Advanced Asymmetric Synthesis of Chiral Indolinone and Angelica Lactone Frameworks for Pharmaceutical Applications
Introduction to High-Efficiency Chiral Framework Synthesis
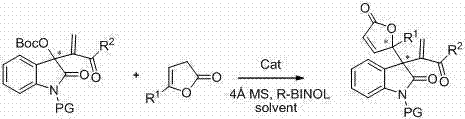
The pharmaceutical industry continuously demands robust and scalable methods for constructing complex chiral scaffolds, particularly those containing indolinone and angelica lactone motifs which are prevalent in bioactive natural products and drug candidates. Patent CN102584800A introduces a groundbreaking asymmetric synthesis method that addresses these needs by utilizing indolinone Morita-Baylis-Hillman (MBH) carbonates and various angelica lactone compounds. This technology leverages chiral amines, specifically derivatives of cinchona alkaloids like beta-isocupreidine (beta-ICD), to achieve high-efficiency and high-enantioselective allylic alkylation. The process is notable for its operational simplicity, mild reaction conditions, and the ability to generate multifunctional products with excellent stereocontrol, positioning it as a critical asset for the development of reliable pharmaceutical intermediate supplier networks.
The significance of this invention lies in its capacity to construct quaternary carbon centers at the C-3 position of the indolinone ring, a structural feature that is notoriously difficult to access with high optical purity using conventional methods. By employing additives such as 4A molecular sieves and R-BINOL alongside the organocatalyst, the reaction system achieves a level of precision that minimizes impurity formation. This technical advancement not only streamlines the synthetic route but also enhances the overall quality of the resulting intermediates, ensuring they meet the stringent purity specifications required for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3-disubstituted indolinone compounds has presented significant challenges to process chemists, primarily due to the steric hindrance associated with forming quaternary stereocenters. Conventional approaches often rely on transition metal catalysis or harsh reaction conditions that can compromise the integrity of sensitive functional groups present in complex molecules. Furthermore, many traditional methods suffer from limited substrate scope, failing to accommodate diverse substitution patterns on the indolinone aromatic ring or the lactone moiety. This lack of versatility often necessitates the development of bespoke synthetic routes for each new analog, leading to increased R&D timelines and higher costs in cost reduction in pharmaceutical intermediate manufacturing.
Another critical drawback of older methodologies is the difficulty in achieving high enantiomeric excess without extensive and costly purification steps. The presence of racemic byproducts or diastereomers can complicate the isolation of the desired active pharmaceutical ingredient, thereby reducing overall process efficiency. Additionally, the use of heavy metal catalysts raises concerns regarding residual metal contamination, which requires additional downstream processing to meet regulatory limits. These factors collectively contribute to longer lead times and reduced supply chain reliability for high-purity chiral intermediates.
The Novel Approach
The methodology disclosed in CN102584800A represents a paradigm shift by utilizing organocatalysis to overcome these historical barriers. By employing chiral cinchona alkaloid-derived catalysts, the reaction proceeds under mild conditions, typically at temperatures around -10°C, which preserves the stability of sensitive functional groups. This approach eliminates the need for transition metals, inherently solving the issue of heavy metal residue and simplifying the workup procedure. The reaction demonstrates remarkable tolerance for a wide array of substituents, including halogens, alkyl groups, and alkoxy groups on the indolinone core, as well as various alkyl substitutions on the angelica lactone ring.
Moreover, the inclusion of specific additives like 4A molecular sieves and R-BINOL plays a pivotal role in enhancing both the yield and enantioselectivity of the transformation. The synergy between the chiral amine catalyst and these additives creates a highly organized transition state that favors the formation of a single enantiomer. This results in products with enantiomeric excess values generally ranging from 80% to 92% and isolated yields between 70% and 91%. Such performance metrics indicate a highly efficient process that significantly reduces the burden on purification resources, thereby facilitating the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Allylic Alkylation
The success of this asymmetric synthesis relies heavily on the unique bifunctional nature of the cinchona alkaloid-derived catalysts. These catalysts typically possess both a basic tertiary amine site and a hydrogen-bond donor group (such as a hydroxyl group), allowing them to activate both the nucleophile and the electrophile simultaneously. In this specific transformation, the catalyst likely activates the angelica lactone through deprotonation to form a reactive enolate species, while concurrently activating the indolinone MBH carbonate via hydrogen bonding interactions with the carbonate leaving group. This dual activation lowers the energy barrier for the nucleophilic attack and ensures that the reaction occurs within a chiral environment defined by the rigid backbone of the cinchona scaffold.
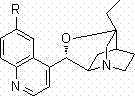
The stereochemical outcome is dictated by the spatial arrangement of the catalyst's substituents, which shield one face of the reacting species while exposing the other. The presence of R-BINOL as an additive further modulates the chirality of the reaction medium, potentially through the formation of a transient chiral complex with the catalyst or by modifying the solvation shell around the transition state. This fine-tuning of the catalytic environment is crucial for achieving the high levels of enantioselectivity observed in the experimental data. Understanding these mechanistic nuances allows process chemists to optimize reaction parameters such as solvent choice and temperature to maximize efficiency.
From an impurity control perspective, the mildness of the organocatalytic conditions minimizes side reactions such as polymerization or decomposition of the MBH carbonate, which are common pitfalls in metal-catalyzed variants. The high specificity of the catalyst ensures that the formation of undesired diastereomers is suppressed, leading to a cleaner crude reaction profile. This inherent selectivity reduces the complexity of the downstream purification process, often allowing for simple column chromatography to afford the pure product. For R&D teams, this means a more predictable and robust process that can be reliably transferred from the laboratory to pilot plant scales.
How to Synthesize Chiral Indolinone-Angelica Lactone Derivatives Efficiently
Implementing this synthesis protocol requires careful attention to reaction conditions and reagent quality to replicate the high performance reported in the patent. The general procedure involves combining the indolinone MBH carbonate and the angelica lactone derivative in an appropriate organic solvent, such as mesitylene or dichloromethane, in the presence of the chiral catalyst and additives. The reaction is typically conducted at low temperatures to maintain stereocontrol, followed by a standard aqueous workup and purification via silica gel chromatography. The detailed standardized synthesis steps are outlined in the guide below.
- Prepare the reaction mixture by adding the chiral cinchona alkaloid-derived b-ICD catalyst, indolinone MBH carbonate, and angelica lactone compound into a clean reaction vessel containing an organic solvent such as mesitylene.
- Maintain the reaction temperature at approximately -10°C and stir the mixture for a duration ranging from 24 to 72 hours to ensure complete conversion.
- Upon completion, isolate the final chiral allylic alkylation product using column chromatography with a petroleum ether and ethyl acetate solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic technology offers substantial strategic benefits beyond mere technical performance. The elimination of expensive and scarce transition metal catalysts directly translates to significant cost savings in raw material acquisition. Furthermore, the avoidance of heavy metals simplifies the regulatory compliance landscape, reducing the need for specialized metal scavenging resins and the associated validation testing. This streamlined workflow enhances the overall economic viability of producing these complex intermediates, making it an attractive option for cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The use of readily available cinchona alkaloid derivatives as catalysts significantly lowers the input cost compared to precious metal complexes. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cryogenic cooling, contributing to a lower carbon footprint and reduced utility costs. The high yields and selectivity minimize waste generation, further optimizing the material balance and reducing disposal expenses.
- Enhanced Supply Chain Reliability: Since the catalysts and starting materials are commercially accessible and do not rely on geopolitically sensitive rare earth elements, the supply chain is more resilient to disruptions. The robustness of the reaction across a wide substrate scope means that a single platform technology can be used to produce a diverse library of intermediates, reducing the need for multiple specialized suppliers and consolidating procurement efforts.
- Scalability and Environmental Compliance: The absence of toxic heavy metals aligns perfectly with green chemistry principles and increasingly strict environmental regulations. This facilitates easier permitting for large-scale production facilities and reduces the risk of environmental liabilities. The simplicity of the workup and purification steps ensures that the process can be scaled up from grams to kilograms without encountering the nonlinear scale-up issues often seen with complex metal-catalyzed reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived from the specific technical disclosures and beneficial effects highlighted in the patent documentation, providing clarity for stakeholders evaluating this process for their own manufacturing pipelines.
Q: What are the key advantages of using cinchona alkaloid catalysts for this synthesis?
A: The use of chiral cinchona alkaloid-derived catalysts allows for mild reaction conditions (-10°C) and avoids the need for expensive transition metals, resulting in high enantioselectivity (up to 92% ee) and yields between 70% and 91%.
Q: Can this method be applied to a wide range of substrates?
A: Yes, the patent demonstrates a broad substrate scope, accommodating various substituents on the indolinone aryl ring (such as F, Cl, Br, methyl, methoxy) and different angelica lactone derivatives, making it versatile for diverse intermediate synthesis.
Q: What downstream transformations are possible with these products?
A: The resulting multifunctional allylic alkylation products serve as versatile precursors that can undergo further transformations, such as cyclization with benzylamine or nitromethane addition, to construct complex polycyclic scaffolds found in bioactive natural products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral building blocks for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patents like CN102584800A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral indolinone and angelica lactone intermediates meets the exacting standards required by global pharmaceutical companies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in organocatalysis can accelerate your drug development timeline while optimizing your manufacturing budget.
