Advanced Manufacturing of Ceftizoxime Sodium: Overcoming Impurity Challenges in Beta-Lactam Production
The pharmaceutical landscape for third-generation cephalosporins is constantly evolving, driven by the critical need for higher purity standards and more sustainable manufacturing processes. Patent CN102807573B introduces a transformative methodology for the preparation of Ceftizoxime Sodium, a broad-spectrum antibiotic essential for treating severe bacterial infections ranging from lower respiratory tract issues to septicemia. This technical disclosure addresses long-standing inefficiencies in beta-lactam synthesis, specifically targeting the persistent issue of toxic residual impurities that plague conventional production lines. By shifting from traditional thioester activation to a sophisticated DCC/DMAP-catalyzed active ester pathway, this innovation offers a robust solution for manufacturers seeking to enhance product quality while streamlining operational complexity. The structural integrity of the final molecule is paramount for its efficacy against gram-negative bacteria, and this new route ensures that the delicate beta-lactam ring remains intact throughout the rigorous synthetic sequence.
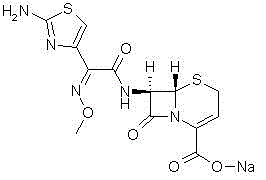
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ceftizoxime Sodium has relied heavily on condensation reactions involving activated esters derived from benzothiazole thioesters. While these methods established the foundation for mass production, they suffer from significant chemical drawbacks that impact both product quality and environmental compliance. The primary deficiency lies in the generation of 2-mercaptobenzothiazole, commonly known as captax, as a stoichiometric byproduct during the coupling phase. This sulfur-containing impurity is not only toxic but also notoriously difficult to remove completely from the final API matrix, often necessitating energy-intensive purification steps such as repeated recrystallization or column chromatography. Furthermore, earlier patents, such as U.S. Pat 4427674, disclosed routes that resulted in numerous side reactions, leading to lower overall yields and a complex impurity profile that complicates regulatory approval. The reliance on these legacy chemistries creates a bottleneck for procurement teams, as the additional processing time extends lead times and increases the cost of goods sold due to solvent consumption and waste disposal requirements.
The Novel Approach
In stark contrast to the problematic thioester pathways, the methodology outlined in CN102807573B employs a highly selective active ester formed via the reaction of 2-(2-amino-4-thiazolyl)-2-methoxyimino acetic acid with N-succinimide. This strategic shift in activation chemistry fundamentally alters the reaction landscape, eliminating the source of sulfur-based contaminants at the molecular level. The use of dicyclohexyl carbodiimide (DCC) and dimethylaminopyridine (DMAP) as a catalytic system facilitates a mild yet potent coupling environment that preserves the stereochemical integrity of the 7-ANCA nucleus. By avoiding the formation of captax, the downstream processing is drastically simplified, removing the need for aggressive purification protocols that often degrade sensitive beta-lactam structures. This approach not only enhances the safety profile of the manufacturing process but also aligns with modern green chemistry principles by reducing the load of hazardous waste. The result is a streamlined workflow that delivers superior economic value through higher throughput and reduced operational overhead.
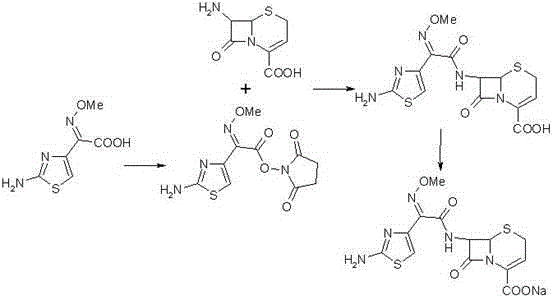
Mechanistic Insights into DCC/DMAP-Catalyzed Active Ester Condensation
The core of this technological advancement lies in the precise mechanistic execution of the active ester formation and subsequent nucleophilic attack. In the first stage, the carboxylic acid group of the side chain precursor is activated by DCC to form an O-acylisourea intermediate, which is then rapidly converted into the more stable and less racemization-prone N-succinimidyl ester in the presence of DMAP. This specific activation mode is crucial because it generates an electrophile that is sufficiently reactive to couple with the sterically hindered amino group of 7-ANCA without requiring harsh thermal conditions that could open the beta-lactam ring. The nucleophilic attack by the 7-amino group of the cephem nucleus on the carbonyl carbon of the active ester proceeds with high fidelity, driven by the excellent leaving group ability of the succinimide moiety. This mechanism ensures that the Z-configuration of the methoxyimino group is preserved, which is essential for the biological activity of the final antibiotic against beta-lactamase-producing organisms.
Impurity control is inherently built into this reaction design through the exclusion of sulfur-based leaving groups. In traditional thioester methods, the cleavage of the carbon-sulfur bond releases free thiols that can oxidize or react further to create colored impurities and difficult-to-separate byproducts. By utilizing an oxygen-based leaving group (succinimide), the reaction byproducts are neutral and easily separable organic compounds that do not interact with the product matrix. Additionally, the use of triethylamine as a base in the condensation step provides a buffered environment that prevents acid-catalyzed degradation of the cephalosporin core. The final salification step using sodium bicarbonate is equally critical, as it ensures the formation of the stable sodium salt without exposing the molecule to strong alkaline conditions that could cause hydrolysis. This meticulous control over pH and reagent selection results in a final product with purity levels exceeding 99.1%, as verified by HPLC analysis, meeting the stringent specifications required for injectable formulations.
How to Synthesize Ceftizoxime Sodium Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize the efficiency of the active ester formation. The process begins with the dissolution of the acid precursor in an alcoholic solvent, preferably ethanol, which serves as an ideal medium for the DCC/DMAP coupling due to its polarity and solubility profile. Following the formation of the active ester, the reaction mixture is transferred to a dichloromethane system for the coupling with 7-ANCA, where temperature control is vital to prevent exothermic runaway. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and crystallization parameters are essential for reproducing the high yields reported in the patent data. For a comprehensive breakdown of the specific operational parameters and safety guidelines required for scale-up, please refer to the technical guide below.
- Dissolve 2-(2-amino-4-thiazolyl)-2-methoxy-imine acetic acid in ethanol and react with N-succinimide using DCC/DMAP catalysis to form the active ester.
- Condense the active ester with 7-ANCA in dichloromethane using triethylamine as a base to generate Ceftizoxime acid.
- React the Ceftizoxime acid with a salifying agent such as sodium bicarbonate to obtain the final Ceftizoxime Sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into tangible operational efficiencies and risk mitigation. The elimination of toxic sulfur byproducts removes a significant regulatory and environmental burden, simplifying the waste treatment process and reducing the associated costs of hazardous material disposal. This cleaner process profile enhances supply chain reliability by minimizing the risk of production stoppages due to environmental compliance issues or equipment corrosion caused by aggressive sulfur compounds. Furthermore, the high yield and purity achieved reduce the need for reprocessing batches, leading to a more predictable production schedule and consistent inventory availability. By optimizing the raw material utilization and reducing solvent consumption through streamlined purification, manufacturers can achieve substantial cost savings without compromising on the quality standards demanded by global pharmaceutical regulators.
- Cost Reduction in Manufacturing: The transition to an active ester method eliminates the need for expensive and complex purification steps such as column chromatography, which are often required in older synthetic routes to remove stubborn impurities. By avoiding the use of benzothiazole thioesters, the process removes the cost associated with sourcing specialized sulfur reagents and managing their toxic waste streams. The high conversion efficiency of the DCC/DMAP system ensures that raw materials are utilized effectively, minimizing waste and maximizing the output per batch. These factors collectively contribute to a significantly optimized cost structure, allowing for more competitive pricing in the global API market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The robustness of this chemical pathway ensures a stable and continuous supply of Ceftizoxime Sodium, which is critical for meeting the demands of the healthcare sector. The use of readily available reagents like N-succinimide and common solvents reduces dependency on niche suppliers, thereby mitigating the risk of raw material shortages. The simplified downstream processing shortens the overall manufacturing cycle time, enabling faster turnaround from production to delivery. This agility allows supply chain managers to respond more effectively to market fluctuations and urgent procurement requests, ensuring that critical antibiotic medications remain available for patients without interruption.
- Scalability and Environmental Compliance: From an environmental perspective, this method represents a significant advancement in green chemistry within the pharmaceutical sector. The absence of heavy metal catalysts and toxic sulfur residues simplifies the effluent treatment process, making it easier to meet strict environmental discharge standards. The process is inherently scalable, as the reaction conditions are mild and do not require specialized high-pressure or high-temperature equipment. This scalability ensures that production can be ramped up to meet commercial volumes without encountering the technical bottlenecks often associated with scaling complex organic syntheses. Consequently, this supports a sustainable manufacturing model that aligns with corporate social responsibility goals and long-term regulatory trends.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of Ceftizoxime Sodium using this advanced methodology. These insights are derived directly from the patent specifications and are intended to provide clarity for technical evaluators and procurement specialists. Understanding these details is crucial for assessing the feasibility of integrating this supply source into your existing vendor portfolio. The answers reflect the specific advantages of the active ester route over traditional methods, highlighting the improvements in purity and process safety.
Q: How does the new active ester method improve purity compared to traditional thioester routes?
A: The novel method utilizes an N-succinimide active ester instead of a benzothiazole thioester. This fundamentally eliminates the generation of 2-mercaptobenzothiazole (captax), a toxic and difficult-to-remove impurity common in prior art, thereby significantly simplifying downstream purification and ensuring higher final product safety.
Q: What are the typical yield improvements observed with this DCC/DMAP catalytic system?
A: Experimental data indicates that the condensation of 7-ANCA with the novel active ester achieves yields exceeding 95%, with final product purity reaching 99.1%. This represents a substantial improvement over older methods that often required complex column chromatography to achieve comparable quality.
Q: Is this synthesis route scalable for industrial commercial production?
A: Yes, the process avoids harsh conditions and uses common organic solvents like ethanol and dichloromethane. The elimination of toxic sulfur byproducts and the high efficiency of the coupling reaction make it highly suitable for large-scale manufacturing without the need for expensive specialized waste treatment for heavy metal or sulfur removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftizoxime Sodium Supplier
The technical potential of this DCC/DMAP-catalyzed synthesis route underscores the importance of partnering with a CDMO expert capable of executing complex beta-lactam chemistry with precision. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Ceftizoxime Sodium meets the highest international standards for safety and efficacy. We understand the critical nature of antibiotic supply chains and are committed to delivering consistent quality that supports your regulatory filings and market success.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this cleaner, more efficient manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify the compatibility of our production capabilities with your quality expectations. Let us collaborate to enhance the reliability and cost-effectiveness of your Ceftizoxime Sodium supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
