Industrial Scale-Up of L-Phenylephrine Hydrochloride via Optimized Rhodium Catalysis
Industrial Scale-Up of L-Phenylephrine Hydrochloride via Optimized Rhodium Catalysis
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective routes for critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN100357255C presents a significant technological breakthrough in the manufacturing of L-phenylephrine hydrochloride, a vital sympathomimetic amine used extensively as a vasoconstrictor in hypotension treatment and ophthalmology. This patent details an improved industrial-scale process that leverages asymmetric hydrogenation as the pivotal stereocontrol step, utilizing a highly efficient rhodium-based catalyst system. By optimizing the catalyst loading and reaction conditions, this method addresses longstanding challenges in purity and throughput that have plagued conventional synthesis routes. For global procurement teams and R&D directors, understanding this technology is essential for securing a reliable L-phenylephrine hydrochloride supplier capable of meeting stringent pharmacopeial standards while minimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing L-phenylephrine, such as those described by Achiwa et al., typically relied on asymmetric hydrogenation using rhodium catalysts but suffered from significant inefficiencies that hindered large-scale adoption. A primary drawback was the excessively high catalyst loading required to achieve acceptable conversion, often necessitating a molar ratio of catalyst to substrate around 1:2000 or 1:1000. This high loading not only inflated raw material costs due to the expense of rhodium and chiral ligands but also complicated downstream purification, as removing trace heavy metals to meet pharmaceutical specifications became a burdensome and costly endeavor. Furthermore, these conventional processes were time-intensive, requiring reaction times of approximately 20 hours to complete the hydrogenation step, which severely limited reactor turnover rates and overall plant capacity.
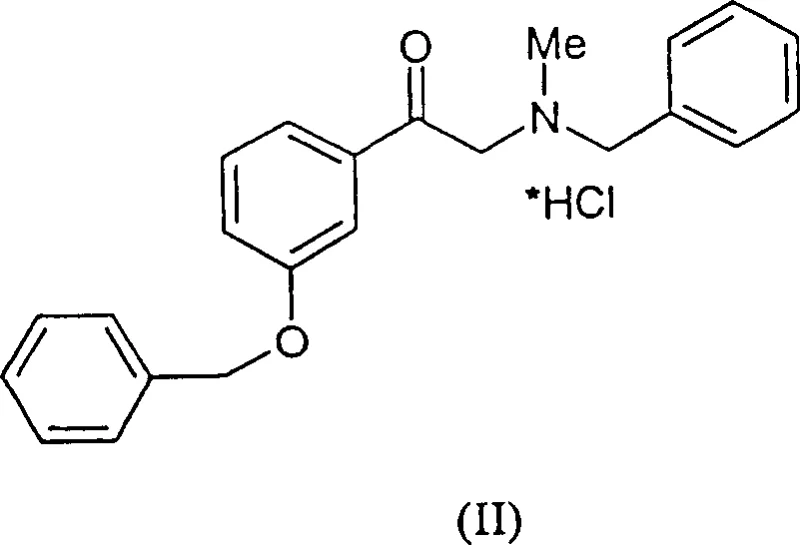
In addition to economic and throughput issues, the optical purity achieved by older methods was often insufficient for direct pharmaceutical use without extensive and yield-loss-inducing recrystallization steps. Conventional routes frequently yielded products containing significant amounts of the undesired D-enantiomer, with optical purity often stagnating around 85% ee. Achieving the requisite >98% ee for drug substance application typically demanded additional, expensive purification protocols that eroded profit margins. The combination of long cycle times, high catalyst consumption, and difficult purification created a supply chain bottleneck, making it challenging for manufacturers to offer cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or safety standards.
The Novel Approach
The methodology outlined in patent CN100357255C introduces a paradigm shift by demonstrating that high optical yields and chemical purity can be achieved with drastically reduced catalyst concentrations. Contrary to the prevailing wisdom that high catalyst loading was necessary for good enantioselectivity, this invention proves that the molar ratio of the rhodium catalyst system to the substrate can be reduced by 10 to 100 times, potentially reaching ratios as low as 1:10,000 or even 1:100,000 while still maintaining robust reaction performance. This reduction is transformative for commercial scale-up of complex pharmaceutical intermediates, as it directly lowers the cost of goods sold (COGS) and simplifies the removal of metal residues. The process utilizes a specific chiral, bicoordinated phosphine ligand, such as (2R,4R)-4-(dicyclohexylphosphino)-2-(diphenylphosphinomethyl)-N-methyl-aminocarbonylpyrrolidine (RR-MCCPM), in conjunction with [Rh(COD)Cl]2 to create a highly active and selective catalytic species.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the sophisticated interplay between the rhodium precursor and the chiral diphosphine ligand, which creates a sterically constrained environment favoring the formation of the L-enantiomer. While the exact mechanistic details of the catalytic cycle remain a subject of academic study, the empirical data confirms that this specific ligand architecture facilitates rapid hydrogen transfer to the prochiral ketone substrate. The reaction proceeds efficiently in protic solvents like methanol or ethanol, which are not only cost-effective but also environmentally preferable compared to chlorinated solvents often used in older methodologies. The catalyst system operates effectively at moderate temperatures ranging from 40°C to 60°C and hydrogen pressures between 10 to 50 bar, striking an optimal balance between reaction kinetics and operational safety.
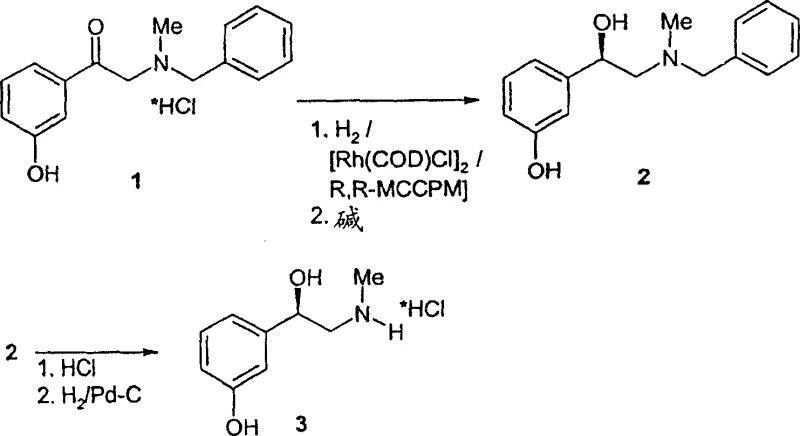
A critical aspect of the impurity control mechanism involves the strategic purification of the intermediate, N-benzyl-L-phenylephrine (compound 2), prior to the final debenzylation step. The patent reveals that while the initial asymmetric hydrogenation yields the intermediate with an optical purity of approximately 92% ee, this intermediate can be easily upgraded to >99% ee through crystallization or precipitation from an ammonia/methanol/water mixture. This "intermediate purification" strategy is far more effective than attempting to purify the final phenylephrine hydrochloride, as the benzyl protecting group alters the physicochemical properties of the molecule, facilitating the separation of the unwanted D-enantiomer. By ensuring the chirality is locked in at the intermediate stage, the subsequent palladium-catalyzed debenzylation proceeds without racemization, delivering the final API with exceptional stereochemical integrity.
How to Synthesize L-Phenylephrine Hydrochloride Efficiently
The synthesis protocol described in the patent offers a clear pathway for laboratory and pilot-scale replication, emphasizing the importance of oxygen-free conditions and precise temperature control during the catalyst activation phase. The process begins with the in-situ or pre-formation of the active rhodium complex in degassed methanol, followed by the introduction of the substrate, N-benzyl-N-methyl-2-amino-hydroxy acetophenone hydrochloride. To enhance solubility, the hydrochloride salt is converted to its free base form using organic or inorganic bases such as triethylamine or sodium carbonate prior to hydrogenation. The detailed standardized synthesis steps below outline the precise conditions required to achieve the high yields and purity reported in the intellectual property.
- Prepare the catalyst system by mixing [Rh(COD)Cl]2 with the chiral ligand RR-MCCPM in degassed methanol under protective gas.
- Conduct asymmetric hydrogenation of N-benzyl-N-methyl-2-amino-hydroxy acetophenone hydrochloride at 50-55°C and 20 bar hydrogen pressure for 4-6 hours.
- Perform catalytic debenzylation using palladium on carbon under hydrogen pressure to yield the final L-phenylephrine hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized synthesis route translates into tangible strategic benefits that extend beyond simple unit price negotiations. The ability to produce high-purity pharmaceutical intermediates with significantly reduced reliance on precious metal catalysts creates a more resilient and cost-stable supply chain. By minimizing the input of expensive rhodium complexes, manufacturers can insulate themselves from volatility in the precious metals market, ensuring more consistent pricing for downstream partners. Furthermore, the streamlined process flow reduces the number of unit operations and the duration of batch cycles, which directly enhances manufacturing throughput and asset utilization rates without requiring additional capital expenditure on new reactor trains.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic reduction in catalyst loading. Since rhodium and specialized chiral ligands represent a significant portion of the variable costs in asymmetric synthesis, lowering the catalyst-to-substrate ratio by up to 100-fold results in substantial cost savings per kilogram of product. Additionally, the simplified purification process eliminates the need for complex chromatographic separations or multiple recrystallizations that typically result in yield loss. This efficiency gain means that more of the raw material input is converted into saleable product, effectively lowering the waste disposal costs and improving the overall mass balance of the production facility.
- Enhanced Supply Chain Reliability: The reduction in reaction time from approximately 20 hours to just 4-6 hours per batch dramatically increases the available capacity of existing manufacturing infrastructure. This acceleration allows suppliers to respond more rapidly to fluctuations in market demand, reducing lead times for high-purity pharmaceutical intermediates. Moreover, the use of common, non-hazardous solvents like methanol and moderate hydrogen pressures reduces the regulatory burden and safety risks associated with production, minimizing the likelihood of unplanned shutdowns due to safety incidents or environmental compliance issues. This reliability is crucial for pharmaceutical companies that require uninterrupted supply to maintain their own production schedules.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing standard equipment and conditions that are easily transferred from pilot plants to multi-ton production reactors. The shift towards lower catalyst usage also aligns with green chemistry principles by reducing the heavy metal load in waste streams, thereby simplifying effluent treatment and lowering environmental compliance costs. The ability to achieve >99% ee through crystallization rather than resource-intensive chromatography further underscores the environmental sustainability of this route, making it an attractive option for companies aiming to reduce their carbon footprint and meet increasingly strict global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of L-phenylephrine hydrochloride using this advanced catalytic method. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process robustness and quality control measures. Understanding these details helps stakeholders evaluate the feasibility of integrating this supply source into their broader procurement strategies.
Q: How does the new rhodium catalyst system improve optical purity compared to prior art?
A: The novel process utilizes a specific chiral bidentate phosphine ligand (RR-MCCPM) with a rhodium precursor, allowing for significantly lower catalyst loading while maintaining high enantioselectivity. Crucially, the process includes a purification step at the N-benzyl-L-phenylephrine intermediate stage, which upgrades the optical purity from approximately 92% ee to over 99% ee before the final debenzylation step.
Q: What are the safety advantages of this manufacturing method?
A: The optimized method reduces the asymmetric hydrogenation reaction time from approximately 20 hours in conventional methods to just 4-6 hours. This drastic reduction in processing time, combined with moderate hydrogen pressures (10-50 bar), significantly lowers the operational risk profile associated with prolonged high-pressure hydrogenation runs in large-scale reactors.
Q: Can this process be scaled for commercial API production?
A: Yes, the patent explicitly describes the method as suitable for technical and industrial scales. The use of common protic solvents like methanol, moderate temperatures (40-60°C), and the ability to reduce expensive catalyst loading by up to 100-fold makes the process economically viable and robust for multi-kilogram to ton-scale manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Phenylephrine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and rigorous process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the rhodium-catalyzed asymmetric hydrogenation process are fully realized in our manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of L-phenylephrine hydrochloride meets or exceeds the optical purity (>99% ee) and chemical purity (>99%) benchmarks set forth in the latest pharmacopeial standards.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic advantages of sourcing from our facility. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that balance quality, cost, and supply security for your critical pharmaceutical supply chain.
