Advanced Multi-Step Synthesis of (E)-2-Benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine for Commercial Scale-Up
Advanced Multi-Step Synthesis of (E)-2-Benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine for Commercial Scale-Up
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as critical cores for bioactive molecules. Patent CN108997251B discloses a robust and versatile method for synthesizing (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine and its dihydro analogues, addressing significant gaps in current synthetic methodologies. This technology is particularly relevant for the development of potential antitubercular agents, anticancer drugs, and 5-HT3 receptor antagonists, where the 1,4-benzoxazepine skeleton plays a pivotal role. By leveraging a multi-step approach that integrates etherification, selective reduction, palladium-catalyzed coupling, and copper-mediated cyclization, this invention provides a reliable pharmaceutical intermediate supplier with a pathway to high-purity compounds. The strategic design of this synthesis not only improves overall yields but also enhances the operational stability required for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,4-benzoxazepine derivatives has been plagued by synthetic inefficiencies and limited molecular diversity. Conventional routes often rely on harsh conditions that compromise functional group tolerance, leading to complex impurity profiles that are difficult to purge during downstream processing. Specifically, traditional etherification steps to install the propargyl moiety frequently utilize strong bases like sodium hydride in polar aprotic solvents, which can result in poor yields ranging typically between 20% and 30% due to competitive side reactions and incomplete conversion. Furthermore, existing methods for closing the seven-membered ring often lack stereoselectivity, producing mixtures of isomers that require tedious purification protocols. These limitations create substantial bottlenecks in reducing lead time for high-purity pharmaceutical intermediates, as process chemists struggle to optimize reaction parameters that balance reactivity with selectivity. The reliance on less efficient reduction methods or expensive catalytic systems in older literature further exacerbates cost issues, making these routes less attractive for large-volume manufacturing.
The Novel Approach
The methodology outlined in CN108997251B introduces a paradigm shift by optimizing each synthetic transformation for maximum efficiency and yield. A standout innovation is the employment of the Mitsunobu reaction or methyl Grignard-assisted etherification in the initial step, which dramatically improves the formation of the nitro-ether intermediate. Unlike the conventional sodium hydride method, the Mitsunobu protocol utilizing triphenylphosphine and azodicarboxylates achieves separation yields exceeding 90% under mild conditions, thereby minimizing waste and maximizing throughput. 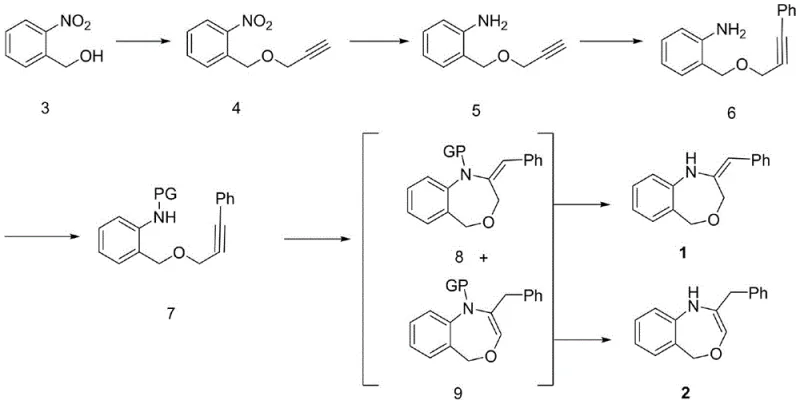 This enhanced efficiency in the early stages sets a strong foundation for the subsequent transformations. Additionally, the patent describes a flexible reduction strategy using either activated iron powder in acetic acid or a nickel-catalyzed system, both of which offer complete conversion of the nitro group to the amine without affecting the sensitive alkyne functionality. This novel approach ensures cost reduction in API manufacturing by utilizing readily available reagents and avoiding the need for high-pressure hydrogenation equipment, thus simplifying the engineering requirements for production facilities.
This enhanced efficiency in the early stages sets a strong foundation for the subsequent transformations. Additionally, the patent describes a flexible reduction strategy using either activated iron powder in acetic acid or a nickel-catalyzed system, both of which offer complete conversion of the nitro group to the amine without affecting the sensitive alkyne functionality. This novel approach ensures cost reduction in API manufacturing by utilizing readily available reagents and avoiding the need for high-pressure hydrogenation equipment, thus simplifying the engineering requirements for production facilities.
Mechanistic Insights into Copper-Catalyzed Cyclization and Coupling
The core of this synthetic strategy lies in the precise orchestration of transition metal catalysis to construct the heterocyclic core. The Sonogashira coupling step, which introduces the phenyl group onto the alkyne chain, utilizes a palladium catalyst such as Pd(PPh3)2Cl2 in conjunction with cuprous iodide and triethylamine. This dual-catalyst system facilitates the cross-coupling of the terminal alkyne with iodobenzene under ambient temperature conditions, ensuring high atom economy and minimal degradation of the amino substrate. Following this, the introduction of a protecting group (PG) such as acetyl, benzoyl, or tosyl is critical to prevent interference of the amine during the subsequent cyclization event. The choice of protecting group dictates the deprotection conditions later in the sequence, offering flexibility depending on the sensitivity of other potential substituents on the molecule. This modularity is essential for medicinal chemists exploring structure-activity relationships (SAR) around the benzoxazepine scaffold.
The final ring-closing step represents a sophisticated application of copper chemistry to form the seven-membered oxazepine ring. Utilizing cuprous bromide-dimethyl sulfide complex and cesium carbonate in dioxane, the reaction proceeds through an intramolecular nucleophilic attack or radical mechanism to close the ring, generating both exocyclic (compound 1) and endocyclic (compound 2) isomers. 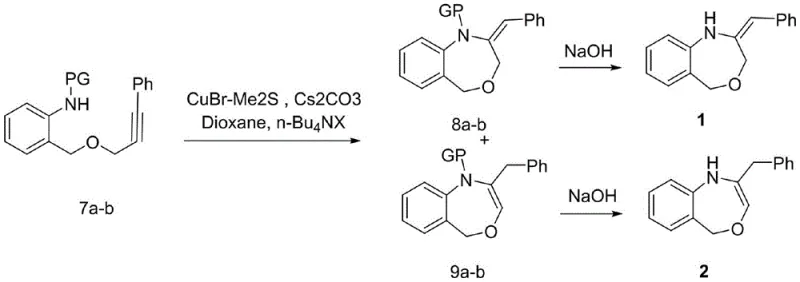 The use of cesium carbonate as the base is particularly noteworthy, as weaker inorganic bases like potassium carbonate fail to drive the reaction to completion, highlighting the specific basicity requirements for this transformation. The resulting N-PG protected intermediates are then subjected to deprotection; for acetyl or benzoyl groups, mild alkaline hydrolysis suffices, whereas the tosyl group requires dissolving metal reduction using sodium and naphthalene. This mechanistic understanding allows for precise control over the final product distribution and purity, ensuring that the output meets the stringent quality standards expected of a reliable agrochemical intermediate supplier or pharma partner.
The use of cesium carbonate as the base is particularly noteworthy, as weaker inorganic bases like potassium carbonate fail to drive the reaction to completion, highlighting the specific basicity requirements for this transformation. The resulting N-PG protected intermediates are then subjected to deprotection; for acetyl or benzoyl groups, mild alkaline hydrolysis suffices, whereas the tosyl group requires dissolving metal reduction using sodium and naphthalene. This mechanistic understanding allows for precise control over the final product distribution and purity, ensuring that the output meets the stringent quality standards expected of a reliable agrochemical intermediate supplier or pharma partner.
How to Synthesize (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine Efficiently
Executing this synthesis requires careful attention to reaction stoichiometry and temperature control, particularly during the etherification and cyclization stages. The process begins with the activation of o-nitrobenzyl alcohol, followed by coupling with the alkyne source, and proceeds through reduction and protection before the final ring closure. Operators must ensure anhydrous conditions during the Mitsunobu reaction to prevent hydrolysis of the reactive intermediates, while the iron powder reduction requires controlled addition of acid to manage exotherms safely. The detailed standardized synthetic steps see the guide below, which outlines the specific molar ratios and workup procedures necessary to achieve the reported high yields. Adhering to these protocols ensures reproducibility and safety, which are paramount when scaling this chemistry from the laboratory bench to pilot plant reactors.
- Perform etherification of o-nitrobenzyl alcohol with propargyl bromide or alcohol using NaH/Grignard or Mitsunobu conditions to form the nitro-ether intermediate.
- Reduce the nitro group to an amine using iron powder/acetic acid or NiCl2(dppp)/tetrahydroxy diboron systems.
- Execute Sonogashira coupling with iodobenzene, followed by amine protection (Ac, Bz, or Ts) and final CuBr-mediated cyclization and deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented route offers distinct advantages that translate directly into operational resilience and cost efficiency for procurement managers. The reliance on commodity chemicals such as o-nitrobenzyl alcohol, propargyl bromide, and iron powder means that raw material sourcing is not constrained by geopolitical risks or single-source supplier dependencies. This abundance of starting materials significantly enhances supply chain reliability, ensuring that production schedules can be maintained even during periods of market volatility. Furthermore, the elimination of specialized high-pressure equipment for the reduction step reduces capital expenditure requirements for manufacturing partners, making the technology accessible to a broader range of contract manufacturing organizations (CMOs). The robustness of the reaction conditions, which tolerate standard industrial solvents like ethanol and dichloromethane, simplifies solvent recovery and recycling processes, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The implementation of the Mitsunobu etherification and iron powder reduction strategies eliminates the need for expensive noble metal catalysts in the reduction phase and drastically improves yields in the initial coupling step. By achieving separation yields of over 90% in the first step compared to the historical average of 20-30%, the overall material throughput is significantly increased, reducing the cost per kilogram of the final active pharmaceutical ingredient. Additionally, the use of inexpensive bases like cesium carbonate and copper salts, rather than exotic ligands, keeps reagent costs low while maintaining high catalytic efficiency. This optimization of reagent selection and yield improvement collectively drives down the total cost of goods sold (COGS), providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The synthetic route is designed with scalability in mind, utilizing reagents that are globally available and stable under standard storage conditions. The avoidance of cryogenic temperatures or ultra-high vacuum systems means that the process can be easily transferred to multipurpose manufacturing plants without extensive retrofitting. This flexibility ensures consistent delivery timelines and reduces the risk of production delays caused by equipment failure or specialized utility shortages. Moreover, the ability to isolate stable intermediates at various stages of the synthesis allows for inventory buffering, further securing the supply chain against unexpected disruptions and enabling just-in-time delivery models for downstream customers.
- Scalability and Environmental Compliance: The process generates waste streams that are manageable through standard treatment protocols, aligning with increasingly strict environmental regulations. The use of iron powder produces iron oxide sludge which is non-toxic and easily disposed of, unlike heavy metal waste from other catalytic systems. The high atom economy of the Sonogashira coupling and the efficient recycling of solvents like dioxane and dichloromethane minimize the environmental footprint of the manufacturing process. This commitment to green chemistry principles not only ensures regulatory compliance but also enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize sustainability in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations and process parameters. Understanding these nuances is critical for process development teams aiming to integrate this technology into their existing manufacturing portfolios. The answers reflect the practical realities of scaling organic synthesis and highlight the specific advantages of this methodology over legacy approaches.
Q: What are the key advantages of the Mitsunobu etherification method described in this patent?
A: The Mitsunobu reaction using triphenylphosphine and DIAD offers significantly milder conditions and higher separation yields (over 90%) compared to traditional sodium hydride methods which often suffer from low conversion and side reactions.
Q: How does the patent address the challenge of stereoselectivity in benzoxazepine synthesis?
A: The process utilizes a specific copper-catalyzed cyclization strategy that favors the formation of the seven-membered oxazepine ring, allowing for the isolation of both exocyclic and endocyclic isomers which can be separated via column chromatography.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the route employs robust reagents like iron powder for reduction and standard solvents like ethanol and dioxane, avoiding extremely sensitive catalysts, which facilitates scalability and cost-effective production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality heterocyclic building blocks for next-generation drug discovery programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of (E)-2-benzylidene-1,2,3,5-tetrahydrobenzo[E][1,4]oxazepine meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of multi-step synthesis, delivering intermediates that empower our clients to accelerate their research and development timelines with confidence.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how adopting this methodology can optimize your budget and improve supply security. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in the global pharmaceutical supply chain. Together, we can drive innovation and bring life-saving therapies to market faster and more efficiently.
