Advanced Manufacturing of High-Purity Amorphous Ertapenem Intermediates for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibiotic intermediates, particularly for broad-spectrum carbapenems like ertapenem. A pivotal advancement in this domain is documented in patent CN101875665B, which discloses a novel preparation method for an ertapenem intermediate characterized by its amorphous solid form and exceptional purity profiles. This technical breakthrough addresses long-standing challenges in the stabilization and isolation of carbapenem derivatives, offering a pathway that significantly enhances the feasibility of industrial-scale production. By utilizing specific carboxyl-protecting groups and a refined acid-quenching isolation technique, the inventors have achieved a material state that is not only chemically stable but also physically manageable, overcoming the tendency of previous iterations to form intractable oils or impure solids. For global procurement and R&D teams, understanding the nuances of this patented methodology is essential for securing a reliable supply of high-quality antibiotic precursors.
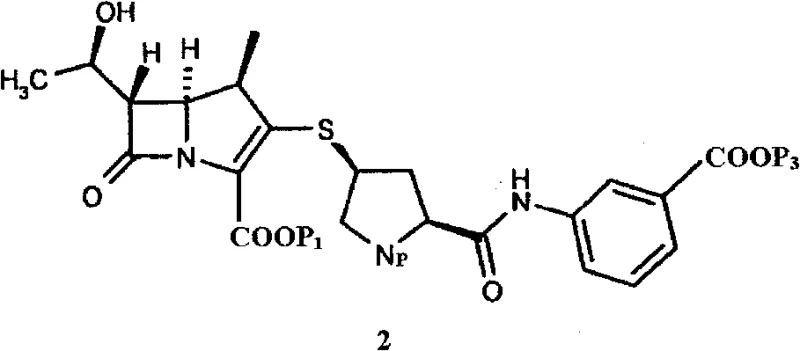
Furthermore, the strategic importance of this intermediate lies in its role as a direct precursor to the final active pharmaceutical ingredient, meaning that any variability in its quality directly impacts the efficacy and safety of the final drug product. The patent details a composition comprising at least 95% of the target intermediate, with preferred embodiments reaching purity levels that minimize downstream purification burdens. This level of control over the impurity profile is critical for regulatory compliance in major markets such as the US and EU. As a result, this technology represents a significant value proposition for manufacturers aiming to optimize their supply chains for carbapenem antibiotics, ensuring consistency and reliability in the face of increasing global demand for effective antimicrobial therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing ertapenem intermediates, such as those disclosed in WO2008062279, frequently encountered severe practical limitations that hindered efficient commercial manufacturing. A primary drawback was the difficulty in isolating the product in a solid form; many conventional processes resulted in the formation of viscous oils or gums that were extremely challenging to handle, filter, and dry on a large scale. This physical state not only complicated the workflow but also trapped impurities, leading to products with purity levels often falling below 90%, which is unacceptable for GMP pharmaceutical production. Additionally, traditional workup procedures often relied heavily on organic solvents for extraction and precipitation, introducing significant environmental hazards and increasing the overall cost of goods sold due to solvent recovery and disposal requirements.
Moreover, the introduction of inorganic salts in certain prior art methods to facilitate crystallization created further downstream processing issues, as these salts were difficult to remove completely and could interfere with subsequent deprotection reactions. The lack of a defined solid state also made structural confirmation via standard analytical techniques like powder X-ray diffraction nearly impossible, creating uncertainty in quality control protocols. These cumulative inefficiencies resulted in extended lead times, higher production costs, and a greater risk of batch failure, posing substantial risks to supply chain continuity for pharmaceutical companies relying on these legacy synthetic routes for their antibiotic portfolios.
The Novel Approach
In stark contrast, the novel approach outlined in CN101875665B introduces a streamlined process that reliably yields the ertapenem intermediate as a free-flowing amorphous solid with high purity. By optimizing the condensation reaction conditions and, crucially, modifying the isolation step to involve pouring the reaction mixture directly into a controlled aqueous acid solution, the inventors have bypassed the formation of oils entirely. This method eliminates the need for extensive organic solvent extraction, thereby reducing the environmental footprint and simplifying the operational complexity of the manufacturing process. The resulting solid exhibits excellent free-running properties, making it easy to store, transport, and feed into subsequent reaction steps without the need for complex milling or conditioning.
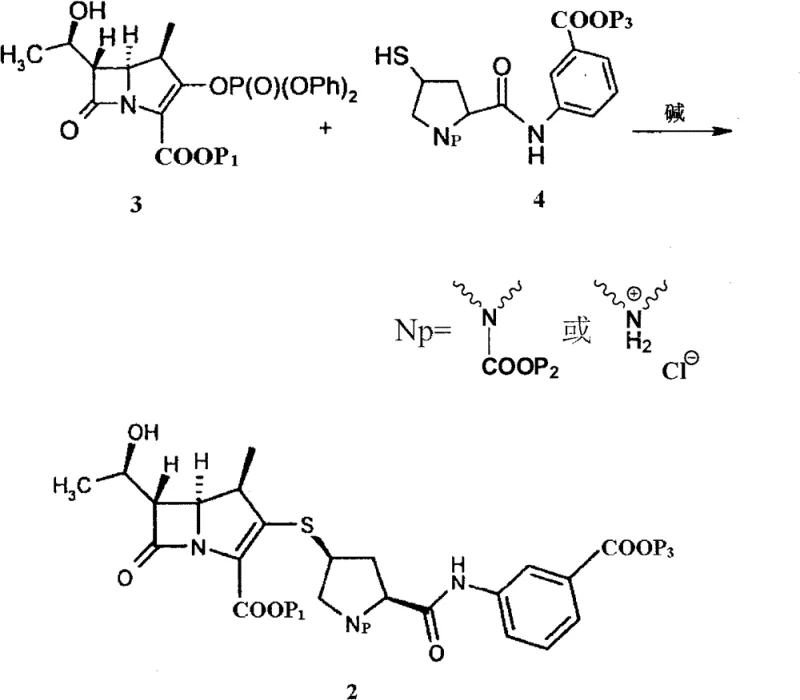
The versatility of this new method is further demonstrated by its compatibility with a wide range of carboxyl-protecting groups, including p-nitrobenzyl, allyl, and various silyl groups, allowing manufacturers to tailor the synthesis to their specific downstream deprotection capabilities. The ability to consistently achieve HPLC purity levels exceeding 98% significantly reduces the burden on purification teams and ensures that the intermediate meets stringent regulatory specifications right out of the reactor. This shift from unpredictable oily residues to a well-defined amorphous solid represents a paradigm shift in carbapenem intermediate manufacturing, offering a robust solution that aligns perfectly with the needs of modern, efficiency-driven pharmaceutical supply chains seeking cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Condensation and Acid Quenching
The core chemical transformation involves the nucleophilic attack of the thiol group on the ertapenem side chain onto the activated phosphate ester of the carbapenem nucleus. This condensation reaction is highly sensitive to reaction conditions, particularly temperature and base strength, requiring precise control to prevent epimerization at the chiral centers of the carbapenem ring. The patent specifies the use of bases like diisopropylethylamine at low temperatures, typically around -35°C, to maintain the stereochemical integrity of the molecule. The choice of protecting groups on the nitrogen and carboxyl moieties plays a critical role in modulating the electronic properties of the reactants, ensuring that the nucleophilicity of the thiol is sufficient to drive the reaction to completion without promoting side reactions that could generate difficult-to-remove impurities.
Following the condensation, the critical innovation lies in the quenching mechanism. Instead of a standard aqueous workup, the reaction mixture is introduced into an aqueous acid solution with a specific pH range, typically between 2 and 5. This acidic environment protonates the basic species and facilitates the precipitation of the product in its free acid form. The rapid change in solubility parameters upon mixing with the aqueous acid prevents the molecules from organizing into a crystalline lattice, instead trapping them in a high-energy amorphous state. This amorphous form is thermodynamically stable enough for storage but lacks the rigid crystal packing that often traps solvent molecules or impurities. This mechanistic understanding allows process chemists to fine-tune the acid concentration and addition rate to maximize yield and purity, ensuring a consistent product quality that is vital for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Ertapenem Intermediate Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and quenching conditions to ensure the formation of the desired amorphous solid. Operators must ensure that the carbapenem nucleus and side chain are fully dissolved and reacted before initiating the precipitation step, as incomplete reaction can lead to contamination with starting materials that are difficult to separate later. The detailed standardized synthesis steps see the guide below, which outlines the precise sequence of addition and the critical control points necessary to replicate the high yields and purity reported in the patent data.
- Dissolve carbapenem nucleus and ertapenem side chain in DMF with base at low temperature.
- Monitor the condensation reaction progress to ensure complete conversion of starting materials.
- Quench the reaction mixture into aqueous acid solution to precipitate the amorphous solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers profound strategic advantages that extend beyond simple chemical yield. The elimination of complex organic solvent extraction steps translates directly into a significantly reduced operational footprint, lowering the costs associated with solvent purchase, recovery, and hazardous waste disposal. Furthermore, the generation of a solid product with good flow properties drastically simplifies logistics and storage, reducing the risk of degradation during transit compared to unstable oils or solutions. This robustness enhances supply chain reliability, ensuring that critical raw materials are available when needed without the delays often caused by difficult purification processes or batch failures associated with older technologies.
- Cost Reduction in Manufacturing: The process design inherently lowers manufacturing costs by removing the need for expensive chromatographic purification steps that were previously required to upgrade the purity of oily residues. By achieving high purity directly through crystallization-like precipitation, the consumption of silica gel and eluents is virtually eliminated, leading to substantial cost savings in consumables. Additionally, the higher yield reduces the amount of expensive starting materials required per kilogram of final product, optimizing the overall material cost structure and improving the margin profile for the final API manufacturer.
- Enhanced Supply Chain Reliability: The simplicity and robustness of the acid quenching method make the process less susceptible to variations in raw material quality or minor operational deviations, resulting in more consistent batch-to-batch performance. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery commitments to downstream API manufacturers. The ability to source key starting materials that are commercially available and the use of standard reaction equipment further mitigate supply risks, ensuring a stable and uninterrupted flow of intermediates even in volatile market conditions.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in organic solvent usage aligns with increasingly stringent global regulations on volatile organic compound emissions and waste generation. The process is inherently greener, facilitating easier regulatory approval and reducing the environmental compliance burden on manufacturing sites. Moreover, the scalability of the precipitation step is excellent, as it does not rely on slow crystallization kinetics that can be problematic in large reactors, allowing for rapid throughput and efficient utilization of production assets for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific ertapenem intermediate. These answers are derived directly from the technical disclosures and experimental data provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for integration into their supply chains. Understanding these details is key to assessing the feasibility and benefits of adopting this advanced synthetic route.
Q: What distinguishes the amorphous form of this ertapenem intermediate from crystalline forms?
A: The amorphous form described in CN101875665B offers superior free-running properties and higher solid purity compared to prior art methods which often resulted in oils or low-purity solids difficult to handle.
Q: How does the acid quenching method improve post-treatment efficiency?
A: By pouring the reaction liquid directly into an aqueous acid solution, the process avoids the use of organic solvents for extraction, significantly simplifying the workup and reducing environmental waste.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the method utilizes common reagents and avoids complex chromatographic purification, making it highly suitable for large-scale commercial manufacturing of carbapenem intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial infections. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot studies to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of ertapenem intermediate we produce meets the highest international standards for safety and efficacy. We are committed to being a reliable Ertapenem Intermediate Supplier who understands the complexities of carbapenem chemistry.
We invite you to collaborate with us to leverage this advanced technology for your own production needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall time to market for critical antibiotic therapies.
