Advanced Chiral Synthesis of L-Phenylephrine Hydrochloride for Commercial Scale-Up
Advanced Chiral Synthesis of L-Phenylephrine Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of critical adrenergic agonists. A recent technological breakthrough, documented in patent CN111689869A, introduces a highly efficient preparation method for L-phenylephrine hydrochloride, a vital active pharmaceutical ingredient used in treating hypotension and as a mydriatic agent. This novel methodology fundamentally shifts the paradigm from traditional, inefficient resolution techniques to a streamlined, asymmetric synthesis route. By leveraging a specific chiral borane reduction strategy, the process achieves superior stereocontrol while utilizing inexpensive, readily available starting materials. For global procurement leaders and R&D directors, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The technology eliminates the reliance on high-pressure hydrogenation equipment and expensive transition metal catalysts, offering a greener, more economically viable alternative that aligns perfectly with modern sustainable manufacturing goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of optically active L-phenylephrine hydrochloride has been plagued by significant technical and economic bottlenecks associated with traditional synthetic routes. The most common legacy method involves the resolution of racemic mixtures, a process inherently flawed by its maximum theoretical yield of only 50 percent, necessitating the disposal or complex recycling of the unwanted enantiomer. Furthermore, the crystallization steps required for resolution are often difficult to control, leading to inconsistent precipitation and challenging mother liquor treatments that generate substantial chemical waste. Alternatively, asymmetric catalytic hydrogenation, while more direct, typically demands the use of prohibitively expensive chiral metal catalysts containing precious metals like rhodium or ruthenium. These hydrogenation processes also require specialized high-pressure reactors, introducing significant safety risks and capital expenditure barriers that hinder flexible manufacturing and rapid scale-up capabilities in standard chemical plants.
The Novel Approach
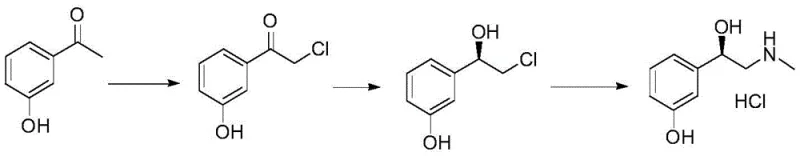
In stark contrast to these legacy challenges, the innovative pathway disclosed in the patent data utilizes a mild, three-step sequence centered around a highly selective chiral reduction. This approach begins with a straightforward chlorination of acetophenone derivatives, followed by the critical asymmetric reduction using a borane-amine complex, and concludes with a simple amination and salification. By employing (1R,2S)-1-amino-2-indanol as a chiral ligand, the process achieves excellent enantioselectivity without the need for high-pressure infrastructure. This method not only simplifies the operational workflow but also drastically reduces the environmental footprint by avoiding heavy metal contamination. The result is a process that delivers product quality meeting stringent pharmacopoeia standards through simpler purification steps, such as recrystallization, rather than complex chromatographic separations. This structural simplicity in the synthesis route translates directly into enhanced process reliability and reduced operational complexity for manufacturing teams.
Mechanistic Insights into Chiral Borane Reduction
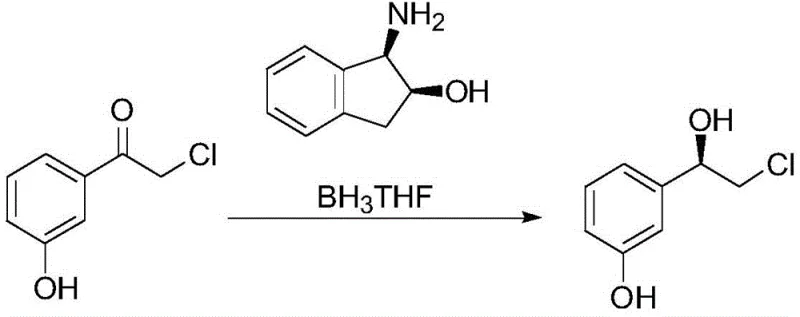
The core of this technological advancement lies in the sophisticated yet practical mechanism of the chiral reduction step, where the stereochemistry of the final drug molecule is established. The reaction utilizes a catalytic system formed in situ from (1R,2S)-1-amino-2-indanol and borane-tetrahydrofuran complex. Mechanistically, the amino alcohol ligand coordinates with the borane species to generate a highly reactive, chiral borane intermediate. This activated complex then approaches the prochiral ketone substrate, 2-chloro-3'-hydroxyacetophenone, in a highly organized transition state. The steric bulk of the indanol framework directs the delivery of the hydride ion specifically to one face of the carbonyl group, ensuring the formation of the desired (R)-configured alcohol intermediate with high fidelity. This precise control over the stereochemical outcome is crucial, as it minimizes the formation of the undesired enantiomer right from the early stages of synthesis, thereby reducing the burden on downstream purification processes.
Furthermore, the impurity profile of this reaction is exceptionally clean due to the mild reaction conditions and the specificity of the reagents. Unlike harsh reduction methods that might reduce other functional groups or cause dehalogenation, this borane-mediated reduction is chemoselective for the ketone moiety in the presence of the aryl chloride. The process operates effectively at temperatures ranging from 0 to 30 degrees Celsius, preventing thermal degradation of sensitive intermediates. The subsequent workup involves a simple quenching with methanol and phase separation, which efficiently removes boron-containing byproducts. This mechanistic elegance ensures that the crude product entering the final amination step already possesses high optical purity, facilitating the final recrystallization to achieve the required greater than 99 percent chiral purity specified by international pharmacopoeias.
How to Synthesize L-Phenylephrine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and temperature control during the chiral reduction phase to maximize enantiomeric excess. The process is designed to be telescoped where possible, minimizing isolation steps and solvent usage. The initial chlorination sets the stage for the reduction, and the final amination locks in the amine functionality. Detailed standard operating procedures regarding reagent addition rates, specific solvent grades, and crystallization parameters are critical for reproducibility. For a comprehensive breakdown of the exact experimental conditions, reagent ratios, and troubleshooting tips derived directly from the patent examples, please refer to the standardized guide below.
- Perform chlorination of 3-hydroxyacetophenone using sulfuryl chloride in an inert solvent like dichloromethane at 0-25°C to obtain 2-chloro-3'-hydroxyacetophenone.
- Conduct chiral reduction using (1R,2S)-1-amino-2-indanol and borane-THF complex at 0-30°C to stereoselectively form (R)-3-(2-chloro-1-hydroxy-ethyl) phenol.
- Execute methylamination followed by salification with hydrochloric acid and recrystallization to achieve final L-phenylephrine hydrochloride with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing route offers profound advantages that directly impact the bottom line and supply security. The elimination of precious metal catalysts removes a major source of cost volatility and supply risk, as the prices of metals like rhodium can fluctuate wildly based on geopolitical factors. Additionally, the avoidance of high-pressure hydrogenation equipment means that production can be outsourced to a wider range of contract manufacturing organizations that possess standard glass-lined or stainless steel reactors, rather than limited facilities with specialized high-pressure capabilities. This flexibility significantly enhances supply chain resilience, allowing for multi-vendor sourcing strategies that mitigate the risk of production stoppages. The simplified purification process also reduces the consumption of solvents and energy, contributing to a lower overall cost of goods sold while aligning with corporate sustainability mandates.
- Cost Reduction in Manufacturing: The substitution of expensive chiral metal catalysts with an organocatalytic borane system results in substantial raw material cost savings. By removing the need for costly metal scavengers and extensive purification steps to meet heavy metal limits, the downstream processing costs are drastically simplified. The high yield and selectivity of the reaction minimize waste generation, further lowering the cost associated with waste disposal and raw material inefficiency. This economic efficiency makes the final API intermediate significantly more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetophenone derivatives, sulfuryl chloride, and methylamine ensures a stable and robust supply of starting materials. These feedstocks are produced on a massive industrial scale globally, meaning there is little risk of shortage compared to specialized chiral ligands or gases required for hydrogenation. The mild reaction conditions also reduce the likelihood of safety-related shutdowns, ensuring consistent delivery schedules. This reliability is paramount for pharmaceutical companies managing tight inventory levels and Just-In-Time manufacturing models.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the engineering complexities associated with handling high-pressure hydrogen gas. Scaling from pilot plant to commercial tonnage is straightforward, requiring only standard agitation and temperature control systems. Furthermore, the absence of heavy metals simplifies environmental compliance and wastewater treatment, reducing the regulatory burden on manufacturing sites. This ease of scale-up ensures that supply can be rapidly ramped up to meet surging market demand for adrenergic medications without lengthy process re-validation periods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived from the specific technical disclosures and comparative data found within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs. The insights provided here clarify the operational benefits and quality assurances associated with this method.
Q: What are the advantages of this chiral reduction method over traditional resolution?
A: Unlike traditional racemate resolution which suffers from low theoretical yield (max 50%) and complex mother liquor treatment, this asymmetric borane reduction directly constructs the chiral center with high efficiency, eliminating the need for multiple recrystallizations to remove unwanted enantiomers.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes an organocatalytic system based on (1R,2S)-1-amino-2-indanol and borane, avoiding the use of costly precious metal catalysts like rhodium or ruthenium often required in asymmetric hydrogenation, thereby significantly reducing raw material costs.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (0-40°C) and do not require high-pressure equipment, making it inherently safer and easier to scale up compared to high-pressure hydrogenation methods, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Phenylephrine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the pharmaceutical sector. Our team of expert chemists has thoroughly analyzed this patented route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to translate this laboratory-scale innovation into a robust, GMP-compliant manufacturing process that guarantees stringent purity specifications. Our rigorous QC labs are prepared to validate every batch against the highest international standards, ensuring that the L-phenylephrine hydrochloride we supply meets the exacting requirements of your final drug formulations.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free process. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your volume needs. Let us help you secure a sustainable, high-quality supply of this essential pharmaceutical intermediate.
