Revolutionizing Taxane Synthesis: A Deep Dive into Nitrate-Catalyzed Selective Acylation for Commercial Scale-Up
Revolutionizing Taxane Synthesis: A Deep Dive into Nitrate-Catalyzed Selective Acylation for Commercial Scale-Up
The global demand for potent oncology therapeutics continues to drive innovation in the synthesis of complex natural product derivatives, particularly within the taxane class. Patent CN101274923B introduces a transformative methodology for the preparation of paclitaxel, baccatin III, and their critical derivatives, addressing long-standing challenges in regioselective acylation. This intellectual property outlines a sophisticated yet practical approach utilizing Group IIIB metal element nitrates as catalysts to selectively acylate hydroxyl groups at the C(10) position of 10-deacetylbaccatin III (10-DAB) and simultaneously at the C(10) and C(2') positions of 10-deacetyl-taxanes. For pharmaceutical manufacturers and supply chain strategists, this represents a pivotal shift away from cumbersome multi-step protection strategies toward a more streamlined, efficient, and economically viable production model for high-value anti-cancer intermediates.
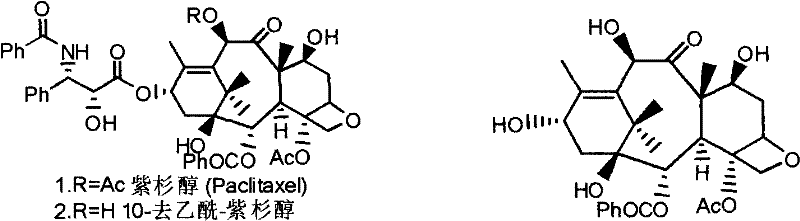
The structural complexity of taxanes, characterized by a rigid tetracyclic core and multiple reactive hydroxyl functionalities, has historically necessitated intricate synthetic routes to ensure correct functionalization. The ability to distinguish between the C(1), C(7), C(10), and C(2') hydroxyls is paramount for generating active pharmaceutical ingredients (APIs) with the requisite biological activity. The disclosed technology leverages the unique Lewis acidic properties of rare earth and scandium group nitrates to orchestrate this selectivity with remarkable precision. By enabling the direct conversion of abundant natural precursors like 10-deacetylbaccatin III into valuable intermediates such as baccatin III, this method unlocks significant potential for cost optimization and supply security in the competitive landscape of generic and branded cancer therapy production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthetic production of paclitaxel and its analogues has been plagued by inefficient acylation protocols that rely heavily on stoichiometric amounts of reagents and extensive use of protecting groups. Traditional methodologies, such as those described by Denis et al., often require the initial protection of the C(7) hydroxyl group using bulky silyl ethers like TES-Cl before acylation can occur at the C(10) position. This adds at least two additional synthetic steps—protection and subsequent deprotection—each introducing yield losses, increased solvent consumption, and extended processing times. Furthermore, alternative approaches utilizing methyleneiminium salts or strong bases like n-BuLi often demand cryogenic conditions or strictly anhydrous environments that are difficult and expensive to maintain on a multi-ton commercial scale. The reliance on hazardous reagents and the generation of substantial chemical waste streams pose significant environmental and safety liabilities for modern manufacturing facilities striving for green chemistry compliance.
The Novel Approach
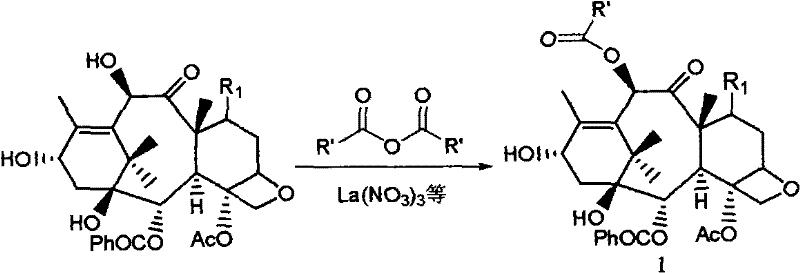
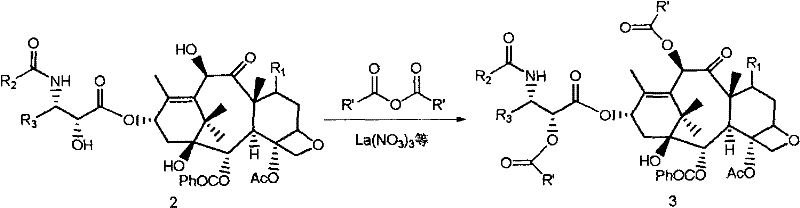
In stark contrast, the methodology detailed in CN101274923B offers a paradigm shift by employing catalytic amounts of metal nitrates to drive the acylation reaction under mild conditions. This novel approach eliminates the absolute necessity for prior protection of the C(7) hydroxyl group in many scenarios, thereby collapsing a multi-step sequence into a single, highly selective transformation. The use of acid anhydrides as acylating agents in the presence of catalysts such as lanthanum nitrate or cerous nitrate allows for the direct and selective modification of the C(10) hydroxyl on 10-DAB. Moreover, when dealing with substrates containing a C(13) side chain, such as 10-deacetyl-taxol, the system demonstrates the unique capability to simultaneously acylate both the C(10) and C(2') positions. This dual functionality not only accelerates the synthesis of advanced intermediates but also provides a versatile platform for generating diverse taxane derivatives, enhancing the strategic flexibility of R&D teams aiming to develop next-generation microtubule stabilizers.
Mechanistic Insights into Nitrate-Catalyzed Selective Acylation
The efficacy of this synthetic route hinges on the specific coordination chemistry between the Group IIIB metal cations and the oxygen atoms of the taxane substrate. Metal nitrates, particularly those of scandium, ytterbium, lanthanum, and cerium, act as potent Lewis acids that coordinate with the carbonyl oxygen of the acid anhydride, increasing its electrophilicity. Simultaneously, the metal center interacts with the hydroxyl groups of the taxane core, effectively lowering the pKa of the target hydroxyl and facilitating nucleophilic attack. The steric environment of the baccatin core, combined with the specific coordination geometry imposed by the metal catalyst, directs the acylation preferentially to the C(10) position, which is sterically more accessible compared to the hindered C(1) and C(7) positions. This mechanistic pathway avoids the formation of reactive intermediates that could lead to elimination or rearrangement side reactions, ensuring the integrity of the delicate oxetane ring and the ester side chains crucial for biological activity.
Furthermore, the choice of solvent plays a critical role in modulating the catalytic activity and selectivity of the system. While ether solvents like tetrahydrofuran (THF) are preferred for their ability to solubilize both the polar catalyst and the organic substrate while maintaining strict anhydrous conditions, the process exhibits robustness across a range of inert organic media including ethyl acetate and methylene chloride. The patent data indicates that moisture control is vital, as water can compete for coordination sites on the metal center, potentially deactivating the catalyst or hydrolyzing the anhydride. However, the tolerance of the system to various acid anhydrides—from acetic anhydride to benzoyl anhydride and even chloroacetic anhydride—demonstrates the broad substrate scope of this catalytic manifold. This versatility allows manufacturers to tailor the electronic and steric properties of the resulting intermediate, paving the way for the synthesis of novel analogues with improved pharmacokinetic profiles or reduced toxicity.
How to Synthesize Baccatin III Efficiently
The implementation of this nitrate-catalyzed acylation protocol offers a straightforward path to high-purity baccatin III and related taxane intermediates, suitable for immediate integration into existing manufacturing workflows. The process begins with the dissolution of the starting material, typically 10-deacetylbaccatin III sourced from renewable plant biomass or fermentation, in a rigorously dried solvent system. The addition of the metal nitrate catalyst, often in sub-stoichiometric quantities ranging from 0.1 to 0.5 equivalents, initiates the activation phase. Subsequent slow addition of the chosen acid anhydride ensures controlled exotherm management and maximizes regioselectivity. Following reaction completion, monitored via standard analytical techniques such as TLC or HPLC, the mixture is quenched with a mild base like sodium bicarbonate. The workup procedure is notably simple, involving standard extraction and drying steps, which facilitates the isolation of the product with high purity and minimal residual metal contamination, a critical parameter for regulatory compliance in API synthesis.
- Dissolve 10-deacetylbaccatin III in a strictly anhydrous inert organic solvent such as tetrahydrofuran (THF) or ethyl acetate under an inert atmosphere.
- Add a catalytic amount of a Group IIIB metal nitrate, such as lanthanum nitrate or cerous nitrate, to the reaction mixture to activate the C(10) hydroxyl group.
- Slowly introduce the appropriate acid anhydride acylating agent at controlled temperatures, allowing the reaction to proceed to completion before quenching and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates directly into tangible operational efficiencies and risk mitigation strategies. The primary economic driver is the drastic simplification of the synthetic route, which inherently reduces the number of unit operations required per batch. By eliminating the need for separate protection and deprotection stages, manufacturers can significantly reduce the consumption of expensive silylating agents, strong bases, and the vast quantities of solvents associated with intermediate isolations. This reduction in material intensity not only lowers the direct cost of goods sold (COGS) but also diminishes the logistical burden of sourcing and storing hazardous chemicals. Furthermore, the shortened cycle time per batch enhances facility throughput, allowing existing production assets to generate higher volumes of critical intermediates without the need for capital-intensive expansion of reactor capacity.
- Cost Reduction in Manufacturing: The elimination of stoichiometric protecting group reagents and the reduction in total synthetic steps lead to substantial savings in raw material costs. Additionally, the use of inexpensive and readily available metal nitrate catalysts replaces costly transition metal complexes or specialized organocatalysts, further driving down the input cost profile. The simplified downstream processing reduces energy consumption associated with distillation and drying, contributing to a leaner and more cost-effective manufacturing footprint that improves overall margin potential for high-volume taxane production.
- Enhanced Supply Chain Reliability: Reliance on commodity chemicals such as acid anhydrides and common metal salts mitigates the risk of supply disruptions often associated with bespoke or highly specialized reagents. The robustness of the reaction conditions, which can proceed effectively at ambient or slightly elevated temperatures, reduces the dependency on complex cryogenic infrastructure, thereby increasing the resilience of the production line against equipment failures or utility fluctuations. This stability ensures a consistent and reliable flow of high-quality intermediates to downstream coupling partners, safeguarding the continuity of the final API supply chain against market volatility.
- Scalability and Environmental Compliance: The atom economy of the direct acylation process is superior to traditional methods, resulting in less chemical waste and a reduced environmental footprint. The avoidance of halogenated solvents in favor of greener alternatives like ethyl acetate or THF aligns with increasingly stringent global environmental regulations and corporate sustainability goals. The scalability of the process is evidenced by its compatibility with standard stainless steel reactors, avoiding the need for specialized glass-lined or Hastelloy equipment, which facilitates rapid technology transfer from pilot scale to multi-ton commercial production while maintaining strict adherence to safety and environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrate-catalyzed acylation technology. These insights are derived directly from the experimental data and scope defined within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What distinguishes this nitrate-catalyzed method from traditional acylation techniques?
A: Unlike conventional methods that often require harsh conditions or multiple protection-deprotection steps, this patent utilizes mild Group IIIB metal nitrates to achieve high regioselectivity at the C(10) and C(2') positions without compromising sensitive functional groups.
Q: Can this process be scaled for industrial production of taxane intermediates?
A: Yes, the use of commercially available metal nitrates and common solvents like THF or ethyl acetate, combined with ambient temperature operation, makes this process highly amenable to large-scale commercial manufacturing.
Q: How does this technology impact the purity profile of the final API intermediate?
A: The high selectivity of the catalyst minimizes the formation of regioisomers and side products, significantly simplifying downstream purification and ensuring a superior impurity profile for subsequent coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced oncology market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of nitrate-catalyzed acylation can be seamlessly translated into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling complex taxane chemistry allows us to navigate the subtleties of regioselectivity and impurity control, guaranteeing a product quality that supports your regulatory filings and clinical timelines.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your competitiveness in the global marketplace. Let us be your partner in securing a sustainable and cost-efficient supply of critical cancer therapy intermediates.
