Advanced Quinazolinone Synthesis: Scalable Green Chemistry for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for high-value heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN112778218A, which discloses a novel method for preparing quinazolinone and its derivatives using a chitosan-loaded copper catalyst. This technology represents a paradigm shift from traditional homogeneous catalysis to a more sustainable heterogeneous system. Quinazolinones are privileged structures in medicinal chemistry, known for their diverse biological activities including antitumor, anti-inflammatory, and antibacterial properties. However, their synthesis has historically been plagued by the use of toxic solvents, harsh reaction conditions, and difficult purification steps to remove metal residues. The innovation presented in this patent addresses these critical pain points by employing a biodegradable chitosan support that stabilizes copper ions, facilitating efficient C-N bond formation under mild conditions. For R&D directors and process chemists, this offers a compelling alternative that simplifies downstream processing while maintaining high conversion rates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the quinazolinone skeleton has relied heavily on the condensation of anthranilic acid derivatives or the cyclization of o-halobenzoic acids using homogeneous transition metal catalysts. These conventional pathways often necessitate the use of high-boiling, toxic organic solvents such as DMF or DMSO, which pose significant challenges for waste management and operator safety. Furthermore, when low-valent copper catalysts are employed in a homogeneous phase, the resulting reaction mixtures contain dissolved metal species that are notoriously difficult to separate from the final product. This metal contamination is a critical quality attribute failure for pharmaceutical intermediates, often requiring expensive and time-consuming scavenging steps or repeated recrystallizations to meet stringent regulatory limits. Additionally, many traditional methods require specialized ligands to stabilize the active catalytic species, adding to the raw material cost and complexity of the supply chain. The reliance on o-amino or o-nitrobenzoic acid derivatives as starting materials further limits the structural diversity accessible through these routes, as these precursors can be expensive or synthetically challenging to functionalize prior to the cyclization step.
The Novel Approach
The methodology described in patent CN112778218A overcomes these hurdles by utilizing a chitosan-supported copper catalyst, specifically immobilizing cuprous ions onto a natural polysaccharide matrix. This heterogeneous approach fundamentally changes the workup procedure; instead of complex extractions to remove dissolved metals, the catalyst can be recovered via simple filtration.  As illustrated in the reaction scheme, the process couples substituted 2-halobenzoic acids with substituted amidine hydrochlorides in a mixed solvent system of isopropanol and water. This solvent choice is particularly advantageous for procurement teams, as it replaces hazardous volatile organic compounds with greener, cheaper, and safer alternatives. The reaction proceeds efficiently at moderate temperatures (around 90°C) without the need for exotic ligands, relying instead on the inherent coordinating ability of the amino and hydroxyl groups within the chitosan backbone to stabilize the copper center. This not only streamlines the operational protocol but also significantly broadens the substrate scope, allowing for the introduction of various substituents on both the benzene ring and the amidine moiety without compromising yield or selectivity.
As illustrated in the reaction scheme, the process couples substituted 2-halobenzoic acids with substituted amidine hydrochlorides in a mixed solvent system of isopropanol and water. This solvent choice is particularly advantageous for procurement teams, as it replaces hazardous volatile organic compounds with greener, cheaper, and safer alternatives. The reaction proceeds efficiently at moderate temperatures (around 90°C) without the need for exotic ligands, relying instead on the inherent coordinating ability of the amino and hydroxyl groups within the chitosan backbone to stabilize the copper center. This not only streamlines the operational protocol but also significantly broadens the substrate scope, allowing for the introduction of various substituents on both the benzene ring and the amidine moiety without compromising yield or selectivity.
Mechanistic Insights into Chitosan-Stabilized Copper Catalysis
The efficacy of this synthetic route lies in the unique interaction between the copper species and the chitosan support. Chitosan, a linear polysaccharide composed of randomly distributed β-(1→4)-linked D-glucosamine and N-acetyl-D-glucosamine units, acts as more than just a passive carrier. The abundant amino (-NH2) and hydroxyl (-OH) groups on the polymer chain serve as coordination sites that anchor the copper ions, preventing their aggregation into inactive metallic clusters. 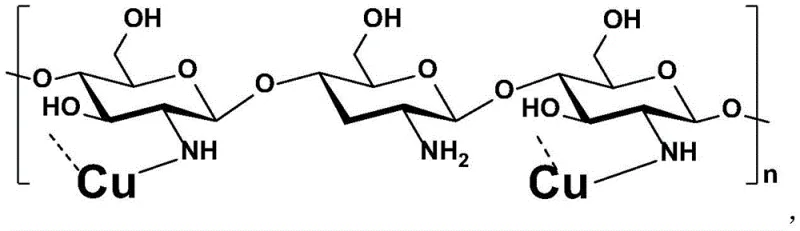 This stabilization is crucial for maintaining the active monovalent copper state (Cu+) throughout the catalytic cycle. Mechanistically, the reaction likely proceeds through an oxidative addition of the aryl halide to the copper center, followed by coordination of the amidine nitrogen. The chitosan matrix facilitates the proximity of reactants on its surface, enhancing the local concentration and promoting the subsequent intramolecular cyclization to form the quinazolinone ring. The reductive elimination step regenerates the active Cu+ species, which remains tethered to the support. This mechanism explains the observed high turnover numbers and the ability to recycle the catalyst multiple times. For quality control professionals, understanding this mechanism is vital because it predicts a very low level of metal leaching. Since the copper is chemically bound to the insoluble polymer matrix, the risk of metal contamination in the filtrate is minimized, directly addressing one of the most common causes of batch rejection in API manufacturing.
This stabilization is crucial for maintaining the active monovalent copper state (Cu+) throughout the catalytic cycle. Mechanistically, the reaction likely proceeds through an oxidative addition of the aryl halide to the copper center, followed by coordination of the amidine nitrogen. The chitosan matrix facilitates the proximity of reactants on its surface, enhancing the local concentration and promoting the subsequent intramolecular cyclization to form the quinazolinone ring. The reductive elimination step regenerates the active Cu+ species, which remains tethered to the support. This mechanism explains the observed high turnover numbers and the ability to recycle the catalyst multiple times. For quality control professionals, understanding this mechanism is vital because it predicts a very low level of metal leaching. Since the copper is chemically bound to the insoluble polymer matrix, the risk of metal contamination in the filtrate is minimized, directly addressing one of the most common causes of batch rejection in API manufacturing.
Furthermore, the use of an inorganic base like sodium carbonate in the mixed solvent system plays a dual role: it neutralizes the hydrochloride salt of the amidine to generate the free base nucleophile and assists in the deprotonation steps required for cyclization. The compatibility of the catalyst with aqueous conditions is a testament to the robustness of the chitosan-copper complex, which does not degrade or lose activity in the presence of water. This tolerance allows for the use of water as a co-solvent, which dramatically reduces the environmental footprint of the process. From an impurity profile perspective, the mild conditions and specific activation mode of this catalyst tend to suppress side reactions such as homocoupling of the aryl halide or hydrolysis of the amidine, leading to cleaner crude products that require less intensive purification.
How to Synthesize Quinazolinone Derivatives Efficiently
Implementing this technology in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and catalyst longevity. The process begins with the rigorous exclusion of oxygen, as cuprous species are susceptible to oxidation which can deactivate the catalyst. The patent outlines a standardized protocol involving vacuum-argon cycles to ensure an inert atmosphere before introducing the reagents. The ratio of substrates is optimized to drive the reaction to completion, typically using a slight excess of the amidine component. The choice of solvent ratio (Isopropanol:Water) is also critical; while the reaction tolerates a range of ratios, a 9:1 mixture has been shown to provide optimal solubility for the organic substrates while maintaining the stability of the heterogeneous catalyst suspension. Following the reaction period, which typically ranges from 2 to 18 hours depending on the substrate electronics, the solid catalyst is separated by filtration. This step is the key differentiator from homogeneous processes, as the recovered solid can be washed and directly reintroduced into the next batch. The liquid filtrate is then concentrated and subjected to standard purification techniques such as column chromatography or recrystallization to isolate the pure quinazolinone derivative.
- Replace residual gas in the reaction vessel with inert gas (Argon/Nitrogen) to ensure an oxygen-free environment.
- Add catalytic amount of chitosan-loaded copper catalyst, substituted 2-halobenzoic acid, substituted amidine hydrochloride, inorganic base, and mixed solvent (Isopropanol: Water).
- Heat the mixture for 2-18 hours, then filter to recover the catalyst, extract the product with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chitosan-copper catalyzed process offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the need for metal scavengers or complex aqueous workups designed to strip metal ions, the overall production time is significantly reduced. This efficiency gain translates directly into lower operational expenditures and increased throughput capacity for existing manufacturing facilities. Moreover, the catalyst itself is derived from chitosan, a renewable and abundant biopolymer, which insulates the supply chain from the volatility associated with synthetic ligand markets. The ability to recycle the catalyst multiple times without significant loss of activity means that the effective cost of the catalyst per kilogram of product is negligible, contributing to substantial cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic model of this process is highly favorable due to the elimination of expensive homogeneous ligands and the reduction in solvent consumption. Traditional methods often require large volumes of toxic solvents that incur high disposal costs and regulatory fees. By switching to an isopropanol-water system, companies can leverage cheaper solvent pricing and reduce waste treatment burdens. Additionally, the simplicity of the filtration-based workup reduces labor hours and equipment usage time, further driving down the cost of goods sold (COGS). The absence of heavy metal residues also mitigates the risk of costly batch failures or reprocessing, ensuring a more predictable financial outcome for each production run.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials and the robustness of the catalyst. Substituted 2-halobenzoic acids and amidine salts are commodity chemicals with stable global supply networks, reducing the risk of raw material shortages. The catalyst's stability allows for long-term storage and transport without special handling requirements, unlike sensitive organometallic complexes that may require cold chain logistics. Furthermore, the scalability of the reaction from gram-scale to multi-ton production is supported by the heterogeneous nature of the catalysis, which behaves predictably in larger reactors where heat and mass transfer are critical factors. This reliability ensures consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this green chemistry approach positions manufacturers favorably for future compliance. The use of water as a major solvent component and the generation of non-toxic waste streams simplify the permitting process for new production lines. The heterogeneous catalyst prevents the release of copper into wastewater, easing the burden on effluent treatment plants. This environmental stewardship not only avoids potential fines but also enhances the corporate reputation of the supplier, making them a preferred partner for multinational corporations with strict ESG (Environmental, Social, and Governance) mandates. The process is inherently safer, reducing the risk of accidents associated with flammable or toxic solvents, thereby protecting both personnel and assets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chitosan-supported copper catalytic system. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational advantages and scope of the technology for potential partners and licensees.
Q: How does the chitosan-supported copper catalyst improve product purity compared to traditional methods?
A: Unlike homogeneous copper catalysts which leave difficult-to-remove metal residues requiring complex purification, this heterogeneous catalyst can be simply filtered off, ensuring negligible metal contamination in the final API intermediate.
Q: Can the catalyst be reused without significant loss of activity?
A: Yes, the patent data demonstrates that the chitosan-immobilized cuprous iodide catalyst can be recovered by filtration and reused for at least five cycles with yields remaining high (91-98%), proving excellent stability and economic efficiency.
Q: What are the environmental benefits of this synthesis route?
A: The process utilizes a green mixed solvent system of isopropanol and water, avoiding toxic organic solvents, and eliminates heavy metal pollution associated with traditional copper-catalyzed reactions, aligning with strict environmental compliance standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
The technological advancements detailed in patent CN112778218A underscore the potential for greener, more efficient synthesis of high-value heterocycles. At NINGBO INNO PHARMCHEM, we recognize the importance of translating such innovative academic and patent research into reliable commercial reality. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary infrastructure to handle heterogeneous catalysis safely and efficiently, ensuring that the benefits of this chitosan-copper method are fully realized at scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinazolinone intermediate meets the exacting standards required for pharmaceutical applications, free from the metal residues that plague older technologies.
We invite procurement leaders and R&D teams to collaborate with us to evaluate the feasibility of this route for your specific project needs. By leveraging our expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can secure a sustainable and cost-effective supply chain for your quinazolinone derivatives, ensuring continuity and quality for your downstream drug development programs.
