Advanced Asymmetric Synthesis of Chiral Indolinone Spiropentacyclic Skeletons for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral scaffolds, particularly those containing indolinone spiropentacyclic skeletons which are prevalent in bioactive natural products. Patent CN102180828A discloses a groundbreaking approach to efficiently synthesizing these challenging structures through a highly enantioselective 3+2 cycloaddition reaction. This innovation leverages chiral amine catalysts, specifically derivatives of cinchona alkaloids like beta-ICD, to drive the transformation of indolinone MBH carbonates with various dipolarophiles. The significance of this technology lies in its ability to generate multifunctional chiral products with exceptional regio- and enantioselectivity under remarkably mild conditions. Unlike traditional methods that often require harsh reagents or expensive transition metals, this organocatalytic strategy offers a greener and more sustainable pathway for producing high-value pharmaceutical intermediates. The broad substrate scope reported in the patent indicates versatility, allowing for the introduction of diverse functional groups essential for downstream drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of optically pure indolinone spiro skeletons has relied heavily on stoichiometric chiral auxiliaries or transition metal catalysis, both of which present significant drawbacks for modern manufacturing. Conventional routes often necessitate the use of heavy metals which pose severe environmental hazards and require rigorous removal steps to meet stringent pharmaceutical purity standards. Furthermore, these traditional processes frequently operate under extreme temperatures or pressures, leading to higher energy consumption and increased operational risks in a production setting. The generation of stoichiometric waste from chiral auxiliaries not only inflates material costs but also complicates waste management protocols, thereby reducing the overall atom economy of the synthesis. Additionally, achieving high enantiomeric excess with older methods often requires extensive optimization and multiple recrystallization steps, which drastically reduces overall yield and extends production lead times. These inefficiencies create bottlenecks in the supply chain, making it difficult to scale up production to meet commercial demand without compromising on quality or cost.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes an organocatalytic system that eliminates the need for toxic transition metals while maintaining superior stereocontrol. By employing cinchona alkaloid-derived catalysts, the reaction proceeds through a well-defined chiral environment that ensures high enantioselectivity without the burden of metal contamination. This method operates at ambient temperature (25°C), significantly reducing energy requirements and simplifying the engineering controls needed for safe operation. The use of 4Å molecular sieves as additives further enhances reaction efficiency by managing water content, ensuring consistent performance across different batches. The operational simplicity of this protocol allows for straightforward workup procedures, typically involving standard column chromatography, which streamlines the purification process. Consequently, this new route offers a more sustainable and economically viable solution for producing complex chiral intermediates, aligning perfectly with the industry's shift towards green chemistry principles.
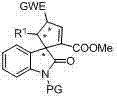
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Cycloaddition
The core of this synthetic breakthrough lies in the intricate mechanistic pathway facilitated by the chiral beta-ICD catalyst, which activates the indolinone MBH carbonate through a network of non-covalent interactions. The catalyst likely functions by forming hydrogen bonds with the carbonate moiety, thereby increasing its electrophilicity and orienting it precisely for nucleophilic attack by the dipolarophile. This activation mode creates a rigid chiral pocket that dictates the facial selectivity of the incoming reagent, ensuring that the 3+2 cycloaddition occurs with high fidelity to produce the desired enantiomer. The presence of the quinoline and quinuclidine rings in the catalyst structure provides the necessary steric bulk to shield one face of the reactive intermediate, effectively minimizing the formation of unwanted stereoisomers. Understanding this mechanism is crucial for R&D teams as it highlights the importance of catalyst loading and solvent choice in maintaining the integrity of the chiral environment throughout the reaction course. The robustness of this catalytic cycle allows for the tolerance of various electron-withdrawing groups on the dipolarophile, expanding the chemical space accessible through this methodology.

Beyond stereocontrol, the mechanism also inherently supports superior impurity control, which is a critical parameter for regulatory compliance in pharmaceutical manufacturing. The high specificity of the organocatalyst reduces the formation of side products that typically arise from non-selective background reactions or competing pathways. By avoiding metal catalysts, the process eliminates the risk of metal-catalyzed decomposition or oligomerization that can plague transition metal-mediated reactions. The mild reaction conditions further prevent thermal degradation of sensitive functional groups, preserving the structural integrity of the final product. This clean reaction profile simplifies downstream purification, as fewer impurities need to be separated, resulting in higher overall recovery rates. For quality assurance teams, this means a more consistent impurity profile across batches, facilitating easier validation and reducing the risk of batch rejection due to out-of-specification impurities.
How to Synthesize Chiral Indolinone Spiro Compounds Efficiently
To implement this synthesis effectively, one must adhere to the specific protocol outlined in the patent which balances catalyst activity with reaction kinetics. The process begins with the precise weighing of the indolinone MBH carbonate and the chosen dipolarophile, ensuring the molar ratios align with the optimized conditions of 1 to 2:1 respectively. The reaction is conducted in an organic solvent such as m-xylene, which provides the appropriate polarity to solubilize reactants while supporting the catalytic cycle. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining indolinone MBH carbonate and various dipolarophile compounds in an organic solvent such as m-xylene.
- Add the chiral quinine-derived beta-ICD derivative catalyst and 4Å molecular sieves to the mixture under inert atmosphere.
- Stir the reaction at 25°C for 12 to 24 hours, then separate the product using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial strategic advantages by decoupling production from the volatility associated with precious metal catalysts. The reliance on organocatalysts derived from renewable cinchona alkaloids ensures a more stable supply chain, as these materials are not subject to the same geopolitical constraints as rare earth metals. This shift significantly mitigates the risk of supply disruptions that can halt production lines and delay product launches in a competitive market. Furthermore, the simplified purification process reduces the consumption of chromatography media and solvents, leading to direct operational cost savings. The ability to run reactions at ambient temperature also lowers utility costs, contributing to a reduced carbon footprint which is increasingly important for corporate sustainability goals. Overall, adopting this method enhances supply chain resilience while driving down the total cost of ownership for these critical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the need for costly metal scavenging resins and extensive analytical testing for residual metals. This simplification of the downstream processing workflow directly translates to lower manufacturing overheads and reduced waste disposal costs. Additionally, the high yields reported in the patent minimize the amount of starting material required per unit of product, optimizing raw material expenditure. The operational simplicity allows for shorter cycle times, increasing the throughput of existing manufacturing assets without the need for capital-intensive equipment upgrades. These factors combine to create a leaner manufacturing process that is highly responsive to market demand fluctuations.
- Enhanced Supply Chain Reliability: Sourcing chiral organocatalysts is generally more straightforward than securing specialized transition metal complexes, which often have long lead times and limited suppliers. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality or minor environmental fluctuations. This reliability ensures consistent delivery schedules, allowing procurement managers to maintain lower safety stock levels and improve working capital efficiency. The scalability of the process from gram to kilogram scale without significant re-optimization further strengthens supply continuity. Consequently, partners can rely on a steady flow of high-quality intermediates to support their own production timelines.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates easier scale-up, as heat management and pressure containment are less critical compared to exothermic metal-catalyzed processes. This reduces the engineering complexity required for commercial reactors, accelerating the timeline from pilot plant to full-scale production. Moreover, the absence of heavy metals simplifies regulatory filings and environmental compliance reporting, reducing the administrative burden on EHS teams. The use of common organic solvents and recyclable molecular sieves aligns with green chemistry initiatives, enhancing the company's environmental stewardship profile. This combination of scalability and compliance makes the technology an attractive option for long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio. We encourage technical teams to review these points thoroughly before initiating any pilot studies or procurement discussions.
Q: What level of enantioselectivity can be achieved with this synthesis method?
A: According to patent CN102180828A, this asymmetric synthesis method achieves high enantioselectivity, typically ranging from 76% to 99% ee, depending on the specific substrate and reaction conditions employed.
Q: What type of catalyst is utilized in this novel synthetic route?
A: The process utilizes chiral amine catalysts, specifically derivatives of cinchona alkaloids such as beta-ICD ((3S,8R,9S)-10,11-dihydro-3,9-epoxy-6'-hydroxycinchonan), which are known for their effectiveness in organocatalytic transformations.
Q: Is this synthetic method suitable for large-scale commercial production?
A: Yes, the method features mild reaction conditions (25°C), simple operation, and readily available catalysts, which collectively suggest strong potential for scalability and commercial viability in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indolinone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at adapting complex organocatalytic routes like the one described in CN102180828A to fit large-scale reactor configurations while maintaining stringent purity specifications. We utilize rigorous QC labs to ensure that every batch meets the highest standards for enantiomeric excess and chemical purity required by global regulatory bodies. Our commitment to quality ensures that the chiral indolinone intermediates supplied are ready for immediate use in sensitive pharmaceutical applications without further purification.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your project volume. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your unique requirements. Contact us today to secure a reliable supply of these high-value chiral building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
