Advanced Ru-Catalyzed Asymmetric Hydrogenation for Commercial L-Phenylephrine Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical vasoconstrictors, and the methodology disclosed in patent CN102234237B represents a significant advancement in the production of L-phenylephrine hydrochloride. This chiral alpha-amino alcohol is a cornerstone active pharmaceutical ingredient (API) widely utilized in hypotension treatment and ophthalmology for its potent sympathetic nerve effects. Traditional manufacturing pathways have often been plagued by the reliance on prohibitively expensive noble metal catalysts and complex multi-step sequences that hinder scalability. The novel approach detailed in this intellectual property leverages a ruthenium-catalyzed asymmetric hydrogenation strategy that fundamentally alters the economic and technical landscape of production. By shifting from rare rhodium complexes to more accessible ruthenium systems, the process offers a viable pathway for cost reduction in pharmaceutical intermediates manufacturing without compromising the stringent purity profiles required for human consumption. This report analyzes the technical merits of this innovation, providing R&D and procurement leaders with a clear understanding of its commercial potential.
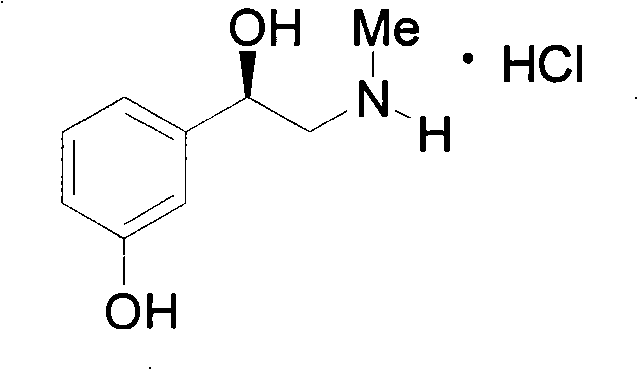
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical precedents for synthesizing L-phenylephrine, such as those documented in Tetrahedron Letters and earlier patent filings like WO2000043345, rely heavily on rhodium-based catalytic systems which present substantial logistical and financial barriers. These conventional methods typically require catalyst-to-substrate ratios that are economically inefficient, often necessitating the use of expensive ligands that are sensitive to oxygen and moisture. The operational complexity is further compounded by the need for rigorous exclusion of air and water, demanding specialized equipment and increasing the risk of batch failure during commercial scale-up of complex pharmaceutical intermediates. Additionally, the purification processes associated with these older routes often involve tedious resolution steps using chiral acids, which inherently limit the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. The cumulative effect of these factors is a high cost of goods sold (COGS) and a supply chain that is vulnerable to fluctuations in the availability of precious metals, creating significant uncertainty for procurement managers seeking long-term stability.
The Novel Approach
In stark contrast, the methodology outlined in CN102234237B introduces a streamlined protocol that utilizes a Ru-BINAP catalyst system, which is not only more cost-effective but also exhibits superior stability under ambient conditions. This novel approach simplifies the synthetic sequence by integrating a debenzylation step prior to the key asymmetric hydrogenation, thereby generating a simpler prochiral substrate that is more amenable to high-selectivity reduction. The use of ruthenium allows for broader tolerance to reaction conditions, including the use of protic solvents like methanol and ethanol, which are cheaper and safer than the aprotic solvents often required by rhodium catalysts. Furthermore, the solid state of the synthesized catalyst facilitates easier handling and dosing, eliminating the need for complex in-situ generation procedures that can introduce variability. This transition to a more robust catalytic system directly addresses the pain points of reducing lead time for high-purity pharmaceutical intermediates by minimizing process development time and enhancing batch-to-batch consistency.
Mechanistic Insights into Ru-BINAP Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the precise orchestration of the asymmetric hydrogenation mechanism, where the chiral environment created by the BINAP ligand dictates the stereochemical outcome of the reaction. The ruthenium center coordinates with the prochiral ketone substrate, facilitating the transfer of hydrogen atoms in a highly stereoselective manner to generate the desired L-enantiomer. The presence of the hydroxyl group in the meta-position of the aromatic ring plays a crucial role in substrate-catalyst interaction, potentially through hydrogen bonding that locks the conformation and enhances enantioselectivity. Understanding this mechanistic nuance is vital for R&D directors as it explains how the process achieves high optical purity without the need for extensive downstream chiral chromatography. The ability to tune the electronic properties of the ligand and the metal center allows for optimization of the turnover number (TON), ensuring that minimal catalyst loading is required to drive the reaction to completion efficiently.
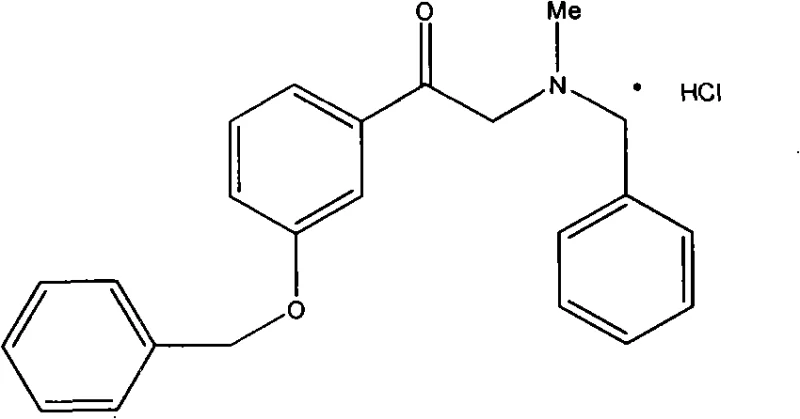
Impurity control is another critical aspect where this mechanism excels, particularly in the management of the D-enantiomer and other structural analogs. The patent details specific purification protocols, such as converting the hydrochloride salt to the free base and re-precipitating, which leverages solubility differences to exclude unwanted isomers. This chemical logic ensures that the final product meets the rigorous specifications for high-purity pharmaceutical intermediates demanded by regulatory bodies. The debenzylation step, utilizing Pd-C, is also designed to minimize over-reduction or side reactions, preserving the integrity of the ketone functionality for the subsequent hydrogenation. By controlling the hydrogen pressure and temperature within specific windows, the process avoids the formation of racemic mixtures, thereby maximizing the yield of the therapeutically active L-form. This level of control over the impurity profile is essential for ensuring patient safety and regulatory compliance in the final drug product.
How to Synthesize L-Phenylephrine Hydrochloride Efficiently
The practical implementation of this synthesis route involves a sequence of well-defined unit operations that can be readily adapted to existing manufacturing infrastructure. The process begins with the preparation of the key intermediate through catalytic debenzylation, followed by the critical asymmetric hydrogenation step using the pre-formed Ru-BINAP complex. Detailed standard operating procedures (SOPs) regarding solvent selection, hydrogen pressure settings, and temperature controls are essential to replicate the high yields and selectivity reported in the patent data. For technical teams looking to adopt this technology, understanding the nuances of catalyst activation and workup procedures is paramount to success. The following guide outlines the standardized synthesis steps derived from the patent embodiments to facilitate technology transfer and process validation.
- Perform catalytic debenzylation on N-benzyl-N-methyl-2-amino-m-hydroxy acetophenone hydrochloride using Pd-C under hydrogen pressure to obtain the N-methyl intermediate.
- Conduct asymmetric hydrogenation on the intermediate using a Ru-BINAP catalyst system in a protic solvent like methanol at moderate temperatures.
- Purify the crude L-phenylephrine hydrochloride through recrystallization or free base precipitation to achieve high optical purity and chemical stability.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Ru-catalyzed route offers compelling advantages that resonate deeply with the strategic goals of procurement and supply chain leadership. The primary benefit stems from the drastic reduction in raw material costs associated with replacing rhodium with ruthenium, which translates directly into improved margin structures for the final API. Beyond mere material costs, the operational simplicity of the process reduces the burden on utility consumption and waste treatment, contributing to a more sustainable and economically viable manufacturing model. For supply chain heads, the robustness of the catalyst and the use of common solvents mitigate the risks associated with supply disruptions of exotic reagents. This reliability ensures a consistent flow of materials, which is critical for maintaining production schedules and meeting market demand without interruption.
- Cost Reduction in Manufacturing: The substitution of expensive rhodium catalysts with ruthenium-based systems eliminates a major cost driver in the synthetic route, leading to substantial savings in direct material expenses. Additionally, the ability to recycle the Pd-C catalyst used in the debenzylation step multiple times further amortizes the cost of consumables over a larger production volume. The simplified purification process reduces the consumption of resolving agents and solvents, lowering the overall environmental footprint and waste disposal costs. These cumulative efficiencies result in a significantly lower cost of goods, allowing for more competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: The use of commercially available and stable catalysts reduces dependency on single-source suppliers of specialized reagents, thereby diversifying the supply base and reducing procurement risk. The solid nature of the Ru-BINAP catalyst allows for long-term storage without degradation, enabling manufacturers to maintain strategic stockpiles that buffer against market volatility. Furthermore, the mild reaction conditions reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime. This operational resilience ensures a steady and predictable supply of reliable L-phenylephrine hydrochloride supplier products to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations that can be easily expanded from pilot plant to full commercial production scales. The use of greener solvents like alcohols and water aligns with increasingly stringent environmental regulations, reducing the need for complex solvent recovery systems. The high atom economy of the hydrogenation reaction minimizes waste generation, supporting corporate sustainability goals and reducing the regulatory burden associated with hazardous waste management. This alignment with green chemistry principles enhances the brand reputation and marketability of the manufactured intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios. The responses cover catalyst performance, purification efficacy, and scalability considerations relevant to industrial application.
Q: How does the Ru-BINAP catalyst improve cost efficiency compared to traditional Rhodium methods?
A: The Ru-BINAP catalyst system utilizes Ruthenium, which is significantly more abundant and cost-effective than the Rhodium complexes traditionally used in asymmetric hydrogenation. Furthermore, the solid nature of the catalyst allows for easier handling and storage, reducing operational overheads.
Q: What purification strategies ensure high optical purity in this synthesis route?
A: The process employs robust purification techniques including recrystallization from alcoholic solvents and conversion to the free base followed by re-salt formation. These steps effectively remove impurities and enhance the enantiomeric excess (ee) to meet stringent medicinal standards.
Q: Is the debenzylation step scalable for industrial production?
A: Yes, the debenzylation step utilizes Pd-C, a heterogeneous catalyst that can be recycled multiple times. The reaction conditions involve moderate hydrogen pressure and temperature, making it highly suitable for large-scale batch processing in standard industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Phenylephrine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the Ru-catalyzed pathway described in CN102234237B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of L-phenylephrine hydrochloride meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis route can be tailored to your specific supply chain requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel method. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate our commitment to quality and innovation. Let us be your trusted partner in delivering high-quality pharmaceutical intermediates that drive your business forward.
