Advanced Synthesis of Sugammadex Sodium Dimer Impurity for Enhanced Quality Control
Advanced Synthesis of Sugammadex Sodium Dimer Impurity for Enhanced Quality Control
The pharmaceutical industry continuously demands higher purity standards for Active Pharmaceutical Ingredients (APIs), particularly for complex molecules like Sugammadex Sodium, a selective relaxant binding agent used to reverse neuromuscular blockade. Patent CN111471121A introduces a groundbreaking synthetic methodology specifically designed to produce the Sugammadex Sodium dimer impurity, a critical related substance that must be monitored to ensure patient safety. This innovation addresses a significant gap in the market where no reliable synthetic route previously existed for this specific dimeric by-product. By establishing a controlled chemical pathway to generate this impurity, manufacturers can now produce authentic reference standards essential for validating analytical methods. This capability is paramount for R&D teams aiming to characterize impurity profiles accurately and for quality control departments tasked with releasing batches that meet rigorous global regulatory compliance. The ability to synthesize this complex dimer on demand transforms how pharmaceutical companies approach the safety assessment of Sugammadex Sodium formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of patent CN111471121A, the pharmaceutical sector faced a substantial challenge regarding the availability of reference standards for Sugammadex Sodium impurities. The background art indicates that while the main API synthesis was established, the specific dimer impurity formed as a side reaction lacked a dedicated synthetic protocol. Traditionally, obtaining such impurities relied on isolating trace amounts from crude reaction mixtures, a process that is notoriously inefficient, costly, and yields insufficient quantities for comprehensive toxicological or analytical studies. Without a pure reference standard, the accuracy of qualitative and quantitative analysis in routine quality control is compromised, potentially leading to false negatives or inaccurate potency assessments. This uncertainty poses a clinical risk, as undetected or misquantified impurities could impact the safety profile of the final injectable medication. Furthermore, the inability to synthesize the impurity independently hinders the development of robust stability-indicating methods, leaving manufacturers vulnerable to regulatory scrutiny during audits.
The Novel Approach
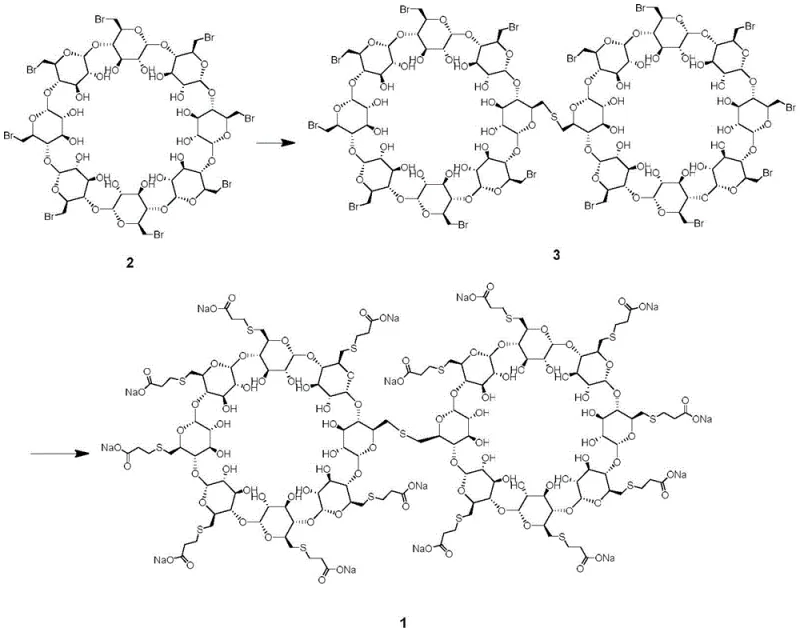
The patented methodology offers a decisive solution by outlining a rational, two-step synthetic route that constructs the dimer impurity from well-defined starting materials. Instead of relying on chance formation during API synthesis, this approach intentionally links two cyclodextrin units using a sulfide bridge before functionalizing the periphery with carboxyethyl thio groups. This strategic inversion of the synthesis logic ensures that the dimeric core is formed first under controlled conditions, maximizing the yield of the desired structural motif. The process utilizes octa-(6-bromo-6-deoxy)-γ-cyclodextrin as a key building block, reacting it with a sulfide source to create the intermolecular thioether linkage. Subsequently, the remaining reactive sites are capped with 3-mercaptopropionic acid to mimic the final API structure. This deliberate construction not only guarantees the structural identity of the impurity but also allows for scalable production, enabling manufacturers to generate gram-to-kilogram quantities of the reference standard as needed for extensive validation campaigns.
Mechanistic Insights into Sulfide-Mediated Dimerization and Thiol Alkylation
The core of this synthetic strategy lies in the precise control of nucleophilic substitution reactions on the cyclodextrin scaffold. In the first stage, the primary bromides at the C6 positions of the gamma-cyclodextrin ring act as excellent leaving groups. When treated with a sulfide source, such as sodium sulfide nonahydrate, in a polar aprotic solvent like DMF, a nucleophilic attack occurs. Crucially, the reaction conditions—specifically the molar ratio of sulfide to cyclodextrin and the temperature range of -5 to 45°C—are tuned to favor intermolecular coupling over intramolecular cyclization or polymerization. This results in the formation of Intermediate 3, where two cyclodextrin macrocycles are covalently linked via a sulfur atom. This step is mechanistically distinct from the standard API synthesis, which aims for mono-substitution on every glucose unit; here, the goal is dimerization, requiring careful stoichiometric management to prevent the formation of higher-order oligomers or unreacted monomers.
Following the formation of the dimeric core, the second stage involves the conversion of the remaining bromide functionalities into the characteristic 2-carboxyethyl thio groups found in Sugammadex Sodium. This is achieved by reacting Intermediate 3 with 3-mercaptopropionic acid in an alkaline aqueous environment. The base deprotonates the thiol group of the mercaptoacid, generating a highly nucleophilic thiolate anion. This anion then displaces the remaining bromide atoms on the dimeric scaffold through an SN2 mechanism. The use of a co-solvent system, typically involving DMSO and water, ensures that both the hydrophobic cyclodextrin intermediate and the hydrophilic thiolate species remain in solution, facilitating efficient mass transfer and reaction completion. The final product is obtained as the sodium salt after neutralization and crystallization, yielding a material that is structurally identical to the process-related impurity found in commercial Sugammadex Sodium batches, thereby serving as a perfect match for chromatographic comparison.
How to Synthesize Sugammadex Sodium Dimer Impurity Efficiently
The synthesis of this high-value reference standard is streamlined into two distinct operational phases that can be easily integrated into existing pilot plant workflows. The initial phase focuses on the construction of the sulfur-bridged dimer backbone, requiring precise temperature control and stoichiometry to maximize the formation of the intermolecular species. The subsequent phase involves the exhaustive functionalization of the dimer with the carboxylic acid side chains, a robust reaction that proceeds efficiently under mild alkaline conditions. Detailed standard operating procedures for reagent preparation, reaction monitoring, and purification protocols are essential to ensure batch-to-batch consistency. For a comprehensive guide on the specific molar ratios, solvent volumes, and workup procedures described in the patent, please refer to the standardized synthesis steps outlined below.
- React octa-(6-bromo-6-deoxy)-γ-cyclodextrin with a sulfide source (e.g., sodium sulfide) in a polar solvent like DMF at mild temperatures to form the intermolecular thioether intermediate.
- Treat the resulting intermediate with 3-mercaptopropionic acid in an aqueous sodium hydroxide solution to substitute remaining bromides and form the final carboxylated dimer impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of reference standard production from the main API manufacturing line. Previously, the scarcity of the dimer impurity meant that quality control labs were dependent on unpredictable isolation yields or expensive external sourcing, creating bottlenecks in batch release schedules. By adopting this dedicated synthesis, organizations can secure a stable, internal supply of critical reference materials, thereby insulating their quality assurance operations from external market volatility. This autonomy significantly enhances supply chain resilience, ensuring that analytical testing can proceed without interruption regardless of API production volumes. Furthermore, the use of commodity chemicals such as sodium sulfide and 3-mercaptopropionic acid keeps raw material costs predictable and low, avoiding the need for exotic or proprietary reagents that often carry high price tags and long lead times.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the elimination of complex isolation procedures. Traditional methods of obtaining impurities often involve processing tons of crude API mixture to isolate milligrams of the target compound, a process that consumes vast amounts of solvents, energy, and labor. In contrast, this directed synthesis builds the impurity directly from inexpensive starting materials with high atom economy relative to the target mass. By removing the need for preparative HPLC or extensive chromatographic purification typically associated with isolation, the overall cost of goods for the reference standard is drastically reduced. Additionally, the mild reaction conditions minimize energy consumption for heating or cooling, further contributing to operational expenditure savings. This cost efficiency allows quality control budgets to be allocated more effectively across other critical testing parameters.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for pharmaceutical manufacturers, especially when dealing with regulated materials. This synthetic route relies on widely available bulk chemicals that are not subject to the same supply constraints as specialized intermediates. The robustness of the reaction conditions, which tolerate a broad temperature range and utilize common polar solvents like DMF and DMSO, means that production can be scaled up or down rapidly in response to demand fluctuations. There is no reliance on single-source suppliers for unique catalysts or reagents, mitigating the risk of supply disruptions. Consequently, procurement teams can negotiate better terms with vendors for standard chemicals and maintain leaner inventory levels of the finished reference standard, knowing that replenishment cycles are short and reliable. This agility is crucial for maintaining just-in-time manufacturing models in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed for straightforward translation from laboratory to commercial scale. The reactions are homogeneous and do not require specialized high-pressure equipment or cryogenic conditions, simplifying the engineering requirements for scale-up. The use of aqueous workups and standard crystallization techniques facilitates the recovery and recycling of solvents, aligning with green chemistry principles and reducing waste disposal costs. The absence of heavy metal catalysts eliminates the need for expensive and time-consuming metal scavenging steps, which are often a regulatory hurdle in API production. This streamlined workflow not only accelerates the time-to-market for new reference standards but also ensures that the manufacturing process remains compliant with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of the Sugammadex Sodium dimer impurity. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on reaction mechanisms and practical implementation. Understanding these details is crucial for technical teams evaluating the feasibility of in-house production versus outsourcing. The answers provided reflect the current state of the art as defined by the patented methodology, ensuring that decision-makers have access to accurate and actionable information.
Q: Why is the synthesis of Sugammadex Sodium dimer impurity critical for API manufacturing?
A: The dimer impurity is a potential by-product during the main API synthesis. Having a certified reference standard allows for accurate qualitative and quantitative analysis, ensuring the final drug product meets stringent safety and purity specifications required by regulatory bodies.
Q: What are the key reaction conditions for the dimerization step?
A: The process utilizes octa-(6-bromo-6-deoxy)-γ-cyclodextrin reacting with a sulfide source such as sodium sulfide nonahydrate. The reaction is conducted in a polar solvent like DMF at controlled temperatures between -5°C and 45°C, preferably around 20-30°C, to ensure selective intermolecular linkage.
Q: How does this patented method improve supply chain reliability for reference standards?
A: Previously, there were no reported synthetic methods for this specific impurity, forcing reliance on isolation or non-existent external supplies. This robust, two-step chemical synthesis enables on-demand production of high-purity reference materials, significantly reducing lead times and dependency on scarce natural sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Dimer Impurity Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality reference standards play in the development and commercialization of safe pharmaceutical products. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex molecules like the Sugammadex Sodium dimer impurity can be manufactured with precision and consistency. We are equipped with state-of-the-art rigorous QC labs capable of verifying structural integrity and purity against the highest international standards. Our commitment to excellence means that every batch of reference material we supply is accompanied by comprehensive analytical data, giving your R&D and quality assurance teams the confidence they need to move forward with their projects. We view ourselves not just as a supplier, but as a strategic partner dedicated to supporting your regulatory success.
We invite you to leverage our technical expertise to optimize your supply chain for critical impurities. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our manufacturing capabilities can meet your exacting specifications. Let us help you secure a reliable source for your Sugammadex Sodium impurity needs, ensuring uninterrupted progress in your drug development and quality control programs.
