Advanced Purification of Ceftizoxime Sodium: Enhancing API Quality and Commercial Scalability
Advanced Purification of Ceftizoxime Sodium: Enhancing API Quality and Commercial Scalability
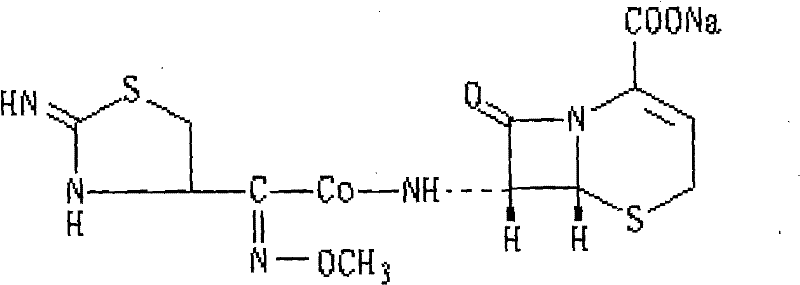
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and safety profile of critical antibiotics, and the technology disclosed in patent CN102329328B represents a significant leap forward in the processing of Ceftizoxime Sodium, also known as SKF-88373. This third-generation cephalosporin is vital for treating a broad spectrum of bacterial infections, yet its clinical efficacy is heavily dependent on the removal of polymeric impurities and residual solvents that often plague conventional synthesis routes. The patented method introduces a sophisticated multi-stage purification protocol that leverages differential solubility and pH-controlled precipitation to achieve purity levels exceeding 94%, drastically outperforming traditional acid-base adjustment techniques. For R&D directors and procurement specialists, understanding this process is crucial as it offers a pathway to higher quality bulk drugs with reduced toxic side effects, ensuring compliance with stringent global pharmacopoeia standards. By addressing the root causes of impurity retention, this innovation not only elevates product quality but also streamlines the manufacturing workflow for reliable ceftizoxime sodium supplier operations globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Ceftizoxime Sodium has relied heavily on simplistic acid-base adjustments and activated carbon decolorization, methods that are increasingly recognized for their inherent inefficiencies and inability to remove intrinsic contaminants. Traditional protocols often involve adjusting the pH using hydrochloric acid followed by neutralization, a process that fails to effectively separate polymeric impurities formed during the high-temperature synthesis or storage of the bulk drug. Furthermore, the reliance on activated carbon, while useful for color removal, introduces the risk of carbon particle contamination and often adsorbs a portion of the active pharmaceutical ingredient, leading to suboptimal yields. These conventional approaches struggle to address the presence of trace heavy metals and bacterial endotoxins, which are critical quality attributes for injectable formulations. Consequently, manufacturers face challenges in consistently meeting the rigorous purity specifications required for parenteral administration, often resulting in batch rejections or the need for costly reprocessing steps that delay time-to-market.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a targeted sequence of liquid-liquid extraction, ammonia-mediated precipitation, and controlled alcoholic recrystallization to systematically eliminate distinct classes of impurities. The process begins with an organic extraction step using solvents like ethyl acetate or cyclohexane, which selectively partitions organic impurities and polymeric byproducts away from the aqueous phase containing the desired antibiotic. Following this, the introduction of ammonia gas or ammonium hydroxide creates an alkaline environment that forces inorganic salts, heavy metal catalysts, and endotoxins to precipitate as insoluble subsalts, which are then mechanically removed via filtration. This is followed by a precise recrystallization using alcoholic solvents, capitalizing on the low solubility of Ceftizoxime Sodium in ethanol to induce the formation of high-purity crystals. This multi-faceted strategy ensures that both organic and inorganic contaminants are addressed sequentially, resulting in a white, free-flowing powder with significantly enhanced dissolution rates and stability profiles suitable for sensitive medical applications.
Mechanistic Insights into Solvent Extraction and Ammonia Precipitation
The core mechanism driving the success of this purification technology lies in the exploitation of physicochemical differences between the target molecule and its contaminants, specifically focusing on solubility parameters and ionization states. In the initial extraction phase, the crude Ceftizoxime Sodium is dissolved in water, creating a polar environment where the ionic antibiotic remains solvated while non-polar or less polar organic impurities, such as residual starting materials and polymeric degradation products, are more soluble in the added organic phase. By utilizing immiscible solvents like ethyl acetate, the process effectively strips these organic contaminants from the aqueous layer without compromising the yield of the active ingredient. This step is critical because polymeric impurities are known to cause discoloration and potential immunogenic responses, and their removal at this early stage prevents them from interfering with subsequent crystallization kinetics. The efficiency of this extraction is governed by the partition coefficient, ensuring that the equilibrium favors the migration of impurities into the organic layer, thereby clarifying the aqueous solution before further processing.
Subsequent to extraction, the ammonia treatment step operates on the principle of selective precipitation driven by pH modulation and complexation chemistry. When ammonia is introduced to the aqueous solution, the pH rises to an alkaline range, typically between 8 and 11, which alters the solubility profile of inorganic impurities such as heavy metal ions and residual catalyst salts. Under these alkaline conditions, these inorganic species form insoluble hydroxides or subsalts that precipitate out of the solution, allowing for their physical removal through filtration. This step is particularly effective against bacterial endotoxins and trace metals that are difficult to remove via standard washing procedures. Furthermore, the removal of residual ammonia via heating prepares the solution for the final recrystallization, where the addition of an alcoholic solvent like ethanol reduces the dielectric constant of the medium. This reduction in polarity decreases the solubility of the Ceftizoxime Sodium, prompting it to crystallize in a highly ordered lattice that excludes remaining trace impurities, thus locking in the high purity achieved in the previous steps.
How to Synthesize Ceftizoxime Sodium Efficiently
Implementing this purification protocol requires precise control over solvent ratios, temperature gradients, and pH levels to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory benchtop optimization to industrial-scale production without losing efficiency. Operators must carefully monitor the extraction times and the volume of organic solvents to ensure complete removal of lipophilic impurities, while the ammonia treatment duration must be sufficient to allow for the complete precipitation of inorganic salts. The final recrystallization step demands slow cooling rates to promote the growth of large, uniform crystals that are easy to filter and dry, minimizing solvent inclusion. For a detailed breakdown of the operational parameters and specific stoichiometric ratios required for optimal results, please refer to the standardized synthesis guide below.
- Dissolve crude Ceftizoxime Sodium in water and extract with cyclohexane or ethyl acetate to remove organic impurities.
- Treat the aqueous phase with ammonia gas or ammonia water to precipitate inorganic impurities and heavy metals, then filter.
- Add an alcoholic solvent to the filtrate for controlled temperature recrystallization, followed by centrifugal washing and drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this advanced purification methodology offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their API sourcing strategies. The elimination of complex and hazardous reagents, combined with the use of common, recoverable solvents like ethanol and ethyl acetate, significantly simplifies the supply chain logistics and reduces the dependency on specialized, high-cost chemicals. This simplification translates directly into cost reduction in pharmaceutical intermediates manufacturing, as the process minimizes waste generation and lowers the burden on wastewater treatment facilities. Moreover, the robustness of the purification steps ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This reliability is paramount for maintaining continuous supply lines for essential antibiotics, especially in a market where demand for high-quality cephalosporins remains steadfast.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive and potentially toxic purification agents with readily available solvents and ammonia, which are cost-effective and easy to source in bulk quantities. By improving the overall yield through the recycling of mother liquor back into the crystallization step, the method maximizes the utilization of raw materials, thereby lowering the cost per kilogram of the final active pharmaceutical ingredient. Additionally, the removal of impurities early in the process reduces the load on downstream polishing steps, further decreasing energy consumption and operational expenses associated with extended processing times.
- Enhanced Supply Chain Reliability: Utilizing common solvents such as ethanol and ethyl acetate ensures that the manufacturing process is not vulnerable to supply disruptions of exotic or regulated chemicals, thereby enhancing the resilience of the supply chain. The simplified workflow reduces the complexity of the production schedule, allowing for faster turnaround times and more predictable delivery windows for customers. This stability is crucial for pharmaceutical companies that require just-in-time delivery of high-purity APIs to meet their own formulation and packaging schedules without the risk of inventory shortages.
- Scalability and Environmental Compliance: The method is inherently scalable, utilizing unit operations like extraction and crystallization that are well-understood and easily adapted for commercial scale-up of complex pharmaceutical intermediates. The use of recyclable solvents and the reduction of hazardous waste streams align with modern environmental, social, and governance (ESG) goals, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility. This compliance not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the global healthcare value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Ceftizoxime Sodium, derived directly from the patent specifications and practical application data. These insights are intended to clarify the mechanistic advantages and operational feasibility of the described method for potential partners and technical evaluators. Understanding these details is essential for assessing the suitability of this technology for specific manufacturing requirements and quality standards.
Q: How does the ammonia treatment step improve the purity of Ceftizoxime Sodium?
A: The ammonia treatment adjusts the solution to alkalinity, causing inorganic impurities, trace catalysts, and heavy metals to precipitate as subsalts, which are then removed by filtration, effectively reducing bacterial endotoxins and metallic residues.
Q: Why is alcoholic solvent preferred for the recrystallization of Ceftizoxime Sodium?
A: Ceftizoxime Sodium exhibits high solubility in water but very low solubility in alcoholic solvents like ethanol. This sharp contrast allows for efficient crystallization and separation of the product from remaining soluble impurities upon cooling.
Q: What specific impurities does the initial organic extraction remove?
A: The initial extraction with immiscible organic solvents like ethyl acetate or cyclohexane removes organic impurities, residual solvents from synthesis, and trace polymeric substances that cause discoloration and reduce active pharmaceutical ingredient content.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftizoxime Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity antibiotics that meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Ceftizoxime Sodium adheres to the highest international standards. Our capability to implement advanced purification technologies, such as the one described in patent CN102329328B, underscores our dedication to providing superior quality products that enhance patient safety and therapeutic outcomes.
We invite procurement leaders and R&D directors to collaborate with us to explore how this optimized purification route can drive value in your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our manufacturing capabilities can support your long-term strategic goals. Partnering with us ensures access to a reliable source of high-quality pharmaceutical intermediates backed by technical expertise and a commitment to excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
