Advanced Synthetic Route for Aztreonam: Enhancing Purity and Commercial Scalability for Global Pharma Markets
Introduction to Advanced Aztreonam Manufacturing Technologies
The global demand for effective monobactam antibiotics continues to drive innovation in the synthesis of aztreonam, a critical therapeutic agent for treating severe Gram-negative bacterial infections. Patent CN101514200B introduces a transformative synthetic methodology that addresses longstanding inefficiencies in the production of this vital API intermediate. Unlike traditional routes that rely on hazardous reagents and complex purification protocols, this novel approach leverages a sophisticated active ester coupling strategy followed by a mild, aqueous-based deprotection sequence. By shifting away from toxic condensing agents and aggressive acids, the technology offers a pathway to higher purity profiles and reduced environmental impact, positioning it as a cornerstone for modern reliable API intermediate supplier networks seeking sustainable manufacturing solutions.
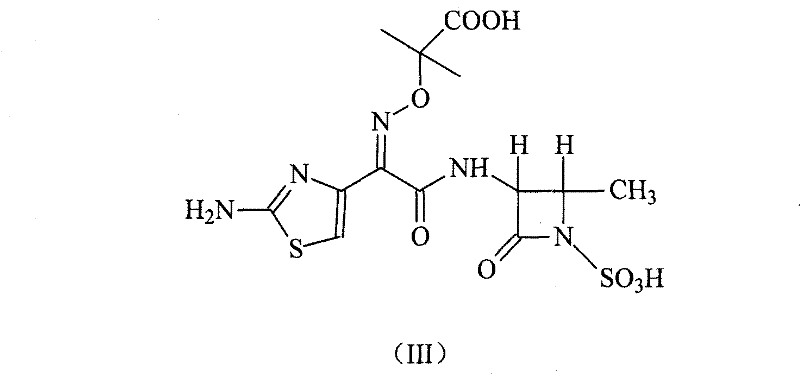
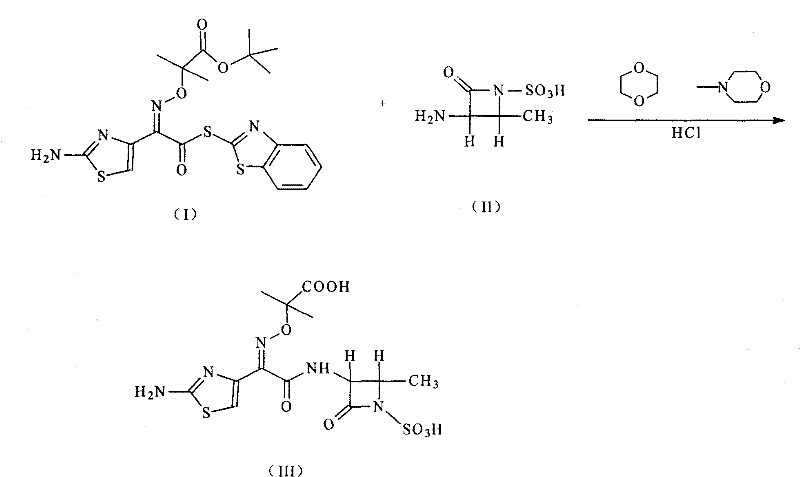
This patent specifically targets the optimization of the final coupling and deprotection stages, which are historically the most yield-limiting steps in aztreonam production. The described method utilizes (2-aminothiazole-4-yl)-2-(tert-butoxycarbonyl)-iminoacetic acid isopropyl ester-2-mercaptobenzothiazole ester as a highly reactive electrophile. When coupled with (3S-trans)-3-amino-4-methyl-2-oxo-1-azetidinyl sulfonic acid in a 1,4-dioxane system, the reaction proceeds with exceptional selectivity. The subsequent removal of protecting groups using a mixed aqueous solution of acetic acid and hydrochloric acid represents a significant departure from the industry-standard use of trifluoroacetic acid, thereby eliminating the need for expensive solvent recovery systems and mitigating safety risks associated with handling strong fluorinated acids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aztreonam has been plagued by significant operational and economic hurdles, primarily stemming from the reliance on carbodiimide-mediated coupling and harsh deprotection conditions. Prior art, such as US Patent 4775670, describes methods utilizing dicyclohexylcarbodiimide (DCC) as a condensing agent. While chemically effective, DCC generates dicyclohexylurea (DCU) as a stoichiometric byproduct, which is notoriously difficult to remove completely from the reaction mixture, often requiring extensive filtration and washing steps that reduce overall throughput. Furthermore, the deprotection of the tert-butyl ester in these legacy processes typically necessitates the use of trifluoroacetic acid (TFA) under anhydrous conditions. TFA is not only prohibitively expensive but also highly corrosive and toxic, imposing severe burdens on equipment maintenance and waste disposal infrastructure. Alternative methods employing sulfuric acid have been reported, yet they frequently suffer from low yields due to the formation of open-ring byproducts, compromising the integrity of the sensitive beta-lactam ring and necessitating complex chromatographic purifications that are impractical at scale.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN101514200B streamlines the entire synthetic sequence through the strategic application of activated ester chemistry. By pre-activating the thiazole side chain as a mercaptobenzothiazole ester, the process bypasses the need for in-situ activation with carbodiimides, effectively eliminating the generation of insoluble urea waste. The reaction is conducted in 1,4-dioxane, a solvent chosen for its ability to solubilize both the polar beta-lactam substrate and the organic side chain, creating a homogeneous reaction environment that enhances kinetics. Following the coupling, the deprotection step is ingeniously managed using a mixed aqueous system of acetic acid and hydrochloric acid. This mild acidic environment is sufficiently robust to cleave the tert-butyl group while being gentle enough to preserve the stereochemical integrity of the Z-oxime and the beta-lactam ring. This shift not only simplifies the workup procedure—allowing for direct crystallization from the aqueous phase—but also drastically reduces the consumption of hazardous reagents, aligning perfectly with the goals of cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Active Ester Coupling and Mild Deprotection
The core of this synthetic advancement lies in the mechanistic efficiency of the nucleophilic acyl substitution between the activated ester and the beta-lactam amine. The mercaptobenzothiazole leaving group is exceptionally stable yet highly labile towards nucleophilic attack by the primary amine of the azetidinone ring. In the presence of N-methylmorpholine, which acts as a non-nucleophilic base, the amine is transiently deprotonated to enhance its nucleophilicity without risking ring opening. The reaction is initiated at low temperatures (0°C to 10°C) to kinetically control the addition and prevent any potential epimerization at the chiral centers. This precise temperature control is critical for maintaining the (3S, trans) configuration of the beta-lactam ring, which is essential for the biological activity of the final antibiotic. The use of 1,4-dioxane ensures that the transition state is well-solvated, facilitating a smooth conversion to the protected intermediate with minimal formation of diastereomeric impurities.
Following the coupling, the deprotection mechanism proceeds via an acid-catalyzed cleavage of the tert-butyl ester. Unlike the aggressive protonation caused by neat TFA, the aqueous acetic acid/hydrochloric acid mixture provides a buffered acidic environment. The protonation of the tert-butyl ester oxygen leads to the formation of a tert-butyl cation, which is rapidly scavenged by water or chloride ions to form volatile isobutylene or tert-butyl chloride, driving the equilibrium towards the free carboxylic acid. Crucially, the presence of water in this system helps to stabilize the zwitterionic nature of the aztreonam molecule, preventing the acid-catalyzed hydrolysis of the beta-lactam amide bond—a common failure mode in anhydrous acidic conditions. This mechanistic nuance allows for the direct isolation of the product as a solid upon pH adjustment and cooling, achieving high purity levels (>99%) without the need for column chromatography, thus demonstrating superior commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Aztreonam Efficiently
The operational protocol derived from this patent offers a robust framework for laboratory and pilot-scale production, emphasizing simplicity and safety. The process begins with the dissolution of the beta-lactam amine salt in 1,4-dioxane, followed by neutralization with N-methylmorpholine to generate the free amine in situ. Careful temperature management is maintained throughout the addition of the active ester solution to ensure exothermic control. After the coupling is complete, the reaction mixture is treated with ethyl acetate and an aqueous acetic acid buffer to facilitate phase separation, allowing for the removal of organic-soluble impurities. The aqueous phase, containing the protected intermediate, is then subjected to mild heating (30°C to 50°C) in the presence of dilute hydrochloric acid to effect deprotection. Finally, the product is induced to crystallize by cooling and pH adjustment, yielding the target compound as a white beta-type crystal. For detailed standardized operating procedures and safety guidelines, please refer to the technical documentation below.
- Dissolve (3S-trans)-3-amino-4-methyl-2-oxo-1-azetidinyl sulfonic acid in 1,4-dioxane with N-methylmorpholine and cool to 0-10°C.
- Add the active ester intermediate (thiazolamine-4-yl derivative) dissolved in 1,4-dioxane and stir for coupling.
- Adjust pH using acetic acid and hydrochloric acid mixtures, warm to 40°C for deprotection, and crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility inherent in antibiotic manufacturing. By eliminating the dependency on specialized and expensive reagents like DCC and TFA, manufacturers can significantly stabilize their raw material costs and reduce exposure to supply disruptions associated with fluorinated chemicals. The simplified workup procedure, which relies on standard liquid-liquid extraction and crystallization rather than complex filtration of urea byproducts or chromatographic purification, translates directly into reduced processing time and lower utility consumption. Furthermore, the ability to recover and reuse solvents like ethyl acetate and 1,4-dioxane enhances the overall atom economy of the process, contributing to substantial long-term operational savings and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of dicyclohexylcarbodiimide (DCC) removes the significant cost burden associated with purchasing this high-value coupling agent and the downstream costs of disposing of its urea byproduct. Additionally, replacing trifluoroacetic acid (TFA) with a mixture of acetic and hydrochloric acid results in drastic raw material cost savings, as mineral acids are commoditized and inexpensive compared to fluorinated organic acids. The process also avoids the need for specialized corrosion-resistant equipment required for handling anhydrous TFA, thereby lowering capital expenditure (CAPEX) for production facilities.
- Enhanced Supply Chain Reliability: The reliance on conventional solvents such as 1,4-dioxane and ethyl acetate ensures a stable and diversified supply base, as these chemicals are produced globally in massive volumes for various industries. Unlike proprietary catalysts or niche reagents that may face allocation issues, the inputs for this process are readily available from multiple vendors, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further securing the continuity of supply for critical antibiotic intermediates.
- Scalability and Environmental Compliance: The aqueous workup and crystallization steps are inherently scalable, allowing for seamless transition from kilogram to multi-ton production scales without the need for process re-engineering. The reduction in hazardous waste generation, particularly the avoidance of fluorinated waste streams and solid urea sludge, simplifies compliance with increasingly stringent environmental regulations. This 'green' profile not only reduces waste disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity, making it a more attractive partner for global pharmaceutical companies focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aztreonam synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on yield expectations, purity profiles, and operational feasibility. Understanding these nuances is essential for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: What are the primary advantages of this aztreonam synthesis method over conventional DCC-based routes?
A: This method eliminates the use of toxic and expensive condensing agents like DCC and hazardous deprotection reagents like trifluoroacetic acid (TFA). It utilizes conventional solvents like 1,4-dioxane and mild acidic hydrolysis, significantly simplifying waste treatment and reducing raw material costs.
Q: How does the new process ensure high stereochemical purity of the Z-isomer?
A: The process employs a pre-formed active ester intermediate which reacts under mild conditions (0-10°C initially) with the beta-lactam amine. This controlled environment prevents isomerization and ring-opening side reactions common in harsher acidic conditions, ensuring the critical Z-configuration is maintained.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is highly scalable. It avoids difficult filtration steps associated with urea byproducts (from DCC) and uses solvents that are easily recoverable. The workup involves simple phase separation and pH-adjusted crystallization, which are standard unit operations in multi-ton manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for life-saving antibiotics like aztreonam. Our technical team has extensively analyzed the innovations presented in CN101514200B and possesses the expertise to implement this advanced active ester coupling strategy at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for downstream API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to leverage these process improvements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can drive value and reliability for your aztreonam projects.
