execution of the condensation and subsequent stereoselective reduction steps, which dictate the optical purity and overall yield of the Rosuvastatin Calcium. The initial condensation reaction involves the coupling of the phosphorane intermediate with 4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonylamino)-5-pyrimidinecarbaldehyde in an organic solvent such as toluene. This step is critical for establishing the carbon-carbon double bond with the correct E-geometry, which is essential for the biological activity of the statin. Following condensation, the deprotection of the tert-butyldimethylsilyl group is performed using acids or fluoride sources, revealing the hydroxyl functionality required for the subsequent reduction. The meticulous control of reaction conditions during these phases ensures that the intermediate cyanoketol is formed with minimal byproduct formation, setting the stage for the high-fidelity transformation into the final diol system. This mechanistic precision is what allows the process to achieve high purity levels, a key metric for R&D directors evaluating the feasibility of technology transfer.
The stereoselective reduction of the cyanoketol to the cyanodiol represents the most technically demanding phase of the synthesis, requiring rigorous temperature control to maintain chiral integrity. The patent specifies the use of diethylmethoxyborane and sodium borohydride in a mixed solvent system containing both alcoholic and non-alcoholic components. The reaction is conducted under cooling conditions, typically between -100°C and 20°C, with a preferred operational window of -80°C to -70°C. This low-temperature environment is crucial for directing the hydride attack to the correct face of the ketone, thereby establishing the (3R, 5S) stereochemistry that defines the active enantiomer of Rosuvastatin. Deviations from these thermal parameters could lead to the formation of diastereomers, which would complicate downstream purification and reduce the overall potency of the drug substance. The ability to consistently reproduce these conditions at scale is a testament to the robustness of the patented process, ensuring that the final product meets the stringent quality specifications required for regulatory approval and patient safety.
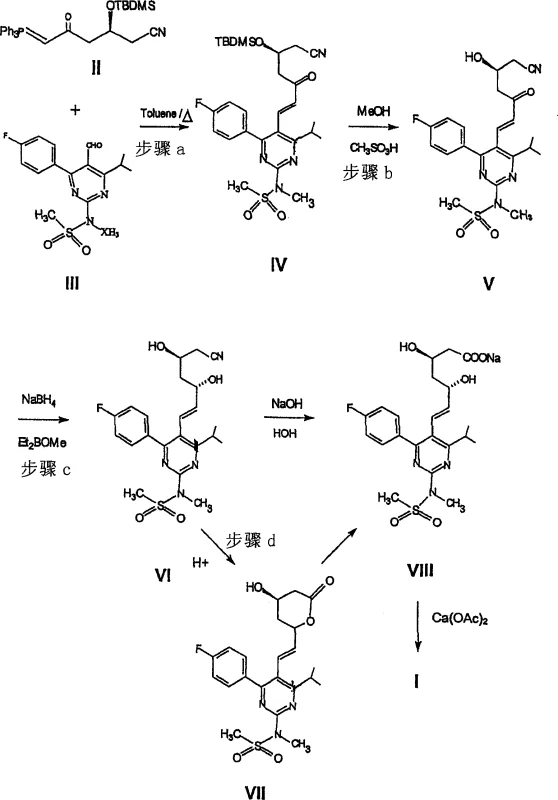
How to Synthesize Rosuvastatin Calcium Efficiently
Implementing this synthesis route requires a disciplined approach to process chemistry, focusing on the four key transformation stages outlined in the patent documentation. The workflow begins with the condensation of the phosphorane and aldehyde precursors, followed by deprotection, reduction, and final hydrolysis to the calcium salt. Each stage demands specific attention to solvent selection, temperature modulation, and work-up procedures to maximize yield and purity. The detailed standardized synthesis steps provided in the guide below are derived directly from the experimental examples within CN1742000A, ensuring that the technical instructions are grounded in verified data. For process engineers and laboratory teams, adhering to these protocols is essential for replicating the commercial success demonstrated in the patent examples. The following section serves as the definitive operational reference for executing this advanced manufacturing pathway.
- Condense 1-cyano-2S-2-[(tert-butyldimethylsilyl)oxy]-4-oxo-5-triphenylphosphoranylidene pentane with 4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonylamino)-5-pyrimidinecarbaldehyde in an organic solvent like toluene.
- Deprotect the tert-butyldimethylsilyl group of the condensation product using an acid or tetrabutylammonium fluoride in an organic solvent to yield the cyanoketol intermediate.
- Reduce the cyanoketol using diethylmethoxyborane and sodium borohydride in a mixed solvent system at low temperatures to form the cyanodiol with high stereocontrol.
- Hydrolyze the cyanodiol with acid to form the lactone, convert to the sodium salt, and finally treat with calcium acetate to obtain the Rosuvastatin Calcium hemi-salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN1742000A process offers substantial strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the significant reduction of manufacturing costs driven by the elimination of the expensive and lengthy phosphorane side chain synthesis found in older methods. By shortening the synthetic route, the process reduces the consumption of high-cost reagents and minimizes the labor hours required for batch processing. This streamlining effect directly impacts the bottom line, allowing for more competitive pricing structures in the global API market. Furthermore, the use of common, readily available solvents such as toluene, methanol, and ethyl acetate mitigates the risk of supply disruptions associated with specialized or hazardous chemicals. This accessibility ensures a more resilient supply chain, capable of maintaining continuous production even during periods of raw material scarcity. The overall simplification of the process also facilitates easier technology transfer between manufacturing sites, enhancing the flexibility of the supply network.
- Cost Reduction in Manufacturing: The economic impact of this novel synthesis route is profound, primarily due to the drastic simplification of the reaction sequence. By removing the need for eight distinct steps to generate the phosphorane side chain, the process eliminates multiple unit operations, each of which carries associated costs for energy, labor, and waste disposal. The reduction in step count also means fewer opportunities for yield loss, resulting in a higher overall output from the same amount of starting material. Additionally, the avoidance of expensive formulations required by the conventional US Patent 5,260,440 method leads to direct savings on raw material procurement. These cumulative efficiencies create a leaner cost structure, enabling manufacturers to offer high-purity Rosuvastatin Calcium at a more attractive price point while maintaining healthy profit margins. The qualitative improvement in cost efficiency makes this route highly desirable for large-volume production contracts.
- Enhanced Supply Chain Reliability: Supply chain continuity is heavily dependent on the availability and stability of raw materials, and this patented process excels in utilizing widely accessible reagents. Unlike methods that rely on niche or custom-synthesized intermediates, the condensation and reduction steps described here utilize standard organic solvents and common reducing agents like sodium borohydride. This reliance on commodity chemicals reduces the risk of bottlenecks caused by supplier constraints or geopolitical trade issues. Moreover, the robustness of the reaction conditions, which tolerate standard industrial equipment and purification techniques like crystallization, ensures that production can be scaled up without requiring exotic infrastructure. For supply chain heads, this translates to a lower risk profile and greater confidence in meeting delivery deadlines. The ability to source materials from multiple vendors further strengthens the supply network, ensuring that production schedules remain uninterrupted.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to commercial production often introduces new challenges, but this method is explicitly designed for industrial feasibility. The reaction conditions, such as the temperature ranges for reduction and the solvent systems for condensation, are compatible with standard reactor vessels and cooling systems found in modern pharmaceutical plants. The process also generates less waste compared to the longer conventional routes, as fewer steps mean fewer byproducts and solvent washes. This reduction in waste volume simplifies effluent treatment and aligns with increasingly strict environmental regulations regarding chemical manufacturing. The ability to purify the product using standard techniques like column chromatography and crystallization further supports scalability, as these methods are well-understood and easily optimized for large batches. Consequently, the process offers a sustainable pathway for meeting global demand while adhering to environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Rosuvastatin Calcium as disclosed in CN1742000A. These answers are derived from the patent's background technology and beneficial effects, providing clarity on how this method compares to existing standards. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their own manufacturing needs. The responses cover aspects of process efficiency, reaction conditions, and scalability, offering a comprehensive overview of the technology's value proposition. This section aims to eliminate commercial doubts and provide the necessary technical context for decision-making.
Q: How does this process improve upon the conventional US Patent 5,260,440 method?
A: The conventional method described in US Patent 5,260,440 requires eight synthetic steps to generate the phosphorane side chain, involving expensive formulations and time-consuming procedures. In contrast, the process disclosed in CN1742000A utilizes a direct condensation strategy that significantly simplifies the synthetic route, reduces the number of operational steps, and employs less expensive reagents, thereby improving overall economic efficiency and production suitability for commercial scales.
Q: What are the critical reaction conditions for the stereoselective reduction step?
A: The stereoselective reduction of the cyanoketol to the cyanodiol is a critical step that requires precise temperature control to ensure high optical purity. The reaction is conducted under cooling conditions, typically ranging from approximately -100°C to 20°C, with a preferred range of -80°C to -70°C. The use of diethylmethoxyborane and sodium borohydride in a mixture of alcoholic and non-alcoholic solvents facilitates the formation of the desired dihydroxyheptenoate stereochemistry essential for biological activity.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the method is explicitly designed to be advantageous for economic efficiency and convenient operation on a commercial scale. By eliminating the need for the complex eight-step phosphorane side chain generation found in prior art, the process reduces material costs and operational complexity. The use of common organic solvents such as toluene, methanol, and ethyl acetate, along with standard purification techniques like crystallization and chromatography, supports robust scalability for industrial production of high-purity Rosuvastatin Calcium.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Supplier
The technical potential of the CN1742000A synthesis route is immense, offering a clear path to high-quality, cost-effective production of this critical cardiovascular medication. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like this stereoselective reduction are executed with precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand the critical nature of API intermediates in the pharmaceutical supply chain and are dedicated to providing a stable, reliable source of Rosuvastatin Calcium that meets the exacting requirements of global regulatory bodies. Our team of experts is ready to collaborate with your R&D and procurement divisions to integrate this advanced manufacturing technology into your supply network.
We invite you to initiate a dialogue regarding your specific supply chain optimization needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this streamlined synthesis route can impact your overall production budget. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project timelines. Our goal is to establish a long-term partnership that drives mutual growth through technical excellence and operational reliability. Let us help you secure a competitive edge in the statin market with our superior manufacturing capabilities and dedication to quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
