Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Commercial Chiral Indolinone Production
The chemical industry is witnessing a significant paradigm shift in the synthesis of complex heterocyclic scaffolds, driven by the urgent demand for enantiomerically pure pharmaceutical intermediates. Patent CN102924360A introduces a groundbreaking methodology for the asymmetric hydrogenation of 3-alkenylene substituted indolinones, utilizing a sophisticated iridium chiral catalyst system. This technology addresses a critical gap in organic synthesis, providing a direct and efficient route to C-3 monosubstituted chiral indolinones, which serve as vital structural motifs in numerous bioactive alkaloids and drug candidates. By leveraging the unique electronic and steric properties of iridium complexes coordinated with specialized P-N ligands, this process achieves remarkable levels of stereocontrol under relatively mild conditions. For R&D directors and process chemists, this represents a robust platform for accessing high-value chiral building blocks that were previously difficult to synthesize with high optical purity. The implications for streamlining the production of anti-tumor agents and other therapeutic compounds are profound, offering a reliable pathway that balances synthetic elegance with practical manufacturability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-3 chiral indolinone cores has relied heavily on indirect strategies that often involve multiple synthetic steps and harsh reaction environments. Prior art, such as the radical cyclization methods reported by Dennis P. Curran, while innovative, frequently suffers from limitations regarding substrate generality and the difficulty in controlling absolute stereochemistry during the bond-forming event. These traditional approaches often require stoichiometric amounts of reagents or generate significant waste streams, complicating the purification process and increasing the overall environmental footprint of the manufacturing campaign. Furthermore, the reliance on radical intermediates can lead to side reactions and lower selectivity, necessitating extensive chromatographic separations that are costly and time-consuming on a large scale. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, creating bottlenecks in the supply chain for critical active pharmaceutical ingredients. The lack of a direct catalytic asymmetric method has long been a recognized bottleneck in the field, limiting the ability to rapidly iterate on molecular designs during drug discovery phases.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102924360A employs a direct asymmetric hydrogenation strategy that transforms 3-alkenylene substituted indolinones into their chiral counterparts with exceptional efficiency. This novel approach utilizes a cationic iridium complex bearing a chiral P-N ligand, which activates molecular hydrogen and transfers it to the exocyclic double bond with high facial selectivity. The reaction proceeds smoothly in common organic solvents such as dichloromethane or toluene, eliminating the need for exotic or hazardous reagents. By operating under moderate hydrogen pressures ranging from 10 to 50 bar and temperatures between -10°C and 40°C, the process ensures safety and operational simplicity while maintaining high reaction rates. This direct functionalization not only improves atom economy but also significantly reduces the number of processing steps required to reach the target molecule. For supply chain heads, this simplification means reduced inventory holding costs and a more resilient production schedule, as the reliance on complex multi-step sequences is minimized. The versatility of this method allows for the accommodation of various substituents on the indolinone ring, making it a versatile tool for diverse medicinal chemistry programs.

Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this transformation hinges on the precise design of the iridium chiral catalyst, which acts as the molecular engine driving the stereoselective reduction. The catalyst system is composed of an iridium(I) center coordinated with cyclooctadiene (COD), a chiral ligand selected from the L1 to L9 library, and a non-coordinating anion such as BArF or PF6. The chiral ligands, predominantly featuring an oxazoline-phosphine backbone, create a well-defined chiral pocket around the metal center. This steric environment dictates the approach of the substrate, ensuring that hydrogen delivery occurs exclusively from one face of the planar alkene moiety. The electronic tuning of the phosphine and the steric bulk of the oxazoline substituents are critical parameters that influence both the activity and the enantioselectivity of the catalyst. Understanding this interplay allows chemists to fine-tune the reaction conditions to maximize the enantiomeric excess (ee) for specific substrates. For instance, subtle changes in the ligand structure can switch the preference for one enantiomer over the other, providing a powerful handle for optimizing the synthesis of specific drug candidates. This level of control is essential for meeting the stringent purity specifications required by regulatory agencies for pharmaceutical products.
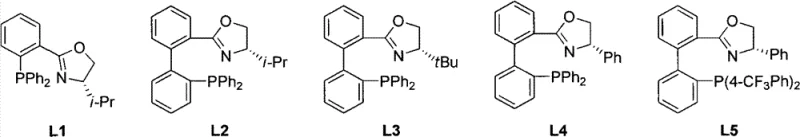
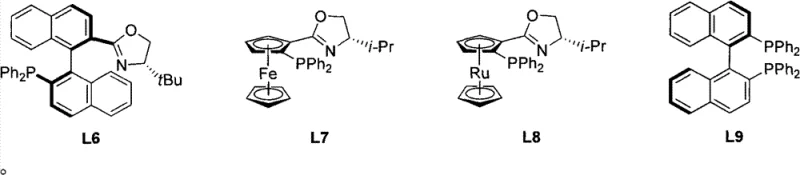
Beyond the primary catalytic cycle, the mechanism also involves careful management of impurity profiles to ensure the quality of the final product. The choice of solvent plays a pivotal role in stabilizing the cationic iridium species and facilitating the heterolytic cleavage of hydrogen. Solvents like dichloromethane and trifluorotoluene have been shown to support high turnover frequencies while minimizing catalyst decomposition. Additionally, the presence of the bulky anion helps to prevent the formation of inactive catalyst dimers, thereby extending the catalyst lifetime and allowing for lower catalyst loadings. This is particularly important for cost reduction in API manufacturing, as precious metal residues must be kept to trace levels. The robustness of the catalyst system against various functional groups present on the substrate further enhances its utility, preventing unwanted side reactions such as over-reduction or isomerization. By rigorously controlling these mechanistic variables, manufacturers can achieve consistent batch-to-batch reproducibility, a key metric for supply chain reliability. The ability to predict and control the impurity profile through mechanistic understanding empowers quality control teams to implement tighter specifications and faster release testing protocols.
How to Synthesize Chiral Indolinones Efficiently
Implementing this patented synthesis route requires a systematic approach to catalyst preparation and reaction engineering to fully realize its potential benefits. The process begins with the in situ or pre-formation of the active iridium species, followed by the introduction of the 3-alkenylene substituted indolinone substrate under an inert atmosphere. Detailed operational parameters, including specific molar ratios and agitation speeds, are critical for maximizing mass transfer in the gas-liquid-solid heterogeneous system. The following guide outlines the standardized procedure derived from the patent examples to ensure optimal performance and safety during scale-up. Adhering to these protocols allows production teams to leverage the full efficiency of the iridium catalytic system while maintaining strict compliance with safety standards.
- Prepare the iridium chiral catalyst by combining Iridium(I), cyclooctadiene (COD), a selected chiral ligand (L1-L9), and an anion source in a suitable solvent.
- Dissolve the 3-alkenylene substituted indolinone substrate in a dry solvent such as dichloromethane or toluene within a high-pressure autoclave.
- Pressurize the system with hydrogen gas to 10-50 bar and maintain the reaction temperature between -10°C and 40°C until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this iridium-catalyzed hydrogenation technology offers substantial strategic advantages for organizations focused on cost optimization and supply chain resilience. By replacing multi-step synthetic sequences with a single catalytic step, companies can drastically simplify their manufacturing workflows, leading to significant reductions in labor, energy, and equipment usage. The mild reaction conditions eliminate the need for extreme temperatures or pressures, lowering the capital expenditure required for specialized reactor infrastructure. Furthermore, the high selectivity of the catalyst minimizes the formation of by-products, reducing the burden on downstream purification units and decreasing solvent consumption. These factors collectively contribute to a leaner, more agile production model that can respond quickly to market demands. For procurement managers, the ability to source high-quality intermediates with a simplified bill of materials translates directly into improved margin protection and competitive pricing strategies. The technology aligns perfectly with green chemistry principles, enhancing the sustainability profile of the manufacturing process which is increasingly valued by global partners.
- Cost Reduction in Manufacturing: The implementation of this catalytic method drives cost efficiency primarily through the elimination of expensive stoichiometric reagents and the reduction of waste disposal costs associated with traditional radical methods. The high turnover number of the iridium catalyst allows for the use of minimal metal loading, which significantly lowers the cost of goods sold despite the use of a precious metal. Additionally, the simplified work-up procedures reduce the consumption of chromatography media and solvents, further driving down operational expenses. This economic efficiency makes the production of complex chiral indolinones commercially viable for a broader range of therapeutic applications. By optimizing the catalyst lifecycle and recycling potential, manufacturers can achieve substantial cost savings over the long term without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this hydrogenation process ensures a steady and predictable supply of critical intermediates. The use of stable and commercially available starting materials mitigates the risk of raw material shortages that often plague complex synthetic routes. The tolerance of the catalyst to various functional groups allows for a flexible supply chain where different substrate variants can be processed using the same core technology platform. This flexibility reduces the need for dedicated production lines for each specific molecule, maximizing asset utilization and throughput. Consequently, lead times for high-purity intermediates can be significantly shortened, enabling faster time-to-market for new drug formulations. The reliability of the process also reduces the risk of batch failures, ensuring continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard high-pressure hydrogenation equipment that is widely available in contract manufacturing organizations. The mild operating conditions facilitate safe scale-up from laboratory to pilot and commercial production scales without encountering significant heat transfer or mixing limitations. Moreover, the high atom economy and reduced solvent usage align with stringent environmental regulations, minimizing the generation of hazardous waste. This compliance reduces the regulatory burden and associated costs of environmental permitting and waste treatment. The ability to operate efficiently within existing infrastructure allows for rapid deployment of capacity to meet surging demand. This scalability ensures that the technology can support the growing global need for chiral pharmaceutical ingredients while maintaining a sustainable environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical application of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers highlight the versatility and robustness of the iridium catalyst system in real-world scenarios.
Q: What is the primary advantage of this iridium-catalyzed method over previous radical cyclization techniques?
A: Unlike prior radical cyclization methods which often suffer from limited substrate scope and harsh conditions, this asymmetric hydrogenation offers mild reaction conditions, higher atom economy, and direct access to C-3 chiral centers with excellent yields.
Q: Which chiral ligands are most effective for this hydrogenation process?
A: The patent identifies a library of P-N type ligands (L1-L9), specifically oxazoline-phosphine derivatives, as highly effective. Ligand selection significantly impacts enantioselectivity, with specific variants achieving up to 91% ee depending on the substrate substituents.
Q: Can this process be scaled for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes standard hydrogenation equipment and operates under moderate pressures (10-50 bar) and temperatures (-10°C to 40°C), making it highly suitable for commercial scale-up and consistent supply of high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102924360A for the future of pharmaceutical manufacturing. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering high-purity chiral indolinones that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch of material supplied adheres to the highest international standards, providing our partners with the confidence they need to advance their clinical and commercial programs. By leveraging our deep technical expertise, we help clients navigate the complexities of chiral synthesis to achieve their project goals efficiently.
We invite you to collaborate with us to explore how this iridium-catalyzed hydrogenation technology can optimize your specific supply chain requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and quality targets. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the production of next-generation therapeutic agents, ensuring a reliable supply of critical intermediates for the global healthcare market.
