Optimized Industrial Synthesis of Cefuroxime Acid via Selective Hydrolysis and Active Ester Coupling
Optimized Industrial Synthesis of Cefuroxime Acid via Selective Hydrolysis and Active Ester Coupling
The pharmaceutical industry continuously demands more efficient and environmentally sustainable pathways for producing critical antibiotic intermediates. Patent CN102093390A introduces a robust methodology for the preparation of cefuroxime acid, a pivotal precursor for second-generation cephalosporins like cefuroxime axetil and cefuroxime sodium. This technical disclosure outlines a streamlined three-step sequence that begins with the selective hydrolysis of 7-aminocephalosporanic acid (7-ACA) to generate 3-deacetylation-7-aminocephalosporanic acid (7-DACA). By leveraging low-temperature inorganic base hydrolysis followed by active ester condensation and final carbamoylation, this process addresses longstanding challenges in purity and yield. For global procurement teams and R&D directors, understanding this specific synthetic architecture is essential for securing a reliable cefuroxime acid supplier capable of meeting stringent pharmacopeial standards while optimizing manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefuroxime acid has been plagued by inefficiencies inherent in traditional acylation strategies. One prevalent conventional route involves the use of methoxyimino furan acetyl ammonium salts converted into acyl chlorides prior to N-acylation with 7-ACA. This approach necessitates strongly basic environments for subsequent hydrolysis of the 3-acetoxy group, which often leads to significant impurity profiles and lower overall purity that struggles to meet medicinal requirements. Furthermore, alternative pathways that attempt to modify the 3-hydroxymethyl group before C7-amino acylation frequently suffer from competitive side reactions where isocyanates react indiscriminately with the 7-amino group. These competing reactions drastically reduce the yield of the desired intermediate, creating bottlenecks in production capacity and inflating the cost of goods sold due to the need for complex downstream purification processes to remove structural analogs.
The Novel Approach
The innovative strategy detailed in the patent data circumvents these pitfalls by reversing the order of functional group modifications and employing milder coupling reagents. Instead of relying on reactive acyl chlorides, the novel approach utilizes 2-(2-furyl)-2-(methoxyimino)acetic acid-(2,5-dioxo-pyrrolidyl)-1-ester, an active ester that facilitates highly selective condensation with the 7-amino group of 7-DACA. This sequence ensures that the sensitive beta-lactam core remains intact while the 3-position is selectively deacetylated under controlled cryogenic conditions using inorganic bases. By prioritizing the formation of the C7-amide bond before modifying the 3-hydroxymethyl group with chlorosulfonyl isocyanate, the process eliminates the risk of unwanted urea formation or polymerization. This logical reordering of synthetic steps results in a cleaner reaction profile, simplified isolation procedures, and a final product with superior specific optical rotation and melting point characteristics indicative of high stereochemical purity.
Mechanistic Insights into Selective Hydrolysis and Active Ester Coupling
The cornerstone of this synthesis lies in the precise control of reaction kinetics during the initial hydrolysis phase. The conversion of 7-ACA to 7-DACA is achieved by dissolving the starting material in a mixed solvent system of water and lower alcohols such as methanol or ethanol, then cooling the mixture to a range of -15 to 0°C. At this temperature, the addition of an aqueous alkaline solution, typically sodium hydroxide, allows for the nucleophilic attack on the 3-acetoxyl ester linkage while minimizing the hydrolysis of the strained four-membered beta-lactam ring. Maintaining the pH between 11 and 12 is critical; deviations can lead to ring opening or incomplete deacetylation. Following this, the condensation step employs triethylamine as a base to scavenge protons during the nucleophilic attack of the 7-amino group on the active ester carbonyl. This mechanism proceeds smoothly at ice-water bath temperatures (0 to 5°C), ensuring that the chiral centers at positions 6 and 7 remain undisturbed, thereby preserving the biological activity of the final cephalosporin scaffold.
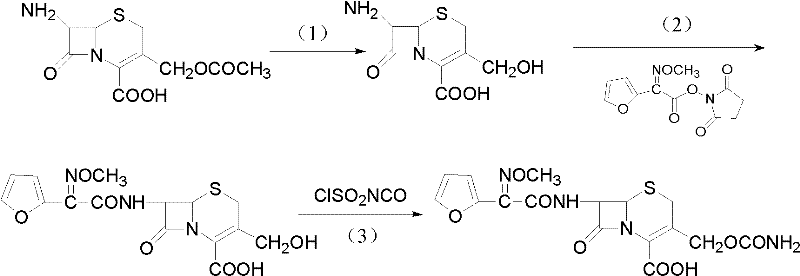
Following the formation of the intermediate 3-decarbamyl-cefuroxime acid (DCCF), the final transformation involves the introduction of the carbamoyloxymethyl side chain. This is accomplished by reacting DCCF with chlorosulfonyl isocyanate in tetrahydrofuran at low temperatures ranging from -20 to 0°C. The mechanism involves the formation of an unstable intermediate which, upon quenching with ice water, rearranges to form the stable carbamate linkage at the 3-position. The use of chlorosulfonyl isocyanate is particularly advantageous because it reacts selectively with the primary alcohol at the 3-position without affecting the newly formed amide bond at the 7-position, provided the reaction temperature is strictly controlled. This selectivity is paramount for preventing the formation of bis-carbamoylated byproducts or degradation of the cephalosporin nucleus, ensuring that the final cefuroxime acid meets the rigorous impurity specifications required for downstream salt formation or esterification into oral prodrugs.
How to Synthesize Cefuroxime Acid Efficiently
The operational protocol derived from this patent offers a clear roadmap for laboratory and pilot-scale execution, emphasizing temperature control and stoichiometric precision. The process begins with the preparation of the 7-DACA intermediate, followed by the coupling reaction to form DCCF, and concludes with the carbamoylation step. Each stage requires careful monitoring via HPLC to ensure complete conversion before proceeding to the next unit operation, particularly to verify the disappearance of the 7-ACA peak and the minimal presence of unreacted 7-DACA. The detailed standardized synthesis steps, including specific solvent ratios, reagent addition rates, and crystallization parameters, are outlined below to guide process engineers in replicating these high-yield results.
- Perform selective hydrolysis of 7-aminocephalosporanic acid (7-ACA) using aqueous alkali at low temperatures (-15 to 0°C) to obtain 3-deacetylation-7-aminocephalosporanic acid (7-DACA).
- Condense 7-DACA with 2-(2-furyl)-2-(methoxyimino)acetic acid-(2,5-dioxo-pyrrolidyl)-1-ester in the presence of triethylamine to form 3-decarbamyl-cefuroxime acid (DCCF).
- Modify the 3-hydroxymethyl group of DCCF using chlorosulfonyl isocyanate at low temperature to finalize the synthesis of cefuroxime acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into enhanced operational efficiency and risk mitigation. The elimination of acyl chloride intermediates removes the need for handling corrosive and hazardous reagents, which simplifies safety protocols and reduces the regulatory burden associated with waste disposal. Furthermore, the high selectivity of the active ester coupling means that less raw material is wasted on side products, effectively lowering the consumption of expensive starting materials like 7-ACA and the furyl-glyoxylate derivative. This efficiency gain is compounded by the simplified workup procedures, which rely on standard pH adjustments and crystallization rather than complex chromatographic separations, thereby reducing cycle times and increasing throughput capacity in existing manufacturing facilities.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive inorganic bases such as sodium hydroxide instead of costly organic catalysts or specialized enzymatic systems. By avoiding the formation of difficult-to-remove impurities, the need for extensive recrystallization or resin treatment is drastically minimized, leading to substantial savings in solvent usage and energy consumption. Additionally, the high yield reported in the embodiments suggests that less feedstock is required per kilogram of final product, directly improving the margin structure for high-purity cefuroxime acid manufacturing and allowing for more competitive pricing in the global API market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like methanol, ethanol, and tetrahydrofuran ensures that the supply chain is not vulnerable to shortages of exotic reagents. The robustness of the low-temperature hydrolysis step allows for consistent batch-to-batch quality, reducing the incidence of out-of-specification results that can disrupt delivery schedules. This stability is crucial for maintaining continuous production lines for downstream derivatives like cefuroxime axetil, ensuring that pharmaceutical customers receive their orders on time without the delays often associated with troubleshooting inconsistent synthetic routes.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in three-waste emissions is a major advantage, as the process generates fewer organic byproducts and utilizes aqueous workups that are easier to treat. The simplicity of the operation makes it highly amenable to commercial scale-up of complex beta-lactam intermediates, allowing manufacturers to transition from pilot batches to multi-ton production with minimal process re-engineering. This scalability ensures that suppliers can rapidly respond to surges in demand for cephalosporin antibiotics without compromising on the environmental standards increasingly demanded by international regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific cefuroxime acid synthesis pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing. Understanding these nuances helps stakeholders make informed decisions about technology transfer and vendor qualification.
Q: What are the advantages of using active ester coupling over acyl chloride methods for cefuroxime acid?
A: The active ester method significantly reduces impurity formation compared to traditional acyl chloride routes. It avoids the harsh conditions associated with acyl chlorides, leading to higher product purity that meets medicinal requirements without extensive purification steps.
Q: How does the low-temperature selective hydrolysis step impact yield?
A: Conducting hydrolysis at -15 to 0°C ensures the selective removal of the 3-acetoxyl group while preserving the integrity of the beta-lactam ring. This precision minimizes degradation byproducts, resulting in consistently high yields suitable for industrial scaling.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for industrial suitability. It utilizes common inorganic bases and solvents, generates less three-waste emission, and involves straightforward operational steps like crystallization and filtration, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate temperature controls and stoichiometric balances required for cefuroxime acid synthesis are maintained with absolute precision. We operate stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and spectroscopic methods to verify that every batch meets the specific optical rotation and melting point criteria essential for therapeutic efficacy. Our commitment to quality assurance means that clients can trust us to deliver intermediates that seamlessly integrate into their final drug product manufacturing processes.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic advantages of switching to this high-yield method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate the potential for reducing lead time for high-purity cephalosporin intermediates within your own production network.
