Advanced Three-Stage Purification Technology for High-Purity Aztreonam Manufacturing
The pharmaceutical industry continuously demands higher purity standards for critical antibiotic intermediates, particularly for beta-lactam compounds like aztreonam which are sensitive to hydrolysis and impurity formation. Patent CN102086196B introduces a groundbreaking refinement methodology that addresses the longstanding challenges of residual solvents, heavy metal contamination, and stereochemical instability in aztreonam production. This novel approach utilizes a sophisticated three-stage purification sequence involving alkali metal alkoxide treatment, macroporous strong basic ion exchange chromatography, and controlled organic acid crystallization. By fundamentally altering the purification landscape, this technology enables manufacturers to achieve purity levels exceeding 99.5%, significantly surpassing the capabilities of conventional recrystallization or weak resin adsorption techniques. The strategic integration of specific chemical mechanisms ensures that not only is the target molecule isolated, but trace ester impurities are chemically converted back into the active pharmaceutical ingredient, enhancing both quality and yield.
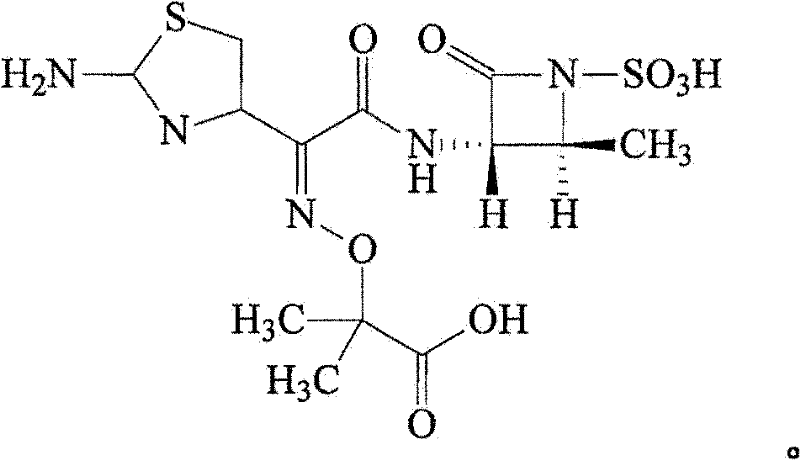
For R&D directors and process chemists, understanding the structural integrity of aztreonam is paramount, as depicted in the molecular diagram above, which highlights the sensitive beta-lactam ring and the sulfonic acid moiety that dictate its solubility and reactivity profiles. The implementation of this patented process represents a significant leap forward in process chemistry, offering a robust solution for producing high-purity aztreonam suitable for parenteral formulations where endotoxin and particulate limits are stringent. As a reliable pharmaceutical intermediates supplier, recognizing the value of such advanced purification protocols is essential for maintaining competitive advantage in the global antibiotic market. The following analysis details how this method overcomes historical bottlenecks in aztreonam manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of aztreonam has been plagued by reliance on harsh reagents and inefficient separation media that compromise both product quality and operational safety. Traditional methods, such as those disclosed in earlier patents like US4775670A, often depend on trifluoroacetic acid (TFA) for deprotection steps, which introduces severe toxicity concerns and necessitates complex, energy-intensive recovery systems to prevent environmental contamination. Furthermore, conventional recrystallization techniques using mineral acids like hydrochloric or sulfuric acid pose a significant risk of partial hydrolysis of the amide linkage and damage to the thiazole ring, leading to the formation of open-loop degradation products that are difficult to remove. Another prevalent issue involves the use of non-ionic macroporous adsorption resins like HP-20, which demonstrate poor purification efficiency for highly polar compounds like aztreonam due to insufficient interaction with the zwitterionic nature of the molecule. These legacy processes frequently result in final products with purity levels struggling to reach 95%, accompanied by unacceptable levels of residual heavy metals and bacterial endotoxins that fail to meet modern pharmacopoeial standards for injectable antibiotics.
The Novel Approach
In stark contrast, the methodology described in CN102086196B employs a gentle yet highly effective chemical strategy that preserves the integrity of the beta-lactam scaffold while aggressively removing impurities. The core innovation lies in the initial treatment with alkali metal or alkaline-earth metal alkoxides, which serves a dual purpose of dissolving the crude material and chemically hydrolyzing trace ester impurities back into the desired aztreonam structure, thereby boosting yield rather than losing material. Following this, the process utilizes macroporous strong basic anion exchange resins, such as Amberlite IRA-900 or D201, which possess a unique dual-phase structure capable of adsorbing not only ionic impurities but also non-electrolyte contaminants, colored bodies, and heavy metals through physical trapping in their macro-reticular channels. The final crystallization step utilizes mild organic acids like benzenesulfonic acid or p-toluenesulfonic acid to adjust the pH, avoiding the corrosive and degradative effects of inorganic acids while promoting the formation of stable, high-purity crystals. This holistic approach ensures that the final product achieves a purity of not less than 99.2%, with residue on ignition and heavy metal content reduced to negligible levels, making it ideal for high-specification pharmaceutical applications.
Mechanistic Insights into Alkali Hydrolysis and Ion Exchange Purification
The first stage of this purification protocol leverages the nucleophilic properties of alkali metal alkoxides, such as sodium ethylate or potassium methylate, to effect a selective transesterification or hydrolysis of impurities. In many synthetic routes for aztreonam, the final steps involve the removal of carboxyl protecting groups, which inevitably leaves behind trace amounts of unreacted esters or side-product esters in the crude bullion. When treated with alkoxides in alcoholic solvents at elevated temperatures ranging from 40°C to 80°C, these ester impurities undergo hydrolysis, cleaving the ester bond to regenerate the free carboxylic acid form of aztreonam. This chemical conversion is critical because it transforms potential impurities into the target product, effectively increasing the overall mass balance and yield of the process. Subsequent adjustment of the pH to the acidic range of 1.0 to 3.0 using suitable acids causes the aztreonam to precipitate out of the solution, leaving behind soluble non-polar impurities and salts in the mother liquor, thus achieving the first level of significant purification.
The second and perhaps most critical mechanistic step involves the interaction between the zwitterionic aztreonam molecule and the quaternary ammonium groups present on the strong basic ion exchange resin. Unlike weakly basic or neutral resins, strong basic resins maintain their ionization capacity across a wide pH range, allowing them to effectively capture anionic impurities and heavy metal complexes that coexist with the aztreonam. The macroporous nature of resins like D201 or IRA-900 provides a high surface area and large pore volume, which facilitates the diffusion of large molecular weight contaminants, such as bacterial endotoxins and polymeric byproducts, into the resin matrix where they are physically adsorbed and retained. During the elution phase, typically using an aqueous or alcoholic solution of alkali metal hydroxide, the aztreonam is selectively desorbed while the tightly bound impurities remain on the column. This chromatographic separation is superior to simple precipitation because it specifically targets the removal of toxicological hazards like heavy metals and pyrogens, which are critical quality attributes for parenteral drugs, ensuring the final eluate is chemically clean before the final crystallization step.
How to Synthesize High-Purity Aztreonam Efficiently
Implementing this three-stage purification process requires precise control over reaction parameters, particularly temperature and pH, to maximize the efficiency of each separation unit operation. The procedure begins with the dissolution of crude aztreonam in a lower alcohol solvent, followed by the addition of an alkoxide solution under heating to facilitate the hydrolysis of ester impurities, after which the pH is carefully lowered to induce precipitation of the primarily purified solid. The detailed standardized synthesis steps, including specific solvent ratios, residence times, and elution gradients for the ion exchange column, are outlined in the structured guide below to ensure reproducibility and compliance with GMP standards.
- Treat raw aztreonam with alkali metal alkoxide in solvent at 40-80°C, then adjust pH to precipitate primarily purified product.
- Adsorb the solution onto macroporous strong basic ion exchange resin, elute with alkali metal hydroxide, and concentrate.
- Adjust pH with organic acid to 1.0-4.0, crystallize the product, centrifuge, wash, and dry to obtain tertiary purified aztreonam.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this advanced refining technology offers substantial strategic benefits by mitigating risks associated with raw material variability and regulatory compliance. The elimination of hazardous reagents like trifluoroacetic acid simplifies waste management protocols and reduces the dependency on specialized recovery infrastructure, leading to a more streamlined and cost-effective manufacturing operation. Furthermore, the use of commercially available strong basic ion exchange resins ensures a stable supply of consumables, preventing bottlenecks that might arise from sourcing proprietary or exotic purification media. The ability to consistently produce ultra-high purity material reduces the likelihood of batch rejection during quality control testing, thereby enhancing overall supply chain reliability and ensuring continuous availability of this critical antibiotic intermediate for downstream formulation partners.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and toxic trifluoroacetic acid, replacing it with readily available alkali alkoxides and organic acids that are significantly cheaper and easier to handle on an industrial scale. By converting ester impurities back into the target product through alkali treatment, the overall yield of the process is improved, which directly lowers the cost of goods sold by maximizing the output from each batch of crude starting material. Additionally, the reduction in heavy metal content and residue on ignition minimizes the need for secondary polishing steps or extensive reprocessing, further driving down operational expenditures and improving the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: The reliance on standard chemical reagents and widely manufactured ion exchange resins, such as the Amberlite or D201 series, ensures that the supply chain is resilient against disruptions caused by the scarcity of specialized catalysts or reagents. The robustness of the purification method allows for the processing of crude aztreonam from various synthetic sources, providing flexibility in sourcing raw materials without compromising the quality of the final refined product. This adaptability is crucial for maintaining consistent delivery schedules to global pharmaceutical clients, as it reduces the risk of production delays associated with raw material specification variances or supplier quality issues.
- Scalability and Environmental Compliance: The unit operations involved in this process, including precipitation, filtration, and column chromatography, are well-established technologies that scale linearly from pilot plant to commercial manufacturing volumes without requiring complex engineering modifications. The substitution of mineral acids with organic acids in the final crystallization step reduces the generation of corrosive wastewater and saline waste, aligning the manufacturing process with increasingly stringent environmental regulations and sustainability goals. This eco-friendly profile not only reduces disposal costs but also enhances the corporate social responsibility standing of the manufacturer, making the supply of aztreonam more attractive to environmentally conscious multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel aztreonam purification technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and relevance for industry stakeholders.
Q: Why is strong basic ion exchange resin preferred over HP-20 for aztreonam purification?
A: Strong basic ion exchange resins possess macroporous structures that effectively adsorb non-electrolyte impurities, bacterial endotoxins, and heavy metals, which HP-20 resins fail to remove efficiently from high-polarity aztreonam solutions.
Q: How does the alkali metal alkoxide treatment improve yield?
A: The alkoxide treatment hydrolyzes trace ester impurities commonly found in crude aztreonam bullion back into the target aztreonam molecule, thereby simultaneously reducing impurity load and increasing overall recovery yield.
Q: What purity levels can be achieved with this refining method?
A: This three-stage process consistently produces aztreonam with a purity of not less than 99.2%, with most batches exceeding 99.5%, while maintaining extremely low residue on ignition and heavy metal content.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Supplier
The technological advancements detailed in patent CN102086196B underscore the immense potential for producing ultra-high purity aztreonam that meets the rigorous demands of modern parenteral therapy. At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such sophisticated purification protocols to life. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of aztreonam we supply is free from critical impurities and ready for formulation. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance guarantees that our clients receive material that performs reliably in their final drug products.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our advanced manufacturing capabilities can optimize your supply chain. Contact us today to discuss how we can support your aztreonam needs with a combination of cutting-edge technology and unwavering supply reliability.
