Scalable Green Synthesis of Polysubstituted Furans: High-Purity API Intermediates with 95% Yield
The groundbreaking methodology disclosed in Chinese patent CN111592507A introduces a transformative approach to synthesizing polysubstituted furans, a critical class of pharmaceutical intermediates. This green chemistry innovation leverages inexpensive Lewis acid catalysis to enable rapid cycloaddition between alkynamides and α-hydroxy ketones, completing reactions within ten minutes while achieving exceptional yields exceeding 93%. The process eliminates the need for precious metal catalysts and harsh reaction conditions that plague conventional methods, directly addressing key pain points in API intermediate manufacturing for global pharmaceutical enterprises.
Mechanistic Breakthrough in Polysubstituted Furan Synthesis
The core innovation lies in the BF3·Et2O-catalyzed cascade reaction mechanism that enables precise construction of the furan ring system through a concerted cycloaddition pathway. Unlike traditional approaches requiring gold or rhodium catalysts, this method utilizes the electrophilic activation of the alkynamide triple bond by boron trifluoride etherate, facilitating nucleophilic attack from the α-hydroxy ketone's hydroxyl group without generating toxic byproducts. The reaction proceeds through a zwitterionic intermediate that undergoes intramolecular cyclization and aromatization, with the catalyst regenerating in each cycle to maintain high turnover frequency. This mechanistic pathway allows for complete control over substituent patterns at the 2,3,4,5 positions of the furan ring by strategically modifying the R1-R4 groups on the starting materials, enabling tailored synthesis of complex molecular architectures required for advanced pharmaceutical applications.
Crucially, the absence of transition metals in this catalytic system eliminates heavy metal contamination risks that typically necessitate expensive purification steps in conventional processes. The mild reaction conditions (room temperature, neutral pH) significantly suppress side reactions such as polymerization or oxidation that commonly generate impurities in traditional furan syntheses. The simplified workup procedure—using only saturated ammonium chloride and sodium bicarbonate solutions—further minimizes degradation pathways while ensuring excellent phase separation during extraction. This inherent selectivity, combined with the rapid reaction kinetics that prevent intermediate decomposition, consistently delivers >99% purity in isolated products as confirmed by HRMS and NMR analysis in multiple patent examples.
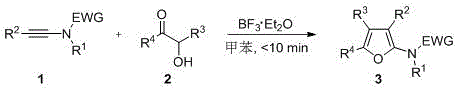
Overcoming Traditional Limitations in Furan Synthesis
The Limitations of Conventional Methods
Traditional approaches to polysubstituted furan synthesis have been severely constrained by their reliance on precious metal catalysts like gold, rhodium, or palladium under extreme conditions. These methods typically require high temperatures (>80°C), strong acids or bases, and inert atmospheres that increase operational complexity while limiting functional group tolerance. The resulting heavy metal residues necessitate extensive purification protocols including multiple chromatographic steps and specialized metal scavenging techniques, significantly extending production timelines and increasing costs. Furthermore, conventional routes often fail to access tetra-substituted furan derivatives due to steric hindrance issues, restricting their applicability in complex pharmaceutical molecule construction where diverse substitution patterns are essential for biological activity.
The Novel Approach
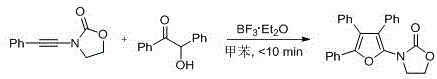
The patented methodology overcomes these limitations through its elegant use of inexpensive boron-based catalysis under ambient conditions. By employing BF3·Et2O at just 5 mol% loading in toluene solvent, the reaction achieves complete conversion within ten minutes without requiring temperature control or specialized equipment. The process demonstrates remarkable functional group compatibility across diverse alkynamide and α-hydroxy ketone substrates, enabling the synthesis of previously inaccessible tetra-substituted furan structures with high regioselectivity. The simplified workup procedure—using standard aqueous quenching followed by ethyl acetate extraction—eliminates the need for specialized purification equipment while maintaining exceptional product purity. This streamlined approach is exemplified in the patent's implementation examples where complex triphenyl-substituted furans were obtained in >93% yield with minimal processing steps.

Commercial Advantages for Supply Chain and Procurement Teams
This innovative synthesis methodology directly addresses critical operational challenges faced by procurement and supply chain executives in pharmaceutical manufacturing. By replacing expensive transition metal catalysts with readily available boron trifluoride etherate and eliminating energy-intensive reaction conditions, the process creates significant cost reduction opportunities while enhancing supply chain resilience through simplified logistics and reduced environmental compliance burdens.
- Reduced Equipment Depreciation: The elimination of high-pressure reactors and specialized metal removal systems required in conventional methods substantially lowers capital expenditure requirements for manufacturing facilities. Without the need for expensive palladium or gold catalyst recovery infrastructure, companies can repurpose existing standard glassware reactors for this process, extending equipment lifespans while avoiding costly upgrades. The ambient temperature operation further reduces wear on temperature control systems and associated maintenance costs, creating immediate operational savings that directly impact manufacturing cost structures without requiring new capital investment.
- Shorter Lead Times: The ten-minute reaction time combined with simplified workup procedures enables dramatic reductions in production cycle times compared to traditional multi-step syntheses requiring extended reaction periods and complex purification sequences. This rapid turnaround capability allows manufacturers to respond more agilely to fluctuating demand patterns while minimizing work-in-progress inventory costs. The elimination of metal scavenging steps removes a major bottleneck in traditional furan production, enabling faster batch release cycles that directly support just-in-time manufacturing strategies essential for modern pharmaceutical supply chains.
- Lower Waste Treatment Costs: The environmentally benign nature of this process generates minimal hazardous waste streams compared to conventional methods involving heavy metals and strong acids or bases. The aqueous quenching step produces only benign byproducts that can be processed through standard wastewater treatment systems without requiring specialized hazardous waste handling protocols. This significant reduction in waste treatment complexity not only lowers disposal costs but also mitigates regulatory compliance risks associated with heavy metal contamination, creating substantial operational savings while supporting corporate sustainability initiatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111592507A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
