Revolutionizing Furan Scaffold Production: A Metal-Free Catalytic Approach for Commercial Scale-Up
The landscape of organic synthesis for heterocyclic scaffolds is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective manufacturing processes. Patent CN111592507A introduces a groundbreaking methodology for the preparation of polysubstituted furans, a class of compounds that serves as a critical backbone in numerous pharmaceutical intermediates and agrochemical agents. This innovation addresses the longstanding challenges associated with furan synthesis, specifically the reliance on precious metal catalysts and harsh reaction conditions that have historically plagued the industry. By leveraging a cheap Lewis acid catalyst, specifically boron trifluoride etherate, this novel route enables the rapid construction of complex furan skeletons through a cycloaddition reaction between alkynamides and alpha-hydroxy ketones. The implications for industrial manufacturing are profound, as this method not only simplifies the operational workflow but also aligns perfectly with modern environmental compliance standards by eliminating heavy metal contamination risks. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and scalable synthetic strategies that do not compromise on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the furan skeleton, particularly for highly substituted variants like tetra-substituted furans, has been a formidable challenge in organic synthesis. Conventional methodologies often rely heavily on the use of transition metal catalysts such as gold, rhodium, or palladium, which are not only prohibitively expensive but also introduce significant supply chain vulnerabilities due to their scarcity and price volatility. Furthermore, these metal-catalyzed reactions frequently require harsh conditions, including strong acids, strong bases, or elevated temperatures, which can lead to poor functional group tolerance and the degradation of sensitive substrates. A critical drawback for pharmaceutical manufacturing is the issue of heavy metal residues; removing trace amounts of precious metals from the final active pharmaceutical ingredient (API) or intermediate requires extensive and costly purification steps, such as specialized scavenging resins or repeated recrystallizations. Additionally, the generation of hazardous waste (three wastes) from these aggressive reaction conditions poses a substantial environmental burden, complicating waste disposal and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN111592507A offers a streamlined, metal-free alternative that fundamentally reshapes the economics of furan production. This approach utilizes inexpensive and readily available boron trifluoride etherate as a Lewis acid catalyst to promote a tandem cycloaddition reaction between alkynamides and alpha-hydroxy ketones. The reaction proceeds with remarkable efficiency at room temperature, completing in less than 10 minutes, which is a dramatic improvement over the multi-hour timelines typical of traditional protocols. This mild condition ensures excellent compatibility with a wide range of functional groups, allowing for the selective synthesis of mono-, di-, tri-, and tetra-substituted furans simply by varying the substituents on the starting materials. The elimination of precious metals not only reduces raw material costs but also simplifies the downstream processing, as there is no need for rigorous heavy metal removal steps. This results in a cleaner crude product profile and facilitates easier isolation of the target compound, thereby enhancing the overall process mass intensity (PMI) and making it an ideal candidate for reliable polysubstituted furans supplier operations.
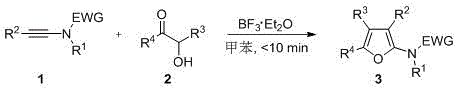
Mechanistic Insights into BF3-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the activation of the carbonyl and alkyne moieties by the boron trifluoride catalyst, which lowers the energy barrier for the cycloaddition cascade. The Lewis acid coordinates with the oxygen atoms of the alpha-hydroxy ketone and potentially the amide carbonyl, increasing the electrophilicity of the system and facilitating the nucleophilic attack by the alkyne. This initiates a rapid cyclization sequence that constructs the furan ring in a single operational step, bypassing the need for multiple intermediate isolations. The reaction pathway is highly concerted, which minimizes the formation of side products and ensures high atom economy. For process chemists, understanding this mechanism is crucial for optimizing reaction parameters; the use of toluene as a solvent provides an ideal medium for solubilizing both organic substrates while maintaining the stability of the Lewis acid catalyst. The rapid kinetics observed, with full conversion achieved in under 10 minutes, suggest a low activation energy profile that is highly favorable for scale-up, as it reduces the residence time required in continuous flow reactors or large batch vessels.
From an impurity control perspective, this metal-free mechanism offers distinct advantages over transition metal catalysis. The absence of gold or palladium species eliminates the risk of metal-ligand complexes forming stable byproducts that are difficult to separate. Furthermore, the mild acidic conditions prevent the polymerization or decomposition of the sensitive furan ring, which can occur under strong acidic or basic conditions used in older methods. The workup procedure described in the patent, involving a simple quench with saturated ammonium chloride and sodium bicarbonate, effectively neutralizes the catalyst and allows for clean phase separation. This simplicity in quenching and extraction means that the organic phase contains primarily the desired product and minimal inorganic salts, streamlining the drying and concentration steps. Consequently, the final product obtained after flash column chromatography exhibits high purity, meeting the stringent specifications required for high-purity pharmaceutical intermediates without the need for complex chiral separations or extensive washing protocols.
How to Synthesize Polysubstituted Furans Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant production. The protocol begins with the dissolution of the alkynamide and alpha-hydroxy ketone substrates in toluene at room temperature, ensuring a homogeneous mixture before the introduction of the catalyst. The addition of a catalytic amount of boron trifluoride etherate triggers the immediate onset of the reaction, which is monitored by TLC and typically reaches completion within a very short timeframe. Following the reaction, a straightforward aqueous workup involving saturated salt solutions allows for the efficient separation of the organic product from the catalyst and any water-soluble byproducts. The detailed standardized synthesis steps, including specific molar ratios and quenching procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve alkynamide and alpha-hydroxy ketone substrates in toluene solvent at room temperature to ensure complete homogeneity before catalysis.
- Add 5 mol% boron trifluoride etherate (BF3·Et2O) to the reaction mixture and stir vigorously for approximately 10 minutes to drive the cycloaddition to completion.
- Quench the reaction with saturated ammonium chloride and sodium bicarbonate solutions, followed by extraction with ethyl acetate and purification via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The substitution of expensive precious metal catalysts with a commodity chemical like boron trifluoride etherate results in a drastic reduction in raw material costs, which is particularly significant when producing large volumes of intermediates. Moreover, the simplified purification process reduces the consumption of solvents and stationary phases required for chromatography, further driving down the cost of goods sold (COGS). The robustness of the reaction conditions also enhances supply chain reliability, as the reagents are commercially available in bulk quantities and are not subject to the geopolitical supply constraints often associated with rare earth metals or precious catalysts. This stability ensures consistent production schedules and mitigates the risk of delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The elimination of gold, rhodium, or palladium catalysts removes a major cost driver from the bill of materials, while the ambient temperature operation significantly lowers energy consumption compared to heating-intensive processes. The high yield and selectivity of the reaction minimize waste generation, reducing the costs associated with waste treatment and disposal. Additionally, the simplified workup procedure reduces labor hours and equipment usage time, contributing to overall operational efficiency and substantial cost savings in polysubstituted furans manufacturing.
- Enhanced Supply Chain Reliability: By relying on widely available and inexpensive reagents, manufacturers can secure a more stable supply chain that is less vulnerable to market fluctuations. The short reaction time of less than 10 minutes allows for higher throughput in existing facilities, effectively increasing capacity without the need for capital investment in new reactors. This agility enables suppliers to respond more quickly to demand spikes from downstream pharmaceutical clients, ensuring timely delivery of critical intermediates and reducing lead time for high-purity polysubstituted furans.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with increasingly strict environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing. The absence of toxic metal residues simplifies the regulatory filing process for new drug applications, as there is no need to demonstrate extensive clearance of metal impurities. Furthermore, the mild conditions and high atom economy make this process inherently safer and more sustainable, facilitating easier scale-up from kilogram to tonnage production while maintaining a green chemistry profile that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this furan synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free furan synthesis over traditional methods?
A: Unlike conventional methods requiring expensive gold, rhodium, or palladium catalysts under harsh conditions, this process utilizes cheap boron trifluoride etherate at room temperature, eliminating heavy metal residues and significantly reducing purification costs.
Q: How does the reaction time impact production throughput?
A: The reaction completes in less than 10 minutes, which drastically increases reactor turnover rates compared to traditional multi-hour protocols, allowing for higher volume production within the same timeframe.
Q: Is this method suitable for generating tetra-substituted furan derivatives?
A: Yes, the method is specifically designed to access tetra-substituted furans by controlling the substituents on the alkynamide and alpha-hydroxy ketone starting materials, offering superior structural diversity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Furans Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the production of high-value furan intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative chemistry can be fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of polysubstituted furans meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering consistent quality and supply continuity, leveraging our technical expertise to optimize this green synthesis method for maximum efficiency and cost-effectiveness.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages this method offers for your specific supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient, metal-free technology into your production pipeline for reliable polysubstituted furans supplier partnerships.
