Scaling Green Paal-Knorr Furan Synthesis: A Commercial Breakthrough for Pharmaceutical Intermediates
Introduction to Patent CN101580498B: A Paradigm Shift in Furan Synthesis
The chemical industry is currently undergoing a rigorous transformation towards greener, more sustainable manufacturing processes, and patent CN101580498B stands at the forefront of this evolution by introducing a novel green synthesis method for polysubstituted furans. This intellectual property details a robust protocol where 1,4-dicarbonyl compounds undergo dehydration cyclization in an aqueous medium, catalyzed by a strong acidic cation exchange resin, effectively replacing hazardous organic solvents and corrosive liquid acids. For R&D directors and process chemists, this represents a critical advancement because it addresses the longstanding environmental and safety liabilities associated with the classical Paal-Knorr reaction, which traditionally relies on benzene, toluene, or chlorinated solvents. The patent demonstrates that by utilizing water as the sole solvent and a solid acid resin as a heterogeneous catalyst, manufacturers can achieve high yields while drastically simplifying the downstream processing workflow. Furthermore, the methodology supports a broad substrate scope, accommodating alkyl, aryl, and ester substituents, making it a versatile platform technology for the production of high-purity pharmaceutical intermediates and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furan derivatives via the Paal-Knorr reaction has been plagued by significant operational and environmental drawbacks that hinder efficient commercial scale-up of complex heterocyclic intermediates. Traditional protocols typically mandate the use of volatile and toxic organic solvents such as benzene or toluene to facilitate the dehydration process, creating severe health risks for operators and imposing heavy burdens on waste management systems. Moreover, the catalysts employed in these legacy methods, including p-toluenesulfonic acid, polyphosphoric acid, zinc chloride, or boron trifluoride etherates, are often homogeneous strong acids or Lewis acids that are difficult to separate from the final product matrix. This difficulty necessitates extensive neutralization, washing, and purification steps, which not only increase the consumption of raw materials and energy but also generate large volumes of acidic wastewater and salt waste. From a supply chain perspective, the reliance on these hazardous reagents complicates logistics and storage, while the potential for residual metal contamination in the final API intermediate poses a critical quality risk that requires expensive additional purification technologies to mitigate.
The Novel Approach
In stark contrast to these conventional limitations, the novel approach described in the patent leverages the unique properties of strong acidic cation exchange resins, specifically sulfonic acid types like Dowex 50W×8-200, to drive the cyclization in a purely aqueous environment. This shift to water as a solvent eliminates the fire hazards and toxicity associated with organic volatiles, aligning perfectly with modern green chemistry principles and regulatory expectations for sustainable manufacturing. The heterogeneous nature of the resin catalyst allows for a simple filtration step to separate the catalyst from the reaction mixture, enabling immediate recovery and regeneration of the catalyst for subsequent batches without significant loss of activity. This innovation fundamentally alters the economic model of furan production by reducing the cost reduction in pharmaceutical intermediate manufacturing through lower solvent procurement costs, minimized waste disposal fees, and streamlined isolation procedures. Additionally, the reaction conditions are flexible, operating effectively either under reflux at 100°C or in a sealed system at 130°C, providing process engineers with the levers to optimize reaction kinetics and throughput based on specific substrate reactivity profiles.
Mechanistic Insights into Resin-Catalyzed Paal-Knorr Cyclization
Understanding the mechanistic underpinnings of this resin-catalyzed transformation is essential for R&D teams aiming to optimize reaction parameters for new substrates. The strong acidic cation exchange resin functions as a solid Brønsted acid, donating protons to the carbonyl oxygen of the 1,4-dicarbonyl starting material to initiate the catalytic cycle. Following protonation, the carbonyl group tautomerizes to its enol form, which then acts as a nucleophile to attack the second protonated carbonyl carbon within the same molecule. This intramolecular nucleophilic attack is widely recognized as the rate-limiting step in the Paal-Knorr mechanism, and its efficiency is heavily influenced by the electronic nature of the substituents attached to the carbonyl carbons. As illustrated in the reaction mechanism below, the formation of the cyclic hemiketal intermediate is followed by a dehydration step that aromatizes the ring to yield the stable polysubstituted furan product.
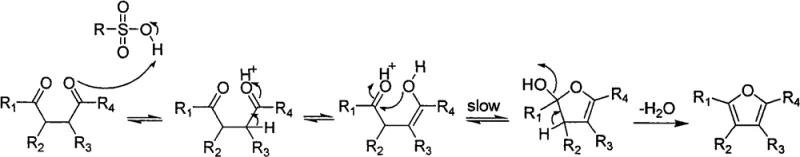
The patent data provides compelling evidence that electron-withdrawing groups, such as phenyl rings, enhance the electrophilicity of the carbonyl carbon, thereby accelerating the rate-limiting nucleophilic attack compared to electron-rich groups like thienyl. This electronic effect explains why 1,4-diphenylbutane-1,4-dione reacts significantly faster than its dithienyl counterpart under identical conditions. Furthermore, the mechanism accounts for the behavior of ester-substituted substrates, where competing pathways of direct cyclization versus decarboxylation can occur. When the cyclization rate outpaces decarboxylation, the non-decarboxylated furan is the major product; conversely, if decarboxylation is kinetically favored, the mono-decarboxylated derivative predominates. This mechanistic clarity allows chemists to predict impurity profiles and adjust temperature or residence time to steer selectivity towards the desired commercial grade high-purity polysubstituted furans.
How to Synthesize Polysubstituted Furans Efficiently
To implement this green synthesis route in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize catalyst efficiency and product recovery. The general protocol involves suspending the 1,4-dicarbonyl substrate in water with a defined loading of the resin catalyst, typically maintaining a molar ratio of substrate to resin functional groups between 1:10 and 1:12 to ensure sufficient acidic sites are available for turnover. The reaction mixture is then heated, with the choice between open reflux at 100°C or sealed heating at 130°C depending on the required reaction speed and substrate stability. Detailed standardized synthesis steps for specific substrates are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Mix 1,4-dicarbonyl compound with water and Dowex 50W×8-200 resin (molar ratio 1: 10-12).
- Heat the mixture to 100°C for reflux or 130°C in a sealed vessel for 6-9 hours.
- Filter to recover the resin catalyst, extract the filtrate with ether, wash, dry, and purify to obtain the furan product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resin-catalyzed aqueous process offers transformative benefits that extend far beyond simple yield improvements, fundamentally reshaping the cost structure and reliability of the supply base. By transitioning away from volatile organic solvents and corrosive liquid acids, facilities can significantly reduce their exposure to fluctuating petrochemical prices and stringent environmental compliance costs associated with hazardous waste treatment. The ability to recycle the solid acid catalyst multiple times without substantial degradation in performance creates a closed-loop system that minimizes raw material consumption and lowers the total cost of ownership for the manufacturing process. Moreover, the simplified workup procedure, which relies on basic filtration and extraction rather than complex distillation or chromatography, shortens the overall production cycle time and increases the throughput capacity of existing reactor infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic organic solvents like benzene and toluene directly reduces raw material expenditures, while the use of water as a benign alternative removes the need for specialized solvent recovery units. Since the catalyst is heterogeneous and easily separated by filtration, the downstream processing costs are drastically simplified, avoiding the expensive neutralization and salt disposal steps required by homogeneous acid catalysts. This streamlined workflow translates into substantial operational savings and a more predictable cost base for long-term supply contracts.
- Enhanced Supply Chain Reliability: Utilizing a robust solid catalyst that can be regenerated and reused enhances supply continuity by reducing dependency on single-use reagents that may face market shortages. The mild reaction conditions and aqueous medium improve process safety, lowering the risk of unplanned shutdowns due to safety incidents or regulatory inspections. Furthermore, the high conversion rates observed for a wide range of substrates ensure consistent output quality, allowing supply chain planners to maintain leaner inventory levels with confidence in the reliability of the production schedule.
- Scalability and Environmental Compliance: The process is inherently scalable because heat transfer and mixing in aqueous slurries are well-understood engineering challenges that do not require exotic equipment. The absence of halogenated solvents and heavy metal catalysts simplifies the environmental permitting process and ensures that the final product meets strict residual solvent and heavy metal specifications required by global regulatory bodies. This alignment with green chemistry standards future-proofs the manufacturing asset against tightening environmental regulations and enhances the brand reputation of the end-product in eco-conscious markets.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology, derived directly from the experimental data and beneficial effects outlined in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this resin-catalyzed method for the production of furan derivatives. Understanding these details is crucial for stakeholders evaluating the transition from legacy chemical processes to this sustainable alternative.
Q: What are the advantages of using cation exchange resin over traditional acids like p-TsOH?
A: Using strong acidic cation exchange resin allows for heterogeneous catalysis in water, eliminating toxic organic solvents like benzene. The resin is easily filtered and regenerated, significantly reducing waste disposal costs and simplifying the purification process compared to homogeneous acid catalysts.
Q: Can this method handle ester-substituted 1,4-dicarbonyl compounds?
A: Yes, the method is effective for tetra-substituted compounds with ester groups. However, reaction conditions influence the product distribution between decarboxylated and non-decarboxylated furans, allowing for tunable selectivity based on temperature and reaction time.
Q: Is the catalyst reusable for industrial scale production?
A: Experimental data confirms the resin can be reused multiple times with only a slight decrease in catalytic efficiency after three cycles. This reusability is a key factor for cost reduction in large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Furans Supplier
As the global demand for sustainable and high-quality chemical building blocks continues to rise, NINGBO INNO PHARMCHEM stands ready to leverage this patented technology to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of polysubstituted furans meets the exacting standards required for pharmaceutical and fine chemical applications. By integrating this green resin-catalyzed process into our manufacturing portfolio, we can offer clients a supply solution that is not only cost-effective but also environmentally responsible and fully compliant with international regulations.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this aqueous protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market.
