Scalable Multi-Step Synthesis of 2-Benzyl-1-5-Dihydrobenzo-Oxazepine Intermediates
Introduction to Advanced Benzoxazepine Synthesis
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly those containing nitrogen and oxygen atoms which are prevalent in bioactive molecules. Patent CN108774190B discloses a highly efficient multi-step methodology for synthesizing 2-benzyl-1,5-dihydrobenzo[e][1,4]oxazepine and its E-isomer, addressing critical limitations in previous synthetic strategies. This technology represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, offering a route that balances high purity with operational simplicity. The core innovation lies in the strategic combination of high-yield etherification, mild reduction techniques, and a specialized copper-catalyzed cyclization that ensures the formation of the challenging seven-membered ring system. By optimizing reaction conditions such as temperature control and catalyst loading, this process minimizes impurity profiles while maximizing overall throughput.
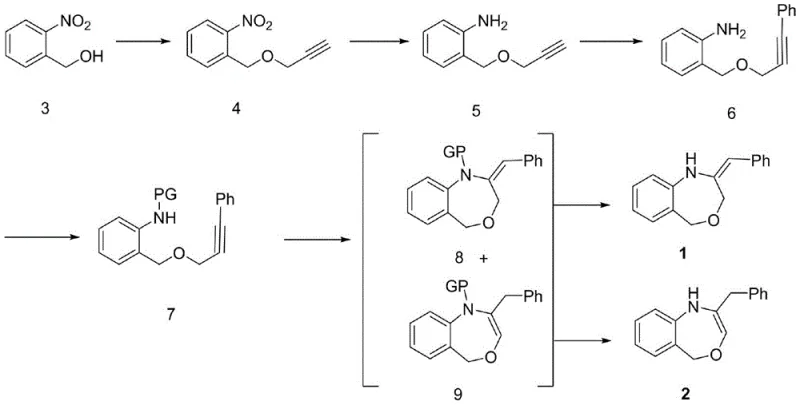
Furthermore, the versatility of this synthetic approach allows for the generation of molecular diversity, which is essential for modern drug discovery programs targeting various therapeutic areas including CNS disorders and oncology. The ability to access both the saturated and unsaturated variants of the oxazepine core from a common intermediate provides medicinal chemists with valuable tools for structure-activity relationship studies. As we delve deeper into the technical specifics, it becomes evident that this patent offers a comprehensive solution for cost reduction in API manufacturing by streamlining what was previously a fragmented and low-yielding sequence of transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,4-benzoxazepine derivatives has been plagued by inefficient etherification steps and harsh reaction conditions that compromise overall yield and purity. Traditional methods often rely on basic alkylation using sodium hydride in polar aprotic solvents, which frequently results in poor conversion rates ranging from merely 20% to 30% due to competing side reactions and incomplete deprotonation. Additionally, the reduction of nitro intermediates using stoichiometric metal powders can generate substantial amounts of solid waste, creating significant disposal challenges and environmental compliance burdens for manufacturing facilities. These conventional routes often lack the stereoselectivity required for specific biological activities, leading to complex mixtures that are difficult and costly to separate. The cumulative effect of these inefficiencies is a supply chain that is vulnerable to delays and cost overruns, making it difficult to secure a reliable source of high-purity intermediates for clinical and commercial needs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent introduces a refined strategy that leverages Mitsunobu etherification or Grignard-mediated alkylation to achieve separation yields exceeding 90% in the initial step. This dramatic improvement in efficiency eliminates the bottleneck typically associated with forming the propargylic ether linkage, ensuring a steady flow of material into subsequent transformation stages. The adoption of catalytic reduction systems, such as nickel-catalyzed protocols with tetrahydroxy diboron, offers a cleaner alternative to traditional iron powder reductions, significantly reducing the burden of heavy metal waste and simplifying downstream purification processes. Moreover, the final cyclization step utilizes a optimized copper catalyst system that promotes the formation of the seven-membered ring with high regioselectivity, overcoming the thermodynamic preferences that often favor six-membered ring formation. This holistic optimization of the synthetic route translates directly into enhanced supply chain reliability and substantial cost savings for procurement teams managing complex API projects.
Mechanistic Insights into Copper-Catalyzed Cyclization
The cornerstone of this synthetic achievement is the final ring-closing step, which employs a sophisticated copper-catalyzed mechanism to construct the benzo[e][1,4]oxazepine core. The reaction proceeds through the activation of the alkyne moiety by a copper species, likely involving the formation of a copper-acetylide intermediate that facilitates nucleophilic attack by the protected amine nitrogen. The presence of cesium carbonate acts as a crucial base to deprotonate the amine and drive the equilibrium towards cyclization, while phase transfer catalysts like tetrabutylammonium iodide enhance the solubility and reactivity of the inorganic base in organic media. Mechanistic studies suggest that the reaction may proceed via an allene intermediate under alkaline conditions, which then undergoes intramolecular hydroamination to close the ring, a pathway that is distinct from standard alkyne activation mechanisms. This specific mechanistic pathway is critical for avoiding the formation of unwanted isomeric byproducts and ensures that the thermodynamic stability of the seven-membered ring is successfully accessed.
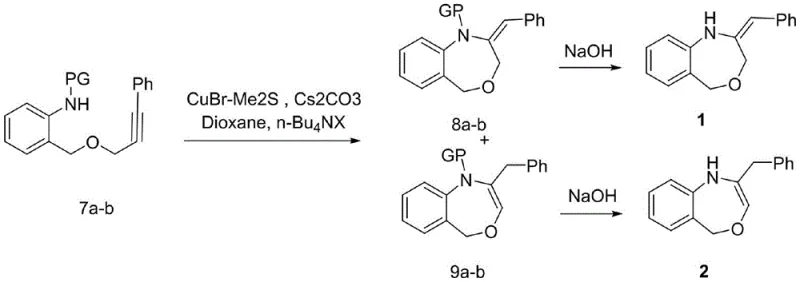
Impurity control is meticulously managed through the selection of protecting groups such as acetyl, benzoyl, or tosyl, which not only prevent premature side reactions but also influence the electronic properties of the nitrogen atom during cyclization. The choice of solvent, specifically 1,4-dioxane, provides an optimal thermal environment for the reaction to proceed at temperatures between 80°C and 110°C without degrading the sensitive alkyne functionality. Following the cyclization, the deprotection step is carefully tuned based on the specific protecting group used; for instance, acetyl and benzoyl groups are removed under mild alkaline hydrolysis, whereas tosyl groups require dissolving metal reduction conditions. This nuanced understanding of the reaction mechanism allows for precise tuning of the process parameters to maximize the ratio of the desired isomers, thereby delivering a product slate that meets the rigorous purity specifications demanded by regulatory agencies.
How to Synthesize 2-Benzyl-1-5-Dihydrobenzo-Oxazepine Efficiently
The synthesis of these valuable heterocyclic intermediates requires a disciplined approach to reaction engineering, beginning with the precise preparation of the nitro-ether precursor using either Mitsunobu conditions or Grignard reagents to ensure high conversion. Subsequent reduction of the nitro group must be monitored closely to prevent over-reduction or degradation of the alkyne handle, utilizing either activated iron powder or the newer nickel-catalyzed protocol depending on waste disposal capabilities. The Sonogashira coupling step introduces the phenyl ring with high fidelity, setting the stage for the critical cyclization event which demands strict control over stoichiometry and temperature to favor the seven-membered ring closure. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures required to execute this synthesis at scale, please refer to the standardized guide below.
- Perform etherification of o-nitrobenzyl alcohol with propargyl bromide or alcohol using Mitsunobu conditions or Grignard reagents to form the nitro-ether intermediate.
- Reduce the nitro group to an amine using iron powder/acetic acid or a nickel-catalyzed system with tetrahydroxy diboron.
- Execute Sonogashira coupling with iodobenzene, followed by amine protection, and finally copper-catalyzed cyclization with cesium carbonate to close the oxazepine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative benefits for organizations seeking to optimize their supply chains for complex heterocyclic intermediates. The shift from low-yielding traditional methods to high-efficiency protocols directly impacts the cost of goods sold by reducing the amount of raw materials required per kilogram of finished product. Furthermore, the use of milder reaction conditions and cleaner catalytic systems reduces the operational complexity of the manufacturing process, lowering the barrier for scale-up from laboratory benchtop to multi-ton commercial production. These factors combine to create a more resilient supply chain that is less susceptible to the volatility often associated with the sourcing of exotic reagents or the management of hazardous waste streams.
- Cost Reduction in Manufacturing: The implementation of high-yield etherification steps, particularly the Mitsunobu reaction which achieves over 90% yield, drastically reduces the consumption of expensive starting materials like o-nitrobenzyl alcohol. By minimizing the loss of material in the early stages of the synthesis, the overall mass balance of the process is improved, leading to significant economic advantages without compromising on quality. Additionally, the ability to use catalytic amounts of nickel for reduction instead of stoichiometric iron powder reduces the volume of solid waste generated, which in turn lowers the costs associated with waste treatment and disposal. This holistic approach to cost management ensures that the final intermediate is produced with maximum economic efficiency, providing a competitive edge in the marketplace.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as propargyl bromide and iodobenzene ensures that the supply chain is not dependent on single-source or custom-synthesized precursors that could introduce bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, means that production can be maintained consistently even when facing minor fluctuations in utility availability or raw material quality. This stability is crucial for maintaining continuous manufacturing operations and meeting the tight delivery schedules required by downstream pharmaceutical customers who depend on just-in-time inventory models. Consequently, partners adopting this technology can offer greater assurance of supply continuity to their own clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are manageable in large-scale reactor systems without requiring exotic high-pressure or cryogenic equipment. The transition to catalytic reduction methods and the optimization of the cyclization step to minimize byproduct formation aligns with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. By reducing the generation of hazardous waste and improving the atom economy of the synthesis, manufacturers can operate with a smaller environmental footprint while simultaneously increasing production capacity. This dual benefit of scalability and sustainability makes the technology highly attractive for long-term commercial partnerships focused on responsible manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these benzoxazepine derivatives, based on the detailed experimental data provided in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing development pipelines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of the Mitsunobu etherification method in this synthesis?
A: The Mitsunobu reaction provides significantly higher yields (over 90%) compared to traditional sodium hydride methods (20-30%), ensuring better material throughput and reduced waste.
Q: How is the regioselectivity of the cyclization step controlled?
A: The use of specific copper catalysts like CuBr-Me2S combined with cesium carbonate and phase transfer catalysts favors the formation of the seven-membered oxazepine ring over other potential isomers.
Q: Is the process suitable for large-scale commercial production?
A: Yes, the route utilizes commercially available starting materials like o-nitrobenzyl alcohol and avoids extremely hazardous conditions, making it viable for scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzyl-1-5-Dihydrobenzo-Oxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics, and we are uniquely positioned to support your needs with our advanced manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage discovery or late-stage clinical trials. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2-benzyl-1,5-dihydrobenzo[e][1,4]oxazepine meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis to your specific project needs and provide a Customized Cost-Saving Analysis for your supply chain. By partnering with us, you gain access to our deep technical expertise and robust infrastructure, allowing you to focus on your core drug discovery objectives while we manage the complexities of chemical manufacturing. Please reach out today to request specific COA data and route feasibility assessments that demonstrate our commitment to being your trusted partner in pharmaceutical innovation.
