Advanced Synthesis of Degradable Photoresist Resin Monomers for High-Resolution Lithography
The semiconductor and display industries are constantly driven by the demand for higher resolution and finer patterning capabilities in lithography processes. Patent CN111606806A introduces a groundbreaking degradable photoresist resin monomer synthesized from dimethylbicyclo[3.3.1]nonanedione, addressing critical limitations in current photolithography materials. This innovation focuses on enhancing the etching resistance and resolution of photoresist patterns, which are essential for the fabrication of advanced integrated circuits and high-definition displays. The structural design incorporates a unique bridged ring system that provides superior mechanical stability and chemical resistance during the development process. By integrating this novel monomer into resin formulations, manufacturers can achieve significantly sharper pattern edges and reduced line edge roughness. 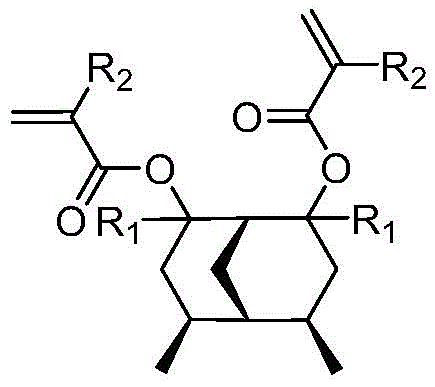 As a reliable display & optoelectronic materials supplier, understanding these molecular advancements is crucial for maintaining competitiveness in the high-tech supply chain.
As a reliable display & optoelectronic materials supplier, understanding these molecular advancements is crucial for maintaining competitiveness in the high-tech supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoresist resins often rely on linear polymer structures with single acid-sensitive groups, which inherently limit their performance in high-resolution applications. These conventional materials frequently suffer from insufficient etching resistance, leading to pattern deformation during the plasma etching steps that follow lithography. Furthermore, the solubility difference between exposed and unexposed areas in standard resins is often marginal, resulting in poor contrast and blurred feature definitions on the substrate. The reliance on simple linear chains also means that the mechanical integrity of the resist film can be compromised under harsh processing conditions. Consequently, achieving the sub-micron precision required for modern electronic devices becomes increasingly difficult with these legacy chemistries. Manufacturers face significant yield losses due to defects arising from inadequate resin performance, driving up the overall cost of production.
The Novel Approach
The technology disclosed in the patent offers a transformative solution by utilizing a monomer capable of forming a three-dimensional network structure upon polymerization. Unlike linear polymers, this new resin monomer contains two unsaturated carbon-carbon double bonds that facilitate cross-linking, creating a robust matrix that withstands aggressive etching environments. The presence of the bridged bicyclic structure further enhances the rigidity and thermal stability of the cured resist film. During exposure, the photoacid generator triggers the cleavage of the ester groups, drastically increasing solubility in the developer only in the exposed regions. 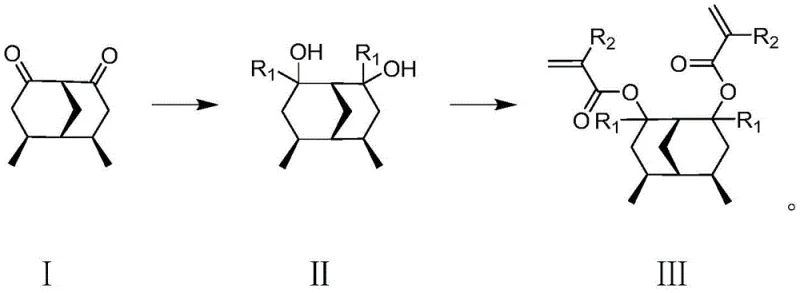 This mechanism ensures a high contrast ratio between exposed and unexposed areas, directly translating to improved resolution and edge roughness. For partners seeking cost reduction in electronic chemical manufacturing, this approach minimizes waste and rework associated with poor patterning.
This mechanism ensures a high contrast ratio between exposed and unexposed areas, directly translating to improved resolution and edge roughness. For partners seeking cost reduction in electronic chemical manufacturing, this approach minimizes waste and rework associated with poor patterning.
Mechanistic Insights into Grignard Addition and Esterification
The synthesis begins with a highly controlled Grignard reaction where (1R, 4R, 5S, 6S)-4,6-dimethylbicyclo[3.3.1]nonane-2,8-dione reacts with alkyl or cycloalkyl Grignard reagents. This step is performed under strict inert gas protection, typically nitrogen, to prevent the quenching of the reactive organomagnesium species by atmospheric moisture or oxygen. The reaction temperature is carefully maintained between 0-30 degrees Celsius to manage the exothermic nature of the addition and ensure selective formation of the diol intermediate. Anhydrous diethyl ether serves as the solvent, providing the necessary coordination environment for the magnesium center. Following the reaction, a careful quenching process with dilute acid or water is employed to protonate the alkoxide, yielding the crude diol which is then purified. 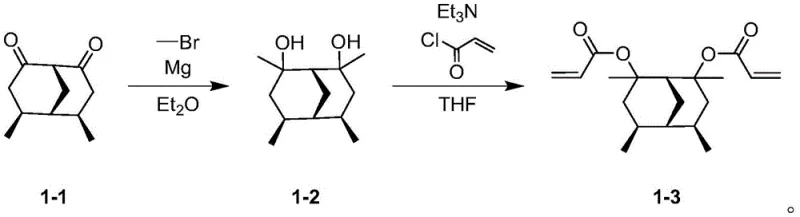 This precise control over reaction conditions is vital for minimizing side products and ensuring the stereochemical integrity of the bridged ring system.
This precise control over reaction conditions is vital for minimizing side products and ensuring the stereochemical integrity of the bridged ring system.
The second stage involves an esterification reaction where the purified diol intermediate is treated with acryloyl chloride or methacryloyl chloride. This transformation occurs under basic conditions, utilizing organic bases such as triethylamine or pyridine to scavenge the hydrochloric acid byproduct generated during the acylation. The reaction temperature ranges from 0-70 degrees Celsius, allowing for flexibility depending on the specific reactivity of the acyl chloride used. Solvents like tetrahydrofuran, toluene, or chloroform are selected to ensure complete dissolution of reactants and efficient heat transfer. The resulting resin monomer possesses the critical polymerizable vinyl groups necessary for cross-linking within the final photoresist formulation. Impurity control is achieved through rigorous workup procedures including extraction, washing with brine, and drying over anhydrous sodium sulfate, ensuring high-purity photoresist resin monomer suitable for sensitive lithographic processes.
How to Synthesize Degradable Photoresist Resin Monomer Efficiently
Implementing this synthesis route requires adherence to strict operational protocols to maximize yield and purity while ensuring safety. The process is designed to be scalable, moving from laboratory benchtop quantities to industrial production volumes without compromising the quality of the final product. Detailed standard operating procedures cover everything from reagent preparation to final purification, ensuring consistency across batches.
- Perform Grignard reaction using dimethylbicyclo[3.3.1]nonane-2,8-dione and alkyl Grignard reagent in anhydrous ether at 0-30°C.
- Quench the reaction with water and purify the intermediate diol compound via extraction and column chromatography.
- Conduct esterification with acryloyl chloride or methacryloyl chloride under basic conditions at 0-70°C to obtain the final resin monomer.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthesis technology offers substantial strategic benefits for procurement and supply chain management within the electronics sector. The elimination of complex transition metal catalysts, often required in alternative coupling reactions, simplifies the purification process and reduces the risk of metal contamination in the final product. This simplification directly correlates to lower processing costs and reduced dependency on scarce precious metal resources. Furthermore, the use of readily available starting materials and common solvents enhances supply chain resilience against market volatility. The robust nature of the reaction conditions allows for flexible manufacturing scheduling, reducing lead time for high-purity photoresist resin monomers. Overall, the process represents a significant step forward in optimizing the cost structure of advanced material production.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of expensive noble metal catalysts, relying instead on magnesium-based Grignard reagents which are cost-effective and widely available. This substitution eliminates the need for specialized metal scavenging steps, thereby reducing both material costs and processing time significantly. The high yields reported in the examples demonstrate efficient atom economy, minimizing raw material waste and disposal costs. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to overall operational expenditure savings.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as dimethylbicyclo[3.3.1]nonanedione and various acyl chlorides, are commercially accessible from multiple global suppliers. This diversity in sourcing options mitigates the risk of supply disruptions that can occur with proprietary or single-source reagents. The stability of the intermediates allows for potential stockpiling or semi-finished goods storage, providing a buffer against demand fluctuations. Consequently, manufacturers can maintain consistent production schedules and meet tight delivery windows for their downstream clients in the semiconductor industry.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the straightforward workup procedures that do not require complex separation technologies. The solvents used, such as ether and THF, are well-understood in industrial settings with established recovery and recycling protocols, supporting environmental compliance goals. The absence of heavy metals simplifies waste treatment and reduces the environmental footprint of the manufacturing facility. This alignment with green chemistry principles enhances the corporate sustainability profile of companies adopting this technology for commercial scale-up of complex electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoresist monomer technology. These insights are derived directly from the patent specifications and practical considerations for industrial application. Understanding these aspects helps stakeholders make informed decisions about integrating this material into their production workflows.
Q: What are the key advantages of this new photoresist resin monomer?
A: The monomer features a bridged ring structure and two unsaturated carbon-carbon double bonds, enabling cross-linking that significantly improves etching resistance and resolution compared to linear polymers.
Q: How does the synthesis method ensure high purity?
A: The process utilizes strict inert gas protection during the Grignard step and specific purification techniques like column chromatography and slurry washing to remove impurities effectively.
Q: Is this material suitable for large-scale semiconductor manufacturing?
A: Yes, the synthesis uses common reagents and moderate temperatures, making it highly scalable for commercial production while maintaining consistent quality for lithography applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Resin Monomer Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of synthesizing complex bridged ring structures and ensuring stringent purity specifications required for electronic grade materials. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality ensures that the photoresist monomers we supply perform consistently in your lithography processes, minimizing downtime and maximizing yield.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our specialized expertise. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to support your project development. Let us help you secure a stable supply of high-performance materials for your next-generation electronic products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
