Advanced Purification Technology for High-Purity Ceftizoxime Sodium Manufacturing
The pharmaceutical industry continuously demands higher standards for antibiotic intermediates, particularly for third-generation cephalosporins like Ceftizoxime Sodium (SKF-88373). Patent CN102796116A introduces a groundbreaking preparation method that addresses the critical limitations of existing purification technologies. This innovation focuses on utilizing strong-acid ion exchange resin adsorption followed by precise recrystallization to achieve exceptional purity levels exceeding 99%. For global procurement leaders and R&D directors, this represents a significant shift towards more robust and reliable supply chains for essential anti-infective agents. The method effectively resolves long-standing issues regarding product clarity, stability in aqueous solutions, and the presence of related substances that often plague bulk drug manufacturing. By integrating this advanced purification logic, manufacturers can ensure a consistent supply of high-quality material suitable for sterile sub-packaging and injection formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis and purification of Ceftizoxime Sodium have been fraught with challenges that impact both cost and quality. Traditional routes, such as those disclosed in earlier patents like U.S. Pat 4427674, often rely on complex protection and de-protection strategies or direct condensation methods that generate significant side reactions. A major bottleneck in these conventional processes is the reliance on column chromatography for purification, which is notoriously difficult to scale industrially. Column chromatography not only consumes vast amounts of expensive silica gel and solvents but also suffers from low throughput and high operational costs. Furthermore, products obtained through these older methods frequently exhibit poor dissolution clarity and instability in aqueous solutions, leading to increased levels of degradation products over time. These impurities can trigger adverse clinical reactions and complicate the regulatory approval process for finished dosage forms. The inability to consistently remove heavy metals and organic by-products using standard filtration or simple recrystallization has been a persistent pain point for generic manufacturers seeking to enter competitive markets.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by replacing cumbersome chromatographic steps with a streamlined ion-exchange and recrystallization protocol. This method leverages the specific adsorption properties of strong-acid cation exchange resins, such as the D001 type, to selectively capture the Ceftizoxime acid from crude mixtures. By eluting with controlled concentrations of mineral acids like hydrochloric or sulfuric acid, the process effectively separates the active ingredient from non-ionic organic impurities. Subsequent neutralization with alkaline sodium salts, preferably sodium carbonate, allows for the precise adjustment of pH to a physiological range of 6.5 to 7.5, minimizing the risk of beta-lactam ring hydrolysis. The final recrystallization step, utilizing an ethanol-water system with strict temperature gradients from 80°C down to 10°C, ensures the formation of well-defined crystals with superior flow properties. This holistic approach not only simplifies the operational workflow but also drastically enhances the overall yield, reportedly achieving rates higher than 95% while maintaining purity above 99%.
Mechanistic Insights into Ion-Exchange Purification and Crystallization
The core of this technological advancement lies in the mechanistic interaction between the Ceftizoxime molecule and the sulfonic acid groups on the styrene-divinylbenzene resin matrix. When the crude drug solution passes through the resin bed, the basic amino groups and other ionizable functionalities on the Ceftizoxime molecule interact strongly with the acidic sites on the resin. This adsorption phase acts as a highly selective filter, retaining the target molecule while allowing neutral organic impurities and colored bodies to wash through. The subsequent elution with dilute acid protonates the resin sites, releasing the Ceftizoxime in its free acid form into a concentrated solution. This phase transfer is critical because it converts the drug into a state that is amenable to further purification without the need for organic extraction solvents that often leave toxic residues. The efficiency of this exchange is governed by the concentration of the eluent, with 10-20% hydrochloric acid or 40-50% sulfuric acid proving optimal for balancing desorption kinetics and resin capacity.
Following the ion-exchange step, the control of crystallization thermodynamics becomes the primary driver for impurity rejection. The addition of absolute ethanol to the aqueous neutralized solution alters the dielectric constant of the solvent system, reducing the solubility of the sodium salt. By heating the mixture to 80°C, any remaining micro-impurities are kept in solution, and the system reaches a homogeneous state. As the temperature is slowly lowered to 10°C, the supersaturation ratio increases gradually, promoting the growth of large, pure crystals rather than the rapid precipitation of amorphous solids that trap mother liquor. This slow cooling regime is essential for excluding structurally similar analogues and ensuring the final crystal lattice is free from solvent inclusions. The result is a product with heavy metal content reduced to trace levels, specifically below 5ppm, which is a stringent requirement for parenteral antibiotics.
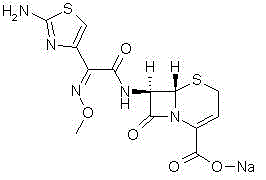
How to Synthesize Ceftizoxime Sodium Efficiently
Implementing this synthesis route requires precise adherence to the patented parameters to replicate the high yields and purity profiles observed in the experimental data. The process begins with the preparation of the ion-exchange column, ensuring the resin is fully conditioned before loading the crude Ceftizoxime solution. Operators must monitor the pH of the effluent closely to determine the breakthrough point, ensuring maximum capture of the active pharmaceutical ingredient. Once loaded, the elution must be performed at a controlled flow rate to maintain equilibrium dynamics within the resin bed. The subsequent neutralization step demands careful addition of the sodium salt solution to avoid local pH spikes that could degrade the sensitive beta-lactam core. Finally, the recrystallization vessel must be equipped with precise temperature control capabilities to execute the heating and cooling cycle accurately. Detailed standardized operating procedures for each of these stages are essential for technology transfer and scale-up.
- Adsorb crude Ceftizoxime onto a strong-acid ion exchange resin (e.g., D001 type) and elute with dilute hydrochloric or sulfuric acid to isolate the free acid form.
- Neutralize the eluted acid solution with an alkaline sodium salt such as sodium carbonate, adjusting pH to 6.5-7.5, and filter hot to remove insoluble impurities.
- Add absolute ethanol to the aqueous solution, heat to 80°C, and slowly cool to 10°C to induce crystallization of high-purity Ceftizoxime Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology offers substantial strategic benefits beyond mere technical specifications. The elimination of column chromatography represents a massive reduction in operational expenditure, as it removes the need for purchasing, disposing of, and regenerating tons of silica gel and associated organic solvents. This simplification of the downstream processing train directly translates to shorter batch cycles and increased plant throughput, allowing suppliers to respond more agilely to market demand fluctuations. Furthermore, the use of common, commodity-grade chemicals like ethanol, hydrochloric acid, and sodium carbonate reduces dependency on specialized reagents that might face supply chain bottlenecks. The robustness of the ion-exchange resin, which can often be regenerated and reused multiple times, further drives down the variable cost per kilogram of the final API. These factors combine to create a more resilient and cost-effective manufacturing model that can withstand raw material price volatility.
- Cost Reduction in Manufacturing: The transition from chromatographic purification to ion-exchange resin technology fundamentally alters the cost structure of Ceftizoxime Sodium production. By removing the most expensive and time-consuming unit operation in the traditional workflow, manufacturers can achieve significant savings in both labor and material costs. The reduction in solvent usage also lowers the burden on waste treatment facilities, decreasing environmental compliance costs associated with VOC emissions and hazardous waste disposal. Additionally, the high yield reported in the patent data means that less starting material is required to produce the same amount of finished goods, optimizing the utilization of valuable cephalosporin intermediates. These cumulative efficiencies allow for a more competitive pricing strategy in the global generic antibiotic market without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of purification steps that are prone to failure or variability. This novel method enhances reliability by utilizing robust, industrial-grade equipment such as fixed-bed reactors and standard crystallizers that are widely available in chemical manufacturing facilities. The process is less sensitive to minor fluctuations in reaction conditions compared to delicate chromatographic separations, resulting in more consistent batch-to-batch quality. This consistency reduces the rate of batch rejections and the need for re-processing, ensuring that delivery schedules are met reliably. Moreover, the raw materials required for this process are globally sourced commodities, mitigating the risk of supply disruptions that can occur with specialized chiral catalysts or exotic reagents used in other synthetic routes.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces unforeseen challenges, but the linear nature of ion-exchange and crystallization makes this technology inherently scalable from pilot plants to multi-ton commercial production. The process generates significantly less hazardous waste compared to traditional methods, aligning with increasingly stringent global environmental regulations and green chemistry principles. The ability to recycle the ion-exchange resin and recover ethanol from the mother liquor further minimizes the environmental footprint of the manufacturing site. This alignment with sustainability goals not only ensures regulatory compliance but also enhances the brand reputation of the supplier among environmentally conscious pharmaceutical partners. The simplified waste stream also reduces the complexity and cost of effluent treatment, making the facility more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Ceftizoxime Sodium preparation method. These insights are derived directly from the patent specifications and practical considerations for industrial application. Understanding these details is crucial for R&D teams evaluating technology transfer and procurement officers assessing supplier capabilities. The answers provide clarity on the mechanism, scalability, and quality control aspects of the process.
Q: How does the ion-exchange method improve Ceftizoxime purity compared to traditional chromatography?
A: The ion-exchange method selectively adsorbs the active pharmaceutical ingredient while allowing many organic impurities to pass through or be washed away, followed by specific elution. This avoids the broad-spectrum separation issues of column chromatography, resulting in significantly higher purity (>99%) and lower heavy metal content (<5ppm) without the need for complex solvent systems.
Q: What are the critical control points for ensuring stability during the recrystallization of Ceftizoxime Sodium?
A: Temperature control is paramount. The process requires heating the ethanol-water mixture to 80°C to ensure complete dissolution and then slowly cooling to 10°C. Rapid temperature changes can trap impurities or cause oiling out, which degrades the beta-lactam ring stability. Maintaining the pH between 6.5 and 7.5 during neutralization is also critical to prevent hydrolysis.
Q: Is this purification process scalable for industrial production of Ceftizoxime Sodium?
A: Yes, the process is highly scalable. It utilizes commercially available strong-acid styrene-type cation exchange resins and common solvents like ethanol and water. The elimination of complex column chromatography steps simplifies the equipment requirements, making it ideal for large-scale manufacturing from 100 kgs to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftizoxime Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity antibiotics that meet the rigorous demands of modern healthcare systems. Our technical team has extensively analyzed the pathway described in CN102796116A and possesses the expertise to implement this ion-exchange purification strategy at a commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities and heavy metals down to ppm levels. We are committed to being a partner that not only supplies materials but also optimizes the manufacturing process to ensure the highest quality standards for Ceftizoxime Sodium.
We invite global pharmaceutical partners to collaborate with us on optimizing their supply chains for this vital antibiotic. By leveraging our technical capabilities, you can secure a stable source of high-quality intermediates that reduce your overall manufacturing risks. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced purification methods can enhance your product portfolio and operational efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
