Scalable Synthesis of Dronedarone Intermediates via Novel Reductive Amination Technology
Introduction to Advanced Dronedarone Synthesis
The pharmaceutical landscape for cardiovascular treatments continues to evolve, driven by the demand for more efficient and safer manufacturing processes for critical active pharmaceutical ingredients (APIs) like Dronedarone. Patent CN103562196A introduces a transformative approach to synthesizing Dronedarone and its pharmaceutically acceptable salts, specifically focusing on a novel reductive amination strategy. This technology represents a significant departure from legacy synthetic routes, addressing long-standing challenges related to safety, yield, and purification complexity. By utilizing a specific amine intermediate compound (Formula II) and reacting it with butyraldehyde or butyric acid under reductive conditions, this method achieves high total recovery and exceptional product purity. For R&D directors and process chemists, this patent offers a robust alternative that mitigates the risks associated with hazardous reagents while streamlining the final alkylation step. The strategic shift towards reductive amination not only enhances the chemical elegance of the synthesis but also aligns with modern green chemistry principles, making it a highly attractive candidate for commercial scale-up in the competitive API intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dronedarone has been plagued by inefficient linear pathways and hazardous reaction conditions that pose significant barriers to industrial viability. As illustrated in prior art such as EP0471609 (Method A), traditional routes often rely on a progressive buildup of the molecular structure, which inherently increases complexity and cost as the synthesis proceeds. A critical bottleneck in these conventional methods is the reliance on aluminum chloride (AlCl3) for the cleavage of methoxy groups, a step that is not only dangerous due to the exothermic nature of the reaction but also generates substantial amounts of corrosive waste that complicates downstream processing. Furthermore, alternative methods like those disclosed in WO02/48078 (Method B) suffer from poor yields in the final amination steps, often reported as low as 61.6%, and necessitate cumbersome chromatographic purification to separate difficult diformylsulfonylation byproducts. These technical deficiencies translate directly into higher production costs, extended lead times, and increased environmental liability, rendering such processes suboptimal for large-scale commercial manufacturing.
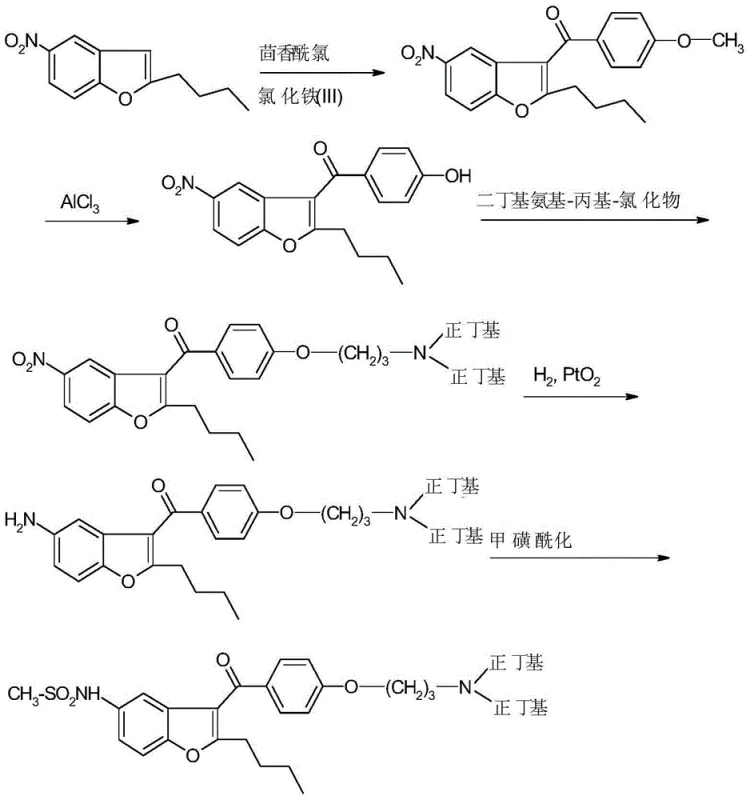
The Novel Approach
In stark contrast to these legacy methodologies, the novel process described in CN103562196A leverages a convergent reductive amination strategy that fundamentally simplifies the construction of the di-n-butylamino side chain. Instead of building the molecule linearly or relying on harsh Lewis acids, this innovative route utilizes a pre-formed amine intermediate (Formula II) which is directly alkylated using butyraldehyde or butyric acid in the presence of mild reducing agents. This approach effectively bypasses the dangerous demethylation steps and eliminates the formation of complex sulfonylated impurities that characterize older techniques. The versatility of the method allows for the use of commercially available reducing agents such as sodium triacetoxyborohydride or sodium borohydride, providing process chemists with flexible options to optimize reaction kinetics and selectivity. By shifting the critical bond-forming event to a reductive amination, the process achieves superior atom economy and significantly reduces the number of purification stages required, thereby enhancing the overall throughput and economic feasibility of Dronedarone production.
Mechanistic Insights into Reductive Amination of Benzofuran Derivatives
The core chemical innovation of this patent lies in the precise execution of the reductive amination reaction on the benzofuran scaffold, a transformation that requires careful control to avoid over-alkylation or degradation of the sensitive sulfonamide moiety. The reaction mechanism involves the initial formation of an imine or iminium ion intermediate between the primary amine of Formula II and the carbonyl group of butyraldehyde, which is subsequently reduced in situ to the secondary amine. Remarkably, the inventors discovered that this process can be driven further to the tertiary amine (the di-n-butylamino group found in Dronedarone) by managing the stoichiometry of the aldehyde and the reducing agent. When using sodium triacetoxyborohydride, the reaction proceeds efficiently at mild temperatures between 10°C and 30°C in solvents like dichloromethane, ensuring that the methanesulfonamide group on the benzofuran ring remains intact. Alternatively, the use of butyric acid with sodium borohydride at slightly higher temperatures (30-60°C) offers a distinct mechanistic pathway that avoids the handling of volatile aldehydes while still achieving high conversion rates. This dual-pathway capability provides a significant advantage for process optimization, allowing manufacturers to select conditions that best fit their existing infrastructure and safety protocols.
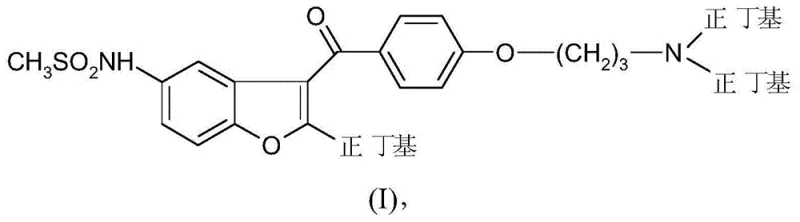
Impurity control is another critical aspect where this novel mechanism outperforms traditional Vilsmeier or Friedel-Crafts approaches. In conventional syntheses, the introduction of the amino side chain often leads to a mixture of mono- and di-alkylated products that are structurally similar and difficult to separate without preparative HPLC. However, the reductive amination protocol described here demonstrates exceptional selectivity, largely due to the specific reactivity profile of the reducing agents employed. Sodium triacetoxyborohydride, for instance, is known for its chemoselectivity in reducing iminium ions over ketones or esters, which prevents unwanted side reactions with the benzoyl ketone functionality present in the molecule. Furthermore, the patent details a highly effective purification strategy involving the formation of oxalate salts, which crystallizes the desired product with high purity (up to 99.7% by HPLC) while leaving organic impurities in the mother liquor. This crystallization-driven purification is far more scalable and cost-effective than chromatographic methods, ensuring that the final API intermediate meets stringent regulatory specifications for residual solvents and related substances without requiring extensive reprocessing.
How to Synthesize Dronedarone Intermediate Efficiently
The practical implementation of this synthesis route involves a series of well-defined steps that begin with the preparation of the key amine intermediate, Formula II, which serves as the convergence point for the entire process. Depending on the available starting materials, Formula II can be accessed either through the hydrogenation of a cyano-precursor (Formula IV) using Raney Nickel or via the deprotection of a protected amine derivative (Formula V). Once the amine intermediate is secured, the critical reductive amination is performed by dissolving the substrate in a halogenated solvent such as methylene chloride or 1,2-dichloroethane, followed by the addition of butyraldehyde and the reducing agent. The reaction mixture is typically stirred for several hours at controlled temperatures to ensure complete conversion, after which the product is isolated through a straightforward aqueous workup and salt formation. For a comprehensive, step-by-step guide including specific molar ratios, stirring speeds, and detailed workup procedures, please refer to the standardized synthesis protocol provided below.
- Prepare the amine intermediate (Formula II) either via hydrogenation of the cyano-derivative (Formula IV) or deprotection of the protected amine (Formula V).
- React Formula II with butyraldehyde (Formula III) or butyric acid in a suitable solvent such as dichloromethane or tetrahydrofuran.
- Add a reducing agent like sodium triacetoxyborohydride or sodium borohydride at controlled temperatures (5°C to 80°C) to complete the reductive amination.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this reductive amination technology offers profound advantages that directly impact the bottom line and operational resilience of API manufacturing. The elimination of hazardous reagents like aluminum chloride not only reduces the cost associated with specialized waste disposal and safety equipment but also simplifies the regulatory compliance burden, facilitating faster approval cycles for new drug master files. Moreover, the reliance on commercially available starting materials such as butyraldehyde and common reducing agents ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague complex synthetic routes dependent on custom-synthesized building blocks. The high yields and purity profiles achieved through this method mean that less raw material is wasted per kilogram of final product, leading to substantial cost savings in material consumption. Additionally, the ability to purify the product via crystallization rather than chromatography drastically reduces the consumption of expensive silica gel and organic solvents, further driving down the cost of goods sold (COGS) and enhancing the overall sustainability of the manufacturing process.
- Cost Reduction in Manufacturing: The transition to this novel reductive amination process eliminates the need for expensive and hazardous Lewis acid catalysts, thereby removing the costly downstream steps required to quench and remove metal residues. By avoiding chromatographic purification in favor of crystallization, manufacturers can significantly reduce solvent usage and labor hours associated with column packing and elution. The high atom economy of the reductive amination step ensures that a greater proportion of input materials are converted into the final product, minimizing waste generation and maximizing resource efficiency. These cumulative efficiencies result in a leaner production model that is less sensitive to fluctuations in raw material pricing and energy costs.
- Enhanced Supply Chain Reliability: The synthetic route relies heavily on commodity chemicals such as butyraldehyde, sodium borohydride, and common organic solvents, all of which are produced at a global scale with robust supply chains. This contrasts sharply with older methods that may require bespoke intermediates with limited supplier bases, creating single points of failure in the supply network. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, further enhances supply chain reliability by allowing for flexibility in manufacturing locations and equipment. Consequently, procurement teams can negotiate better terms with multiple vendors and secure long-term supply agreements with greater confidence, ensuring uninterrupted production schedules for critical cardiovascular medications.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as evidenced by the successful execution of reactions at multi-gram scales in the patent examples without loss of efficiency. The absence of exothermic hazards associated with aluminum chloride cleavage makes the process safer to scale up to tonnage quantities, reducing the need for specialized reactor engineering. From an environmental standpoint, the reduction in heavy metal waste and organic solvent consumption aligns with increasingly stringent global environmental regulations, such as REACH and EPA guidelines. This proactive compliance minimizes the risk of regulatory fines and shutdowns, ensuring long-term operational continuity and protecting the company's reputation as a responsible manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Dronedarone intermediates. These insights are derived directly from the experimental data and claims within patent CN103562196A, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method. The answers highlight the specific operational parameters and quality outcomes that distinguish this process from conventional alternatives.
Q: What are the advantages of the reductive amination method over traditional Friedel-Crafts routes?
A: The reductive amination method avoids the use of hazardous aluminum chloride (AlCl3) required for methoxy group cleavage in older linear syntheses. It also eliminates the need for complex chromatographic purification associated with diformylsulfonylation byproducts, resulting in higher purity and safer operations.
Q: Which reducing agents are preferred for this synthesis?
A: The patent specifies sodium triacetoxyborohydride (NaBH(OAc)3) when using butyraldehyde, typically at 10-30°C. Alternatively, sodium borohydride (NaBH4) can be used with butyric acid at slightly elevated temperatures (30-60°C), offering flexibility based on reagent availability.
Q: How is product purity ensured in this new process?
A: High purity (up to 99.7% by HPLC) is achieved through the formation of stable oxalate salts during the workup phase. This crystallization step effectively removes impurities without the need for column chromatography, which is a significant bottleneck in previous methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dronedarone Intermediate Supplier
As the global demand for high-quality cardiovascular therapeutics continues to rise, partnering with a technically proficient CDMO like NINGBO INNO PHARMCHEM is essential for securing a competitive edge in the marketplace. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative reductive amination process described in CN103562196A can be seamlessly transferred from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Dronedarone intermediate meets the highest international standards. Our commitment to technical excellence means that we do not just supply chemicals; we deliver validated, scalable solutions that de-risk your supply chain and accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this more efficient route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us collaborate to optimize your API supply chain with superior chemistry and unmatched reliability.
