Advanced Manufacturing of L-Phenylephrine Hydrochloride via Chiral Borane Reduction Technology
Introduction to Next-Generation L-Phenylephrine Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical cardiovascular agents, and the preparation method disclosed in patent CN111689869A represents a significant technological leap for L-phenylephrine hydrochloride production. This specific intellectual property outlines a streamlined three-step sequence that effectively bypasses the historical bottlenecks associated with traditional manufacturing, such as low yields and harsh reaction conditions. By leveraging a sophisticated chiral reduction strategy, the process ensures high stereochemical fidelity while utilizing cost-effective reagents that are readily available on the global chemical market. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for evaluating potential supply chain partners who can deliver high-purity pharmaceutical intermediates with consistent reliability. The methodology not only addresses the growing demand for optically active drugs but also aligns with modern green chemistry principles by minimizing waste generation and eliminating the need for high-pressure equipment.
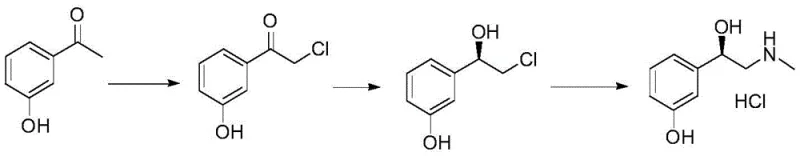
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-phenylephrine hydrochloride has been plagued by significant inefficiencies inherent to racemate resolution and asymmetric catalytic hydrogenation techniques. The racemate resolution approach, while chemically straightforward, suffers from a fundamental theoretical yield limitation of merely 50%, necessitating the disposal or complex recycling of the unwanted enantiomer, which drastically inflates production costs and environmental burden. Furthermore, this traditional method often involves multiple crystallization steps that are difficult to control, leading to inconsistent crystal precipitation and challenging mother liquor treatments that complicate scale-up operations. On the other hand, asymmetric catalytic hydrogenation, although capable of high enantioselectivity, relies heavily on expensive chiral metal catalysts containing precious metals like rhodium or ruthenium, which pose severe contamination risks and require rigorous removal protocols to meet pharmacopoeial standards. Additionally, the requirement for specialized high-pressure hydrogenation equipment introduces substantial capital expenditure and safety hazards, making the process less attractive for flexible multi-purpose manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a clever combination of electrophilic chlorination followed by a highly stereoselective borane reduction mediated by a chiral amino-alcohol ligand. This route begins with the efficient chlorination of acetophenone derivatives under mild conditions, setting the stage for a pivotal asymmetric reduction that installs the critical chiral center with high fidelity without the need for pressurized hydrogen gas. The use of (1R,2S)-1-amino-2-indanol as a chiral inducer allows for the formation of a reactive borane complex in situ, which delivers hydride to the ketone substrate with exceptional facial selectivity, thereby generating the desired (R)-configured intermediate directly. This strategy effectively decouples the synthesis from the constraints of precious metal catalysis and high-pressure infrastructure, offering a safer and more economically viable pathway that is inherently easier to scale from kilogram to multi-ton quantities. The subsequent amination and salt formation steps are equally robust, utilizing common reagents like methylamine and hydrochloric acid to finalize the API intermediate structure with minimal purification overhead.
Mechanistic Insights into Chiral Borane Reduction
The core of this synthetic innovation lies in the mechanistic elegance of the chiral reduction step, where the interaction between the borane-tetrahydrofuran complex and the chiral catalyst dictates the stereochemical outcome of the reaction. The catalyst, (1R,2S)-1-amino-2-indanol, acts as a Lewis base that coordinates with the electron-deficient boron atom, creating a rigid chiral environment around the reducing agent. When the prochiral ketone substrate, 2-chloro-3'-hydroxyacetophenone, enters this coordination sphere, the steric bulk of the indanol framework forces the hydride transfer to occur from a specific face of the carbonyl group, thus ensuring the formation of the (R)-alcohol configuration with high enantiomeric excess. This mechanism is particularly advantageous because the catalyst can be generated in situ without isolation, reducing handling steps and exposure to air-sensitive materials, which is a critical factor for maintaining process stability in a commercial setting. The reaction proceeds smoothly at temperatures ranging from 0°C to 30°C, demonstrating remarkable tolerance to thermal variations that might otherwise degrade sensitive intermediates in less optimized processes.
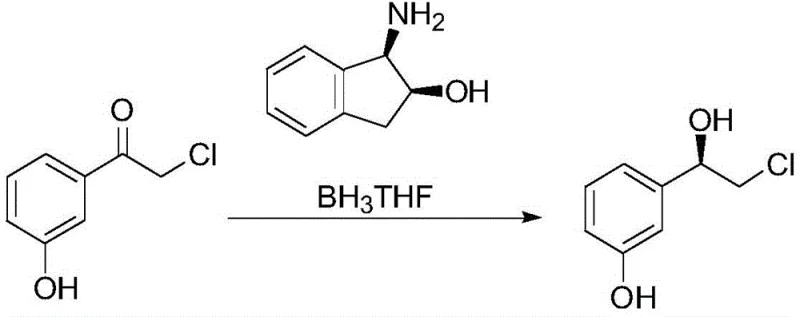
Impurity control within this mechanism is achieved through a combination of kinetic selectivity during the reduction phase and thermodynamic control during the final purification stages. While the crude chiral reduction product may exhibit enantiomeric purities in the range of 80% to 88%, the process design incorporates a powerful recrystallization or chiral resolution step post-amination that elevates the final optical purity to exceed 99.0%. This two-tiered purity assurance strategy ensures that even if minor variations occur in the upstream reduction step, the downstream processing acts as a robust filter to remove off-spec material, guaranteeing that the final L-phenylephrine hydrochloride meets the stringent requirements of major pharmacopoeias. Furthermore, the choice of solvents such as tetrahydrofuran and methanol facilitates easy separation of by-products and unreacted starting materials, preventing the accumulation of genotoxic impurities or heavy metal residues that are often associated with transition metal-catalyzed reactions.
How to Synthesize L-Phenylephrine Hydrochloride Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and stereochemical integrity, beginning with the careful addition of chlorinating agents to maintain regioselectivity. The standardized protocol involves dissolving the acetophenone precursor in an inert solvent like dichloromethane and maintaining the temperature between 0°C and 10°C during the dropwise addition of sulfuryl chloride to prevent over-chlorination or side reactions. Following the isolation of the chlorinated ketone, the critical chiral reduction is performed by pre-mixing the amino-indanol ligand with borane-THF before introducing the substrate, a sequence that ensures the active catalytic species is fully formed prior to the onset of reduction. Detailed standardized synthesis steps see the guide below.
- Perform chlorination of acetophenone using sulfuryl chloride in an inert solvent to obtain 2-chloro-3'-hydroxyacetophenone.
- Conduct a chiral reduction reaction on the chlorinated intermediate using a catalyst system of (1R,2S)-1-amino-2-indanol and borane-THF.
- Execute methylamination followed by salification with hydrochloric acid and recrystallization to achieve final pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this manufacturing route offers profound advantages by fundamentally altering the cost structure and risk profile associated with producing L-phenylephrine hydrochloride. The elimination of expensive noble metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth elements, leading to a more stable and predictable pricing model for long-term contracts. Moreover, the avoidance of high-pressure hydrogenation equipment reduces the barrier to entry for contract manufacturing organizations, allowing for a broader base of qualified suppliers who possess standard glass-lined or stainless steel reactors rather than specialized autoclaves. This accessibility translates directly into enhanced supply chain resilience, as production can be easily shifted between different facilities without the need for extensive retrofitting or safety recertification processes that are mandatory for high-pressure operations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly chiral metal catalysts with inexpensive organic amino-alcohols and commodity borane reagents. By removing the necessity for precious metals, manufacturers avoid the complex and expensive downstream processing steps required to reduce metal residues to parts-per-million levels, which significantly lowers the operational expenditure per kilogram of finished product. Additionally, the high atom economy of the chlorination and reduction steps minimizes raw material waste, further contributing to substantial cost savings in large-scale production runs where material efficiency is a key driver of profitability.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as acetophenone, sulfuryl chloride, and methylamine ensures that the supply chain is not vulnerable to the bottlenecks often seen with specialized chiral ligands or high-purity hydrogen gas. Since the reagents are commodity chemicals produced by multiple global vendors, procurement teams can diversify their sourcing strategies to prevent single-point failures, ensuring continuous availability of critical inputs even during market disruptions. This robustness is further supported by the mild reaction conditions, which reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents, thereby guaranteeing consistent delivery schedules for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at near-ambient pressures and moderate temperatures, allowing for seamless technology transfer from pilot plants to multi-ton commercial reactors without encountering the heat and mass transfer limitations typical of exothermic high-pressure reactions. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and waste disposal protocols, reducing the regulatory burden and associated costs of environmental compliance. The use of common organic solvents that can be readily recovered and recycled further enhances the sustainability profile of the manufacturing process, aligning with the increasing corporate mandates for greener supply chains and reduced carbon footprints in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of L-phenylephrine hydrochloride using this advanced chiral reduction methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's viability. Understanding these details is crucial for technical teams assessing the feasibility of integrating this intermediate into their own drug substance manufacturing workflows.
Q: What is the key chiral catalyst used in this synthesis?
A: The process utilizes (1R,2S)-1-amino-2-indanol complexed with borane-tetrahydrofuran, which avoids the need for expensive noble metal catalysts typically required in asymmetric hydrogenation.
Q: How is the optical purity of the final product ensured?
A: Optical purity is controlled through the stereoselective chiral reduction step and further enhanced by recrystallization or chiral resolution using D-tartaric acid in the final purification stage.
Q: Why is this method superior to traditional racemate resolution?
A: Unlike racemate resolution which suffers from a maximum theoretical yield of 50% and complex mother liquor treatment, this asymmetric synthesis offers higher yields and simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Phenylephrine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN111689869A to ensure the consistent supply of high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art chiral HPLC and NMR instrumentation to verify that every batch of L-phenylephrine hydrochloride meets or exceeds the requirements of the Chinese and European Pharmacopoeias. Our commitment to quality assurance means that we do not just supply chemicals; we provide validated solutions that de-risk your drug development pipeline.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your bottom line while securing your supply chain against market volatility. Contact us today to discuss how our expertise in chiral synthesis can support your next project milestone.
