Advanced Synthesis of 3-Aminopiperidine: A Scalable Route for High-Purity API Intermediates
Advanced Synthesis of 3-Aminopiperidine: A Scalable Route for High-Purity API Intermediates
The pharmaceutical industry's relentless pursuit of safer and more efficient synthetic routes for critical heterocyclic intermediates has found a significant breakthrough in patent CN103373953A. This document details a robust preparation method for 3-aminopiperidine and its optical isomers, specifically addressing the severe safety and scalability limitations inherent in legacy manufacturing processes. As a key structural motif in the synthesis of DPP-IV class antidiabetic medicines such as Alogliptin and Linagliptin, the demand for high-purity 3-aminopiperidine is escalating globally. The disclosed technology leverages inexpensive, readily available starting materials derived from glutamic acid to achieve high yields under moderate reaction conditions. For R&D directors and procurement strategists, this patent represents a pivotal shift away from hazardous reagents like sodium azide and lithium aluminum hydride, offering a pathway to cost reduction in pharmaceutical intermediate manufacturing while ensuring stringent safety compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminopiperidine has been plagued by methods that are fundamentally unsuited for modern, large-scale industrial application due to safety hazards and poor atom economy. One prevalent legacy route involves the conversion of 3-hydroxypiperidine through mesylation followed by azidation and reduction. As illustrated in the reaction scheme below, this pathway relies heavily on sodium azide, a compound known for its extreme toxicity and explosive potential, creating unacceptable operational risks in a production facility.
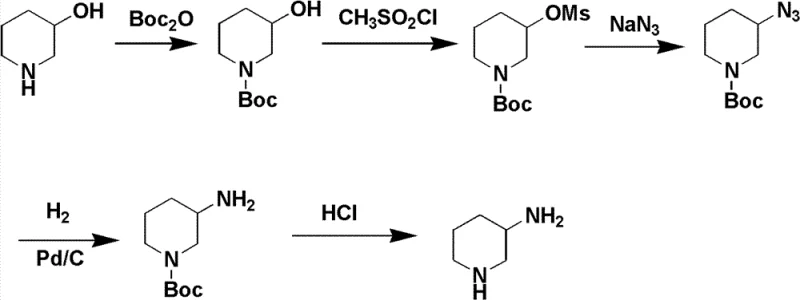
Furthermore, alternative methods utilizing 2,5-ornithine often suffer from racemization during the cyclization step, leading to a significant drop in the enantiomeric excess (ee%) of the final product. Other approaches requiring catalytic hydrogenation of 3-aminopyridine necessitate high-pressure equipment and struggle with incomplete reactions and difficult by-product removal. These technical bottlenecks not only inflate the cost of goods sold (COGS) but also introduce substantial variability in supply continuity, making them unreliable for meeting the rigorous quality standards of global regulatory bodies.
The Novel Approach
In stark contrast to these hazardous and inefficient legacy routes, the method disclosed in CN103373953A introduces a streamlined cyclization strategy that prioritizes safety and stereochemical integrity. The core innovation lies in the reaction of a protected glutamic acid derivative (Formula I) with an amine source (R2NH2) to directly construct the piperidine ring. This approach eliminates the need for explosive azides and dangerous reducing agents like lithium aluminum hydride. Instead, it utilizes mild thermal conditions, typically between 20°C and 60°C, to drive the intramolecular nucleophilic substitution.
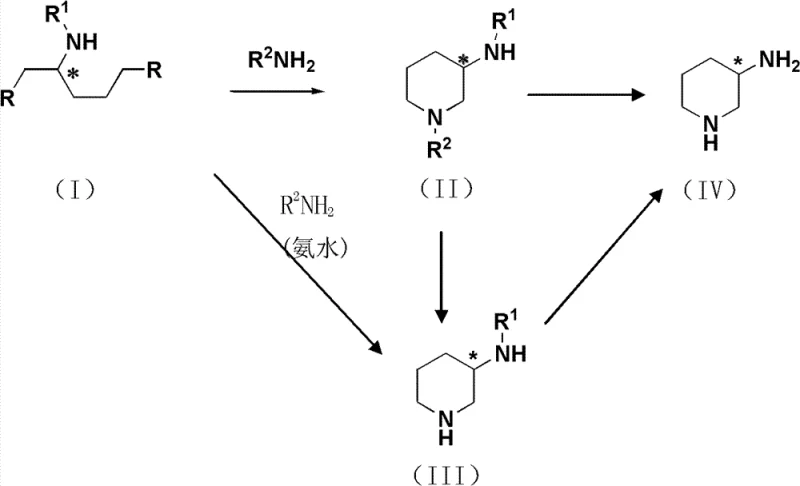
The versatility of this novel approach allows for the synthesis of both racemic and optically pure isomers simply by selecting the appropriate chirality of the glutamic acid starting material. The reaction proceeds with high selectivity and minimal by-product formation, significantly simplifying downstream purification. For a reliable pharmaceutical intermediate supplier, this translates to a process that is not only safer to operate but also inherently more scalable, capable of transitioning smoothly from kilogram-level pilot batches to multi-ton commercial production without the engineering complexities associated with high-pressure or cryogenic chemistry.
Mechanistic Insights into Intramolecular Cyclization and Deprotection
The mechanistic elegance of this synthesis lies in the precise control of the nucleophilic attack during the ring-closing step. When the compound of Formula I, containing leaving groups such as alkyl sulfonates (e.g., mesylates), reacts with an amine like allylamine or benzylamine, the nitrogen atom acts as a nucleophile attacking the electrophilic carbon adjacent to the leaving group. This intramolecular displacement forms the six-membered piperidine ring with high fidelity. Crucially, because the chiral center is established in the precursor glutamic acid derivative and the reaction conditions are mild (avoiding strong bases or high heat that could induce epimerization), the optical purity is effectively locked in. This mechanism ensures that the resulting intermediate (Formula II or III) retains the desired stereochemistry, whether it be the (R)- or (S)-configuration.
Following cyclization, the removal of protecting groups is achieved through highly selective catalytic hydrogenation or acid hydrolysis. For instance, when benzyloxycarbonyl (Cbz) or allyl groups are used as protecting groups, they can be cleanly removed using palladium on carbon (Pd/C) catalysts under atmospheric hydrogen pressure. This step is critical for impurity control, as it avoids the formation of complex side products often seen with harsh chemical deprotection methods. The process described in the patent demonstrates that by carefully selecting the protecting group R1 (such as Boc or Cbz) and the temporary amine protector R2, manufacturers can tailor the solubility and crystallization properties of intermediates, thereby facilitating easier isolation and higher overall purity of the final high-purity 3-aminopiperidine product.
How to Synthesize 3-Aminopiperidine Efficiently
The operational simplicity of this patented route makes it an ideal candidate for technology transfer and rapid scale-up. The process generally follows a logical sequence of cyclization, deprotection, and salt formation, utilizing standard reactor equipment found in most fine chemical facilities. The reaction parameters are forgiving, with broad temperature windows and tolerance for various solvent systems including alcohols, esters, and aqueous mixtures. Below is a structured overview of the standardized synthesis steps derived from the patent examples, designed to guide process engineers in implementing this superior methodology.
- React a protected glutamic acid derivative (Formula I) with a secondary amine or ammonia to form the piperidine ring structure (Formula II or III) via intramolecular cyclization.
- Remove the amino protecting group (R2) from the intermediate using a metal catalyst such as Pd/C under hydrogenation conditions to yield the free amine or protected amine (Formula III or IV).
- Perform final deprotection of the primary amine group (R1) using acid hydrolysis or hydrogenolysis to obtain the final 3-aminopiperidine product or its corresponding salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers profound strategic advantages beyond mere technical feasibility. The shift from specialized, hazardous reagents to commodity-grade starting materials fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the dependency on sodium azide and lithium aluminum hydride, manufacturers remove significant safety liabilities and the associated costs of specialized waste disposal and insurance. This qualitative improvement in process safety directly correlates to lower operational expenditures and reduced downtime risks, ensuring a more stable supply of critical intermediates for downstream API production.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the utilization of low-cost, abundant raw materials such as glutamic acid derivatives, which are significantly cheaper than the chiral esters or protected diols required in alternative methods. Additionally, the elimination of expensive transition metal catalysts (other than standard Pd/C) and the avoidance of high-pressure hydrogenation equipment reduce capital expenditure (CAPEX) and maintenance costs. The high yields reported in the patent examples, often exceeding ninety percent in key steps, minimize material loss and maximize throughput, leading to substantial cost savings per kilogram of finished product.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term API contracts, and this method excels by relying on feedstocks that are widely available in the global chemical market. Unlike niche chiral building blocks that may have single-source suppliers and long lead times, glutamic acid is a fermentation product produced at massive scales worldwide. This abundance ensures that production schedules are not held hostage by raw material shortages. Furthermore, the robustness of the reaction conditions means that manufacturing can proceed with consistent quality even if minor variations in utility supplies occur, enhancing the overall reliability of the supply chain.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior to legacy methods. The absence of toxic azides and the use of catalytic hydrogenation instead of stoichiometric metal hydride reductions drastically reduce the generation of hazardous waste streams. This simplifies wastewater treatment and lowers the burden of environmental compliance, making it easier to obtain permits for capacity expansion. The mild reaction conditions also imply lower energy consumption for heating and cooling, aligning with modern sustainability goals and reducing the carbon footprint of commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on how this method outperforms traditional alternatives in terms of safety, purity, and operational efficiency.
Q: Why is the traditional azide method for 3-aminopiperidine synthesis considered unsafe for industrial scale-up?
A: The conventional method utilizing sodium azide involves significant operational hazards due to the explosive nature of azides and poor controllability during the reaction. Furthermore, the subsequent reduction steps often require harsh conditions that complicate waste treatment and increase safety risks in large-scale manufacturing environments.
Q: How does the new cyclization method improve optical purity compared to the ornithine route?
A: Unlike the ornithine-based route which is prone to racemization during the ring-closing step, the novel method utilizes chiral glutamic acid derivatives where the stereocenter is preserved throughout the mild cyclization process. This ensures the final optical isomer achieves high optical purity without the need for complex resolution steps.
Q: What are the cost advantages of using glutamic acid derivatives as starting materials?
A: Glutamic acid and its derivatives are commodity chemicals with low market prices and high availability compared to specialized chiral esters or protected diols. This abundance significantly reduces raw material costs and mitigates supply chain risks associated with sourcing niche intermediates for pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a safer, more efficient synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN103373953A are fully realized in practical, GMP-compliant manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-aminopiperidine meets the exacting standards required for diabetes medication synthesis.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss route feasibility assessments and to obtain specific COA data for our high-purity intermediates, ensuring your project moves forward with confidence and efficiency.
