Optimized Multi-Step Synthesis of 2-Benzyl-1,5-Dihydrobenzo[e][1,4]Oxazepine for Commercial Scale-Up
Introduction to Advanced Oxazepine Synthesis
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting broad pharmacological activities. Patent CN108997250B discloses a robust, multi-step methodology for synthesizing 2-benzyl-1,5-dihydrobenzo[e][1,4]oxazepine and its benzylidene analogues, compounds that have garnered significant attention for their potential as anticancer, antitubercular, and neuroprotective agents. This technical disclosure outlines a strategic pathway starting from readily available o-nitrobenzyl alcohol, navigating through etherification, reduction, palladium-catalyzed coupling, and a critical copper-mediated cyclization. For R&D teams and procurement specialists alike, understanding the nuances of this sequence is vital, as it addresses historical limitations in yield and stereoselectivity associated with 1,4-benzoxazepine derivatives. The protocol not only ensures high purity but also optimizes reaction conditions to be mild and environmentally friendlier, positioning it as a viable candidate for cost reduction in API manufacturing where complex heterocyclic cores are required.
![Overall synthetic route from o-nitrobenzyl alcohol to 2-benzyl-1,5-dihydrobenzo[e][1,4]oxazepine showing etherification, reduction, coupling, and cyclization steps](/insights/img/2-benzyl-1-5-dihydrobenzo-e-1-4-oxazepine-synthesis-pharma-supplier-20260313193858-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-benzoxazepine derivatives has been plagued by several synthetic bottlenecks that hinder their widespread application in drug discovery pipelines. Conventional strategies often rely on harsh reaction conditions that compromise functional group tolerance, leading to complex impurity profiles that are difficult to purge during downstream processing. Specifically, traditional etherification steps to install the propargyl side chain frequently suffer from poor conversion rates, with yields often stagnating between 20% and 30% when using standard alkali metal bases in common solvents. Furthermore, existing literature describes few stereoselective methods capable of differentiating between exocyclic and endocyclic double bond formations, limiting the molecular diversity accessible to chemists. These inefficiencies translate directly into higher production costs and extended lead times, creating a significant barrier for commercial scale-up of complex pharmaceutical intermediates. The reliance on stoichiometric amounts of aggressive reagents also generates substantial chemical waste, conflicting with modern green chemistry mandates.
The Novel Approach
The methodology detailed in the patent represents a paradigm shift by introducing high-yielding alternatives for every critical transformation in the sequence. A standout innovation is the adoption of Mitsunobu etherification conditions using triphenylphosphine and azodicarboxylates, which boosts the initial ether formation yield to over 90%, drastically outperforming traditional alkylation. Additionally, the reduction of the nitro group is optimized through either a classic iron/acetic acid system or a modern nickel-catalyzed protocol using tetrahydroxy diboron, both achieving conversions exceeding 85% with minimal byproduct formation. The subsequent Sonogashira coupling proceeds with exceptional efficiency, reaching yields of approximately 98%, ensuring that the precious phenylalkyne motif is installed with near-quantitative fidelity. By integrating these high-efficiency steps with a versatile protecting group strategy (Ac, Bz, or Ts), the novel approach provides a flexible platform that enhances supply chain reliability and reduces the overall cost of goods sold for these valuable heterocyclic scaffolds.
Mechanistic Insights into Copper-Catalyzed Cyclization
The heart of this synthetic strategy lies in the fifth step, where the linear precursor undergoes an intramolecular cyclization to form the seven-membered oxazepine ring. This transformation is mediated by a copper(I) catalyst, specifically cuprous bromide-dimethyl sulfide complex, in the presence of cesium carbonate and a phase transfer catalyst like tetrabutylammonium iodide. Mechanistically, this likely involves the activation of the alkyne by the copper species, facilitating a nucleophilic attack by the protected amine nitrogen or the adjacent oxygen, followed by proton transfer and aromatization or isomerization. The choice of base is critical; screening revealed that inorganic carbonates like potassium or sodium carbonate were ineffective, whereas cesium carbonate drove the reaction to completion. This specificity suggests a delicate balance of basicity and solubility is required to deprotonate the nitrogen without decomposing the sensitive alkyne functionality. The reaction temperature, maintained between 80°C and 110°C in dioxane, provides the necessary thermal energy to overcome the entropic penalty of forming a seven-membered ring, a notoriously difficult task in organic synthesis.
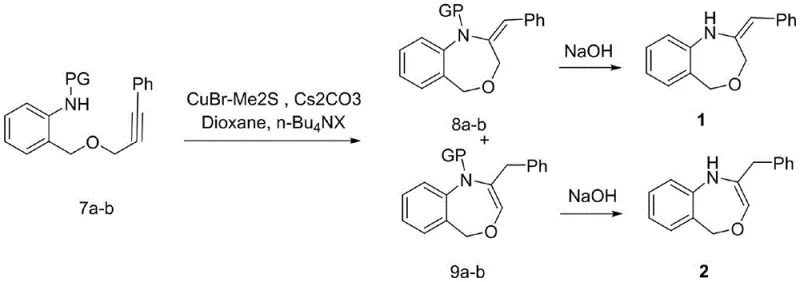
Furthermore, the mechanism elegantly accounts for the formation of two distinct isomeric products: the exocyclic benzylidene derivative (Compound 1) and the endocyclic benzyl derivative (Compound 2). The ratio of these isomers can be subtly influenced by the nature of the protecting group on the nitrogen, with acetyl and benzoyl groups favoring one pathway and the tosyl group altering the electronic environment to shift the product distribution. This mechanistic insight is invaluable for process chemists aiming to steer the reaction towards a specific isomer for biological testing. The final deprotection step, utilizing sodium hydroxide for acyl groups or sodium/naphthalene for the sulfonyl group, cleanly reveals the free amine without disrupting the newly formed heterocyclic core. This robustness ensures that the final high-purity pharmaceutical intermediates meet stringent quality specifications required for preclinical and clinical studies, minimizing the risk of toxic impurities carrying over into the final drug substance.
How to Synthesize 2-Benzyl-1,5-Dihydrobenzo[e][1,4]Oxazepine Efficiently
Executing this synthesis requires precise control over reaction parameters, particularly during the etherification and cyclization stages where side reactions can erode yield. The process begins with the activation of o-nitrobenzyl alcohol, followed by a sequential build-up of the carbon skeleton. Operators must pay close attention to the stoichiometry of the copper catalyst and the phase transfer agent in the final ring-closing step, as deviations here can lead to incomplete conversion or polymerization of the alkyne. The detailed standardized synthetic steps, including specific solvent volumes, temperature ramps, and workup procedures derived from the patent examples, are outlined below to ensure reproducibility and safety in your laboratory or pilot plant.
- Perform etherification of o-nitrobenzyl alcohol with propargyl bromide or propargyl alcohol using Mitsunobu conditions or Grignard reagents to form the nitro-ether intermediate.
- Reduce the nitro group to an amine using iron powder/acetic acid or a nickel-catalyzed system with tetrahydroxy diboron.
- Execute a Sonogashira coupling with iodobenzene to introduce the phenylalkyne moiety, followed by amine protection.
- Conduct the critical ring-closing reaction using CuBr-Me2S and cesium carbonate to form the oxazepine core.
- Finalize the synthesis by deprotecting the amine group using sodium hydroxide or sodium/naphthalene to yield the target heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthetic route offers compelling advantages that directly address the pain points of sourcing complex heterocyclic building blocks. The reliance on commodity chemicals such as o-nitrobenzyl alcohol, propargyl bromide, and iodobenzene ensures that raw material availability is not a bottleneck, mitigating the risk of supply disruptions that often plague specialty chemical procurement. Moreover, the high yields observed in key steps, particularly the Mitsunobu etherification and Sonogashira coupling, mean that less starting material is required to produce a given amount of final product, effectively lowering the material cost per kilogram. The ability to use standard solvents like dichloromethane, ethanol, and dioxane further simplifies logistics, as these are universally available and easily recycled in most manufacturing facilities. This operational simplicity translates into a more resilient supply chain capable of meeting fluctuating demand without significant lead time penalties.
- Cost Reduction in Manufacturing: The elimination of exotic or prohibitively expensive catalysts in favor of earth-abundant metals like iron and copper significantly drives down the direct material costs. While palladium is used in the coupling step, the low loading (approx. 2.5 mol%) and high turnover frequency minimize its impact on the overall budget. Additionally, the high isolated yields across the five-step sequence reduce the volume of waste solvent and reagents that must be treated and disposed of, leading to substantial savings in waste management fees. By optimizing the atom economy and reducing the number of purification cycles needed due to cleaner reaction profiles, manufacturers can achieve a leaner, more cost-effective production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate ambient moisture and oxygen in several steps (such as the iron reduction), reduces the need for specialized inert atmosphere equipment, thereby lowering capital expenditure requirements for production lines. The flexibility to choose between different protecting groups (Ac, Bz, Ts) allows procurement teams to source the most cost-effective reagents at any given time without redesigning the entire process. This adaptability ensures that production schedules remain uninterrupted even if specific reagents face temporary market shortages. Furthermore, the scalability of the process from gram to kilogram scales has been demonstrated through the detailed examples, providing confidence that the supply can be ramped up rapidly to support clinical trial material needs or commercial launch volumes.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by offering a nickel-catalyzed reduction alternative that generates less solid waste compared to the traditional iron sludge, simplifying effluent treatment and regulatory compliance. The use of cesium carbonate, while a heavier metal salt, is employed in stoichiometric amounts that allow for potential recovery and reuse, aligning with sustainability goals. The mild reaction temperatures (mostly reflux or room temperature) reduce energy consumption compared to high-pressure or cryogenic processes. These factors collectively make the technology attractive for contract manufacturing organizations (CMOs) looking to expand their portfolio of eco-friendly synthetic capabilities while maintaining strict adherence to environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route, focusing on yield optimization, impurity control, and scalability. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering practical guidance for process development teams evaluating this technology for adoption.
Q: What are the key advantages of the Mitsunobu etherification method described in the patent?
A: The patent highlights that using Mitsunobu conditions (PPh3/DIAD) for the initial etherification step significantly improves the isolated yield to over 90%, compared to traditional alkylation methods which often suffer from lower yields around 20-30% or require harsh Grignard conditions.
Q: How does the new cyclization method improve molecular diversity?
A: The copper-catalyzed cyclization step allows for the simultaneous generation of both exocyclic (benzylidene) and endocyclic (benzyl) ring-closed products. This dual formation capability offers greater flexibility in generating molecular diversity for drug discovery compared to rigid six-membered ring synthesis strategies.
Q: Is the reduction step scalable for industrial production?
A: Yes, the process offers two scalable reduction pathways. The traditional iron powder/acetic acid method is cost-effective and robust, while the alternative nickel-catalyzed reduction using tetrahydroxy diboron provides a cleaner profile with easier waste solid disposal, making both suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzyl-1,5-Dihydrobenzo[e][1,4]Oxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are equipped with state-of-the-art rigorous QC labs and analytical instrumentation to guarantee stringent purity specifications for every batch of 2-benzyl-1,5-dihydrobenzo[e][1,4]oxazepine we deliver. Whether you require custom synthesis of specific isomers or large-scale supply of the racemic mixture, our infrastructure is designed to meet the demanding timelines of the pharmaceutical industry while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to optimize your supply chain for this valuable heterocyclic scaffold. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By leveraging our manufacturing expertise, you can secure a stable source of this critical intermediate, allowing your R&D team to focus on what they do best: discovering the next generation of life-saving medicines. Contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
