Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in medicinal chemistry. A significant breakthrough in this domain is detailed in Chinese Patent CN113045503B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing readily available trifluoroethylimidoyl chloride and various amines as starting materials. The introduction of the trifluoromethyl group is strategically vital, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, properties that are critical for modern drug design. This patent not only addresses the synthetic challenges associated with these structures but also provides a scalable pathway applicable to the production of complex drug molecules such as Rutaecarpine, thereby offering substantial value to reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinone compounds has been fraught with significant technical and economic hurdles that impede large-scale commercialization. Conventional literature methods often rely on the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions, which frequently necessitates harsh reaction environments and rigorous temperature control. Alternative approaches involving the reaction of anthranilates with unstable trifluoroacetamides or the use of isatoic anhydride with trifluoroacetic anhydride suffer from poor atom economy and the generation of difficult-to-remove byproducts. Furthermore, methods promoted by coupling reagents like T3P, while effective on a small scale, introduce substantial cost burdens due to the expense of the reagents and the complexity of the downstream purification processes required to remove phosphorus-containing impurities. These traditional routes are generally characterized by narrow substrate scopes, low yields, and the requirement for pre-activated substrates, making them unsuitable for the cost reduction in API manufacturing demanded by today's competitive market.
The Novel Approach
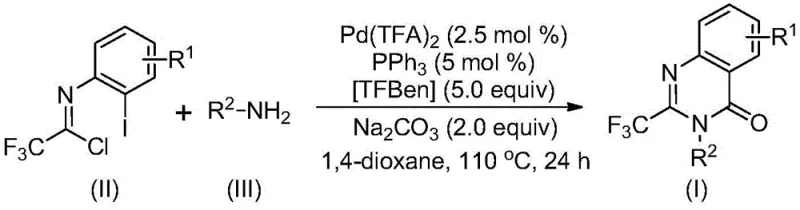
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a palladium-catalyzed carbonylation cascade that fundamentally simplifies the synthetic logic. By employing cheap and easily accessible trifluoroethylimidoyl chloride and amines as the primary building blocks, the process eliminates the need for expensive pre-activation steps or hazardous gaseous carbon monoxide sources. The reaction proceeds efficiently in common organic solvents like 1,4-dioxane at a moderate temperature of 110°C, demonstrating exceptional functional group tolerance across a wide range of substituents including halogens, alkyl groups, and trifluoromethyl moieties. This methodology not only streamlines the operation by reducing the number of unit operations but also significantly improves the overall reaction efficiency, with experimental data showing isolated yields reaching as high as 98% for certain substrates. The ability to directly access diverse 2-trifluoromethyl quinazolinone derivatives through simple substrate design makes this approach a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The core of this technological advancement lies in the intricate catalytic cycle mediated by palladium trifluoroacetate and triphenylphosphine. The reaction mechanism is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the trifluoroethylimidoyl chloride and the amine, generating a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst undergoes oxidative addition into the carbon-iodine bond of the aromatic ring, forming a key divalent palladium intermediate. A critical feature of this system is the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, which releases CO under heating conditions to insert into the carbon-palladium bond, thereby forming an acyl palladium species. This insertion step is pivotal as it constructs the carbonyl functionality of the quinazolinone ring without the safety risks associated with handling high-pressure CO gas.
Following the CO insertion, the presence of a base such as sodium carbonate facilitates the formation of a palladium-nitrogen bond, leading to the generation of a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted quinazolinone product and regenerates the active palladium catalyst. This mechanistic pathway ensures high purity and minimizes the formation of side products, as the cascade nature of the reaction drives the equilibrium towards the desired cyclic product. Understanding this mechanism is crucial for R&D directors aiming to further optimize reaction parameters or adapt the chemistry to novel substrates, as it highlights the delicate balance between ligand selection, base strength, and temperature required to maintain catalyst turnover and prevent deactivation.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory equipment such as Schlenk tubes or pressure reactors capable of maintaining 110°C. The protocol involves mixing the palladium catalyst, ligand, CO source, additive, and substrates in an aprotic solvent, followed by heating for a defined period of 16 to 30 hours. Post-reaction processing is straightforward, typically involving filtration to remove inorganic salts, followed by silica gel treatment and column chromatography to achieve high-purity isolation. The detailed standardized synthesis steps, including precise molar ratios and workup procedures validated across multiple examples, are outlined below to guide process chemists in replicating these results.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits regarding cost structure and supply reliability. The shift from expensive, specialized reagents like T3P or isatoic anhydride to commodity chemicals such as trifluoroethylimidoyl chloride and simple amines drastically reduces the raw material cost base. Furthermore, the use of a solid CO source (TFBen) eliminates the logistical complexities and safety compliance costs associated with storing and transporting high-pressure carbon monoxide cylinders, thereby enhancing the overall safety profile of the manufacturing facility. The robustness of the reaction conditions allows for flexible scheduling and reduces the risk of batch failures due to sensitive parameters, ensuring a consistent supply of high-purity intermediates.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the utilization of inexpensive, commercially available starting materials and the high atom efficiency of the cascade reaction. By avoiding the use of stoichiometric amounts of costly coupling reagents and minimizing waste generation, the process significantly lowers the cost of goods sold (COGS). Additionally, the high yields observed across a broad substrate scope mean that less raw material is wasted on failed reactions or extensive purification, leading to substantial cost savings in the overall production budget without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available chemical feedstocks ensures that the supply chain is resilient against market fluctuations or shortages of niche reagents. Since the amines and imidoyl chlorides used are common industrial chemicals, sourcing is straightforward and can be diversified across multiple vendors to mitigate risk. The simplified operational workflow also reduces the dependency on highly specialized operators or complex equipment, allowing for more agile production planning and faster response times to changing market demands for these valuable pharmaceutical building blocks.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles by reducing solvent usage and avoiding hazardous gas handling. The reaction has been demonstrated to be effective on gram scales and is amenable to kilogram-level production, facilitating a smooth transition from pilot plant to commercial manufacturing. The reduced generation of toxic byproducts simplifies waste treatment protocols, helping manufacturers meet stringent environmental regulations while maintaining high throughput capabilities for the production of complex drug intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy for technical decision-makers evaluating this process for integration into their existing workflows.
Q: What are the advantages of using TFBen over gaseous CO in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, significantly enhancing operational safety by eliminating the need for high-pressure CO gas cylinders while ensuring efficient in-situ CO release at 110°C.
Q: What is the substrate scope for the amine component in this reaction?
A: The method demonstrates excellent compatibility with various amines including n-butyl, t-butyl, benzyl, cyclohexyl, and substituted aryl amines, allowing for the synthesis of diverse quinazolinone derivatives with yields up to 98%.
Q: Can this method be applied to the synthesis of complex drug molecules?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the efficient total synthesis of the bioactive alkaloid Rutaecarpine, achieving a 77% overall yield through a three-step sequence.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN113045503B for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, allowing us to deliver 2-trifluoromethyl quinazolinone derivatives that meet the exacting standards required for clinical and commercial API manufacturing.
We invite global pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for your drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, cost, and reliability in the competitive landscape of fine chemical manufacturing.
