Advanced Multi-Step Synthesis of Benzoxazepine Derivatives for Scalable Drug Manufacturing
Introduction to Advanced Benzoxazepine Synthesis
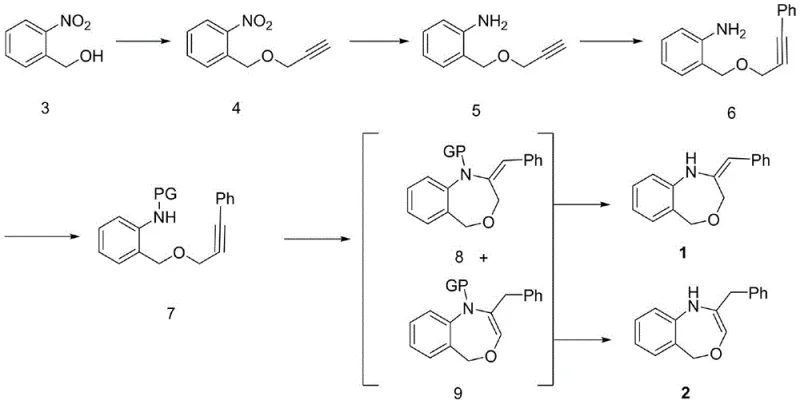
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly those exhibiting potent biological activities. Patent CN108997251B discloses a highly efficient, multi-step methodology for synthesizing (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine and its dihydro analogues. These 1,4-benzoxazepine derivatives represent a critical class of intermediates in modern drug discovery, known for their diverse pharmacological profiles including anticancer, antitubercular, and neuroprotective properties. The disclosed technology addresses significant limitations in existing literature by providing a route that ensures high stereochemical control and operational simplicity. By leveraging a strategic combination of etherification, transition metal-catalyzed coupling, and copper-mediated cyclization, this process enables the reliable production of high-purity pharmaceutical intermediates essential for developing next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dihydro-1,4-benzoxazepines and related structures has been plagued by challenges regarding molecular diversity and stereoselectivity. Traditional approaches often suffer from low yields during the critical ring-closing steps or require harsh reaction conditions that compromise the integrity of sensitive functional groups. Furthermore, many reported methods lack the flexibility to easily access both exocyclic and endocyclic alkene isomers, which are often required for structure-activity relationship (SAR) studies. The inability to efficiently separate these isomers or control their formation ratio has frequently bottlenecked the development of potential antitubercular and antihypertensive drugs based on this scaffold. Consequently, there has been a persistent demand for a synthetic strategy that can overcome these structural limitations while maintaining cost-effectiveness and scalability for industrial applications.
The Novel Approach
The innovative route detailed in the patent data introduces a streamlined five-to-six-step sequence starting from readily available o-nitrobenzyl alcohol. A key breakthrough lies in the first step, where etherification is optimized using either methyl Grignard reagents or Mitsunobu conditions, achieving isolation yields exceeding 90 percent, a significant improvement over standard nucleophilic substitutions. This is followed by a highly selective reduction and a palladium-catalyzed Sonogashira coupling to install the phenylalkyne motif. The culmination of the synthesis involves a sophisticated copper-catalyzed cyclization that simultaneously generates the seven-membered oxazepine ring with excellent regiocontrol. This approach not only simplifies the operational workflow but also ensures that the final products can be easily purified, making it an ideal candidate for a reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing portfolio.
Mechanistic Insights into Copper-Catalyzed Cyclization
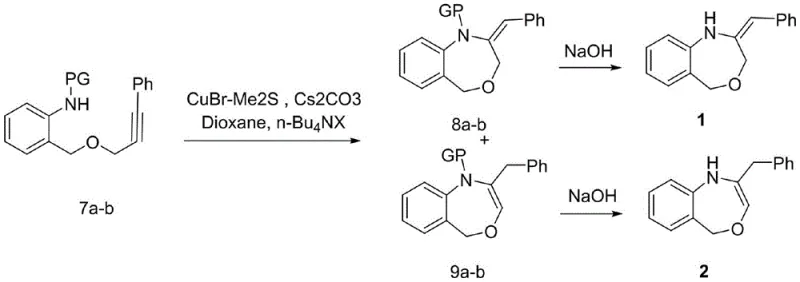
The core of this synthetic innovation is the fifth step, which involves a copper-mediated intramolecular cyclization to form the seven-membered oxazepine ring. This transformation utilizes a specific catalytic system comprising cuprous bromide-dimethyl sulfide complex (CuBr-Me2S), cesium carbonate (Cs2CO3), and a phase transfer catalyst like tetrabutylammonium iodide. The mechanism likely proceeds through the activation of the terminal alkyne or the aryl halide equivalent (if generated in situ), facilitating a nucleophilic attack by the protected amine nitrogen onto the alkyne system. The use of Cs2CO3 is critical here, as weaker bases fail to drive the reaction to completion, highlighting the necessity of strong basic conditions for this specific ring closure. The reaction temperature is carefully maintained between 80°C and 110°C in solvents like 1,4-dioxane to ensure optimal kinetics without degrading the sensitive intermediates.
Following the cyclization, the process yields a mixture of N-protected isomers, specifically the exocyclic alkene (compound 8) and the endocyclic alkene (compound 9). The ability to control and separate these isomers is vital for downstream applications. The patent data indicates that the ratio of these isomers can be influenced by the choice of the protecting group (Ac, Bz, or Ts), with separation achieved via standard column chromatography. Subsequent deprotection strategies are tailored to the specific protecting group employed; for instance, acetyl and benzoyl groups are removed under mild alkaline hydrolysis, while the tosyl group requires dissolving metal reduction using sodium and naphthalene. This mechanistic versatility allows chemists to tune the final product profile, ensuring cost reduction in API manufacturing by minimizing waste and maximizing the recovery of the desired active isomer.
How to Synthesize (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine Efficiently
The synthesis of these valuable heterocycles is achieved through a logical progression of functional group transformations that prioritize yield and purity at every stage. Beginning with the etherification of o-nitrobenzyl alcohol, the process moves through reduction, coupling, and protection before the final ring closure. Each step has been optimized to use commercially viable reagents and standard laboratory equipment, ensuring that the transition from bench-scale discovery to pilot plant production is seamless. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the structured guide below to assist technical teams in replicating this high-efficiency route.
- Perform etherification of o-nitrobenzyl alcohol with propargyl bromide or propargyl alcohol using NaH/Grignard or Mitsunobu conditions to form the nitro-ether intermediate.
- Reduce the nitro group to an amine using iron powder/acetic acid or a nickel-catalyzed system to obtain the aniline derivative.
- Execute a Sonogashira coupling with iodobenzene to introduce the phenylalkyne moiety, followed by amino protection with Ac, Bz, or Ts groups.
- Conduct the key ring-closing reaction using CuBr-Me2S and Cs2CO3 in dioxane to form the seven-membered oxazepine ring isomers.
- Finalize the synthesis by removing the protecting group via alkaline hydrolysis or sodium/naphthalene reduction to yield the target benzoxazepines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical elegance. The reliance on commodity chemicals such as o-nitrobenzyl alcohol, propargyl bromide, and iodobenzene ensures a stable and continuous supply of raw materials, mitigating the risks associated with sourcing exotic or proprietary reagents. Furthermore, the elimination of complex purification steps and the use of robust catalytic systems significantly streamline the manufacturing timeline. This efficiency translates directly into enhanced supply chain reliability, allowing for faster response times to market demands and reducing the lead time for high-purity pharmaceutical intermediates. The process is designed to be inherently scalable, meaning that production volumes can be increased from kilograms to tons without encountering the nonlinear scale-up issues often seen in fine chemical synthesis.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing high-yielding reactions that minimize raw material waste. For example, the initial etherification step achieves yields over 90 percent using Mitsunobu conditions, drastically reducing the cost per kilogram of the intermediate compared to lower-yielding alternatives. Additionally, the use of iron powder for reduction is a classic, low-cost method that avoids the expense of precious metal catalysts in that specific step. By optimizing the stoichiometry of expensive reagents like palladium catalysts in the Sonogashira coupling to low loading levels (1.5-2.5%), the overall material cost is kept competitive, driving down the total cost of goods sold for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The synthetic pathway is constructed using reagents that are widely available in the global chemical market, reducing dependency on single-source suppliers. The robustness of the reaction conditions, such as the tolerance of the cyclization step to various solvents and the stability of the intermediates, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted production schedules in a GMP environment. The ability to easily separate isomers via chromatography further guarantees that the final product meets stringent purity specifications without requiring complex recrystallization protocols that could delay shipment.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process favors reagents and solvents that are manageable on a large scale. The replacement of hazardous reagents with safer alternatives where possible, combined with efficient workup procedures like filtration and extraction, simplifies waste treatment. The high atom economy of the coupling and cyclization steps means less chemical waste is generated per unit of product. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing facility, supporting long-term sustainability goals for the organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of benzoxazepine derivatives. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or research pipelines.
Q: What are the primary advantages of this synthetic route over conventional methods?
A: This method offers significantly improved yields, particularly in the initial etherification step (up to 92% via Mitsunobu), and utilizes mild, scalable conditions avoiding harsh reagents often found in traditional heterocyclic synthesis.
Q: How is the stereoselectivity and isomer separation managed in the final steps?
A: The cyclization produces both exocyclic and endocyclic isomers which are readily distinguishable by TLC and can be effectively separated via standard column chromatography using petroleum ether and ethyl acetate gradients.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process relies on commercially available starting materials like o-nitrobenzyl alcohol and employs robust catalytic systems (Pd/Cu) and workup procedures that are highly amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex heterocycles or large-scale supply of established intermediates, our infrastructure is designed to support your growth from early-stage discovery through to commercial launch.
We invite you to collaborate with us to explore how this advanced synthetic route can benefit your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your time to market while optimizing your manufacturing costs.
