Advanced Metal-Free Synthesis of Benzoxazole Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing privileged heterocyclic scaffolds. Patent CN113264895B introduces a groundbreaking synthetic method for preparing benzoxazole compounds directly from catechol compounds and amine compounds. This technology represents a significant paradigm shift away from traditional transition metal-catalyzed processes, offering a metal-free alternative that utilizes 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) as a mild oxidant. Benzoxazole derivatives are critical structural motifs found in numerous bioactive molecules, exhibiting potent antibacterial, anti-inflammatory, and anticancer properties. The ability to synthesize these compounds without heavy metal contamination is of paramount importance for regulatory compliance in drug development. This report analyzes the technical merits and commercial implications of this novel route, highlighting its potential to serve as a reliable benzoxazole supplier strategy for global pharmaceutical manufacturers seeking to optimize their supply chains and reduce production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzoxazole core has relied heavily on the condensation of o-aminophenol with carboxylic acids or their derivatives, often necessitating harsh reaction conditions. The preparation of the o-aminophenol substrate itself is a major bottleneck, typically involving the nitration of phenol followed by reduction, a process that utilizes strong corrosive mixed acids and poses significant safety risks due to the exothermic nature of nitration reactions. Furthermore, many modern synthetic strategies for benzoxazole formation employ transition metal catalysts such as copper, palladium, or iron to facilitate C-H activation or oxidative cyclization. While effective, these metal-catalyzed methods introduce the persistent risk of heavy metal residues in the final active pharmaceutical ingredient (API), requiring expensive and time-consuming purification steps to meet stringent ICH Q3D guidelines. The reliance on precious metal ligands also drives up the raw material costs, making the overall process less economically viable for large-scale manufacturing of commodity intermediates.
The Novel Approach
In stark contrast, the method disclosed in patent CN113264895B utilizes readily available catechol compounds and amine compounds as starting materials, bypassing the hazardous nitration steps entirely. This metal-free oxidative cyclization proceeds under mild conditions, typically between 25°C and 70°C, using DDQ as the sole oxidant in common organic solvents like ethyl acetate or 1,2-dichloroethane. The reaction system is remarkably simple, avoiding the need for complex ligand systems or inert atmosphere rigor beyond standard nitrogen protection. This approach not only enhances the safety profile of the manufacturing process by eliminating explosive nitration hazards but also simplifies the downstream processing. The absence of transition metals means that the costly metal scavenging steps are rendered unnecessary, directly contributing to cost reduction in pharmaceutical intermediates manufacturing. The broad substrate tolerance allows for the synthesis of diverse 2-aryl, 2-alkenyl, and 2-alkyl substituted benzoxazoles, providing a versatile platform for drug discovery and process development teams.
Mechanistic Insights into DDQ-Mediated Oxidative Cyclization
The core of this innovation lies in the efficient oxidative cyclization mechanism facilitated by DDQ. The reaction initiates with the oxidation of the catechol compound to the corresponding o-quinone intermediate by DDQ. This highly electrophilic o-quinone then undergoes a condensation reaction with the nucleophilic amine compound to form an imine or Schiff base intermediate. Subsequent intramolecular cyclization occurs, where the phenolic hydroxyl group attacks the imine carbon, closing the oxazole ring. Finally, a second oxidation step by DDQ restores aromaticity to the benzoxazole system, releasing water as the only byproduct alongside the reduced form of DDQ. This mechanism is highly atom-economical and avoids the generation of stoichiometric metal waste. The use of DDQ ensures a clean reaction profile with high conversion rates, as evidenced by the experimental yields ranging from 60% to 97% across various substrates. The mild oxidative power of DDQ is sufficient to drive the reaction forward without degrading sensitive functional groups such as halogens, esters, or trifluoromethyl groups, which are often present in advanced drug candidates.
From a quality control perspective, this metal-free mechanism offers superior impurity control. Traditional metal-catalyzed routes often generate metal-organic complexes or side products resulting from metal-mediated homocoupling, which are difficult to separate and can act as genotoxic impurities. By eliminating the metal catalyst, the impurity profile of the resulting high-purity benzoxazole derivatives is significantly simplified. The primary impurities are typically unreacted starting materials or over-oxidized byproducts, which are easily removed via standard aqueous workup and column chromatography. This cleanliness is crucial for commercial scale-up of complex heterocyclic compounds, as it reduces the burden on analytical laboratories and ensures consistent batch-to-batch quality. The reaction's tolerance to various substituents on both the catechol and amine rings further demonstrates its robustness, allowing for the synthesis of structurally diverse libraries without the need for extensive process re-optimization for each new analog.
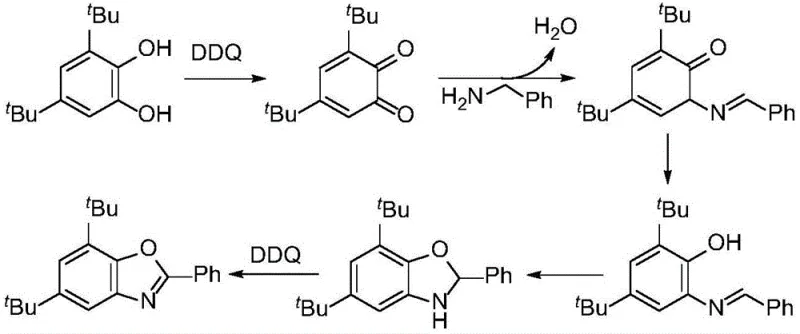
How to Synthesize 5,7-di-tert-butyl-2-phenylbenzo[d]oxazole Efficiently
To implement this synthesis in a laboratory or pilot plant setting, the procedure involves mixing the specific catechol derivative, such as 3,5-di-tert-butylcatechol, with the corresponding amine, like benzylamine, and DDQ in a molar ratio of approximately 1:1.2:2. The reaction is conducted in ethyl acetate under a nitrogen atmosphere at room temperature or with mild heating. Detailed standardized synthesis steps see the guide below.
- Mix catechol compound, amine compound, and DDQ oxidant in a reaction vessel under inert gas protection.
- Add solvent such as ethyl acetate or 1,2-dichloroethane and heat the mixture to 25-70°C for 3-12 hours with stirring.
- Cool the reaction, wash with saturated NaCl, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply chain. Catechol compounds and simple amines are commodity chemicals available from multiple global suppliers, reducing the risk of supply disruption compared to specialized metal catalysts or ligands which may have limited sources. This diversification of supply sources enhances supply chain reliability and provides leverage in price negotiations. Furthermore, the elimination of expensive transition metals like palladium or copper directly lowers the bill of materials cost. The simplified workup procedure, which avoids metal scavenging resins and complex filtration steps, reduces the consumption of auxiliary materials and shortens the overall production cycle time. These factors collectively contribute to substantial cost savings and improved margin potential for high-volume production campaigns.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and ligands from the process equation eliminates a significant cost center associated with precious metal procurement and recovery. Additionally, the avoidance of metal scavenging agents, which are often expensive disposable materials, further reduces operational expenditures. The use of common solvents like ethyl acetate, which are cheaper and easier to recycle than specialized polar aprotic solvents, adds to the economic efficiency. By streamlining the purification process and reducing the number of unit operations required to meet purity specifications, the overall manufacturing cost per kilogram is significantly optimized without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks such as catechols and amines ensures a stable and continuous supply of raw materials, as these are produced on a massive scale for various industries. This reduces the vulnerability to supply shocks that often affect specialized catalytic systems. The robustness of the reaction conditions, which tolerate mild temperature variations and do not require stringent anhydrous conditions beyond standard inert gas protection, minimizes the risk of batch failures due to operational deviations. This reliability is critical for maintaining consistent delivery schedules to downstream pharmaceutical clients, thereby strengthening long-term partnership agreements and reducing lead time for high-purity benzoxazoles.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The absence of heavy metal waste simplifies wastewater treatment and disposal, reducing the environmental footprint and associated compliance costs. The reaction's exotherm is manageable, and the use of DDQ, while an oxidant, is handled safely in standard organic synthesis setups. This makes the process highly scalable from gram to ton scale without the need for specialized reactor linings or containment systems required for corrosive acid-based traditional routes. The ease of scale-up ensures that commercial production can meet market demand fluctuations efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzoxazole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN113264895B, ensuring accuracy and relevance for process development teams evaluating this route for potential licensing or contract manufacturing opportunities.
Q: What are the primary advantages of this metal-free benzoxazole synthesis method?
A: This method eliminates the need for expensive transition metal catalysts and ligands, significantly reducing raw material costs and avoiding heavy metal residue contamination in the final pharmaceutical product.
Q: How does this process improve safety compared to traditional o-aminophenol routes?
A: By using catechol compounds instead of o-aminophenol, the process avoids the hazardous nitration reactions and strong corrosive mixed acids typically required, thereby reducing explosion risks and improving operational safety.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (25-70°C), use common solvents like ethyl acetate, and involve simple workup procedures, making it highly scalable for industrial manufacturing of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-value benzoxazole intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzoxazole derivative meets the exacting standards required by global regulatory bodies. We understand the critical nature of supply continuity in the pharmaceutical sector and have established robust procurement channels for key raw materials like DDQ and substituted catechols to prevent production bottlenecks.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to obtain specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable benzoxazole supplier capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
