Advanced Aldopentose-Derived Photoresist Monomers for High-Resolution Semiconductor Lithography
Introduction to Next-Generation Lithography Materials
The relentless pursuit of miniaturization in the semiconductor industry has placed immense pressure on the development of advanced photolithography materials capable of resolving sub-micron features with exceptional fidelity. Patent CN111116605B introduces a groundbreaking class of photoresist resin monomers synthesized from aldopentose, addressing critical limitations in resolution and edge roughness found in conventional systems. Unlike traditional monomers that rely solely on side-chain deprotection, these novel compounds incorporate a sugar-derived backbone that enables main-chain degradation upon exposure. This fundamental shift in chemical architecture allows for a dramatic increase in the solubility contrast between exposed and unexposed regions, which is the cornerstone of high-resolution patterning. The general structural formula, depicted below, highlights the versatile nature of this chemical platform, where R1 can be hydrogen or methyl, and R2 represents alkyl or cycloalkyl groups, offering tunable properties for specific lithographic requirements.
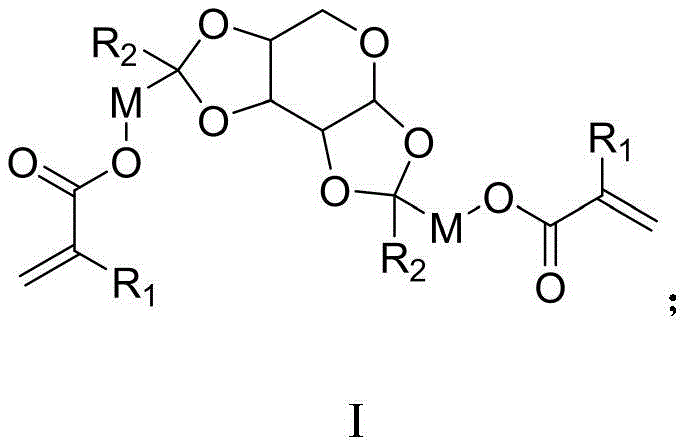
Furthermore, the inclusion of alicyclic rings and polar functionalities within the monomer structure significantly enhances corrosion resistance and adhesion to wafer substrates. For R&D directors and procurement specialists in the electronic chemicals sector, this technology represents a reliable photoresist resin monomer supplier opportunity that aligns with the industry's demand for higher performance materials. The ability to fine-tune the divalent linking group M, potentially incorporating ether bonds or ester groups, provides a robust framework for optimizing the physical and chemical properties of the final resist film. As we delve deeper into the technical specifics, it becomes evident that this aldopentose-based approach offers a compelling solution for cost reduction in electronic chemical manufacturing while simultaneously pushing the boundaries of lithographic resolution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoresist technologies predominantly utilize linear polymers formed by copolymerizing monomers with acid-sensitive protecting groups on their side chains. In these conventional systems, the exposure to light generates an acid that catalyzes the deprotection of these groups, altering the solubility of the resin in the developing solution. However, a significant drawback of this mechanism is that the polymer main chain remains intact throughout the development process. This structural integrity limits the magnitude of the solubility change, often resulting in insufficient dissolution rates for the exposed areas and consequently poorer resolution. Moreover, the linear nature of these polymers can lead to issues with pattern collapse and edge roughness, particularly as feature sizes shrink to the nanometer scale. The inability of the main chain to fragment means that large polymer molecules may remain partially insoluble, creating defects and limiting the overall fidelity of the transferred pattern on the substrate.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique chemistry of aldopentose to create monomers that facilitate main-chain scission. By incorporating a hemiacetal or ketal linkage derived from the reaction between aldopentose and hydroxyl-containing carbonyl compounds, the resulting polymer backbone becomes susceptible to acid-catalyzed cleavage. Upon exposure, the generated acid not only deprotects side chains but also breaks the hemiacetal bonds in the main chain, fragmenting the polymer into small, highly soluble segments. This dual mechanism of action—side-chain deprotection coupled with main-chain degradation—drastically amplifies the solubility difference between exposed and unexposed regions. The specific embodiments shown below, ranging from Formula II to Formula VI, illustrate the structural diversity achievable with this method, including variations with adamantyl groups and other cyclic structures that further enhance etch resistance and thermal stability.
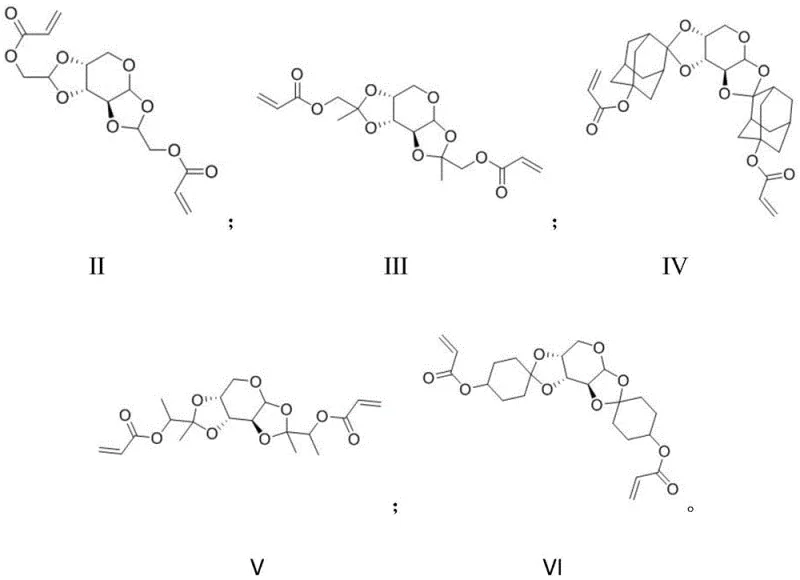
This structural innovation directly translates to improved edge roughness and resolution, addressing the core pain points of modern lithography. The presence of alicyclic rings in many of these derivatives, such as those seen in Formula IV, adds rigidity and corrosion resistance to the resin, which is crucial for withstanding harsh etching processes. For supply chain heads, understanding this distinction is vital, as it signifies a move towards more efficient materials that require less processing time to achieve high-quality patterns. The versatility of the synthesis allows for the creation of both positive and negative tone resists, broadening the application scope across various semiconductor manufacturing nodes. This novel approach effectively overcomes the solubility ceiling imposed by linear polymers, offering a clear pathway to next-generation lithographic performance.
Mechanistic Insights into Acid-Catalyzed Acetalization and Polymer Degradation
The synthesis and function of these monomers rely heavily on the principles of acetal and ketal chemistry, specifically the formation and cleavage of hemiacetal linkages under acidic conditions. The core reaction involves the condensation of hydroxyl groups on the aldopentose ring with the carbonyl group of an acrylic-functionalized aldehyde or ketone. This reaction is typically catalyzed by strong organic acids such as p-toluenesulfonic acid or sulfuric acid in azeotropic solvents like toluene to drive the equilibrium towards product formation by removing water. The resulting structure locks the acrylic polymerizable group and the sugar backbone together via a stable yet acid-labile bridge. From a mechanistic standpoint, the electron-withdrawing nature of the adjacent oxygen atoms in the acetal/ketal moiety makes the central carbon highly susceptible to protonation. Once protonated during the exposure phase of lithography, the bond cleaves readily, regenerating the carbonyl and hydroxyl components and effectively severing the polymer chain.
Impurity control is paramount in this synthesis to ensure the high purity required for electronic grade materials. The reaction conditions must be carefully managed to prevent the polymerization of the acrylic double bond during the monomer synthesis stage. This is typically achieved by adding radical inhibitors and maintaining controlled temperatures during the reflux process. Furthermore, the purification steps, often involving column chromatography as described in the examples, are critical for removing unreacted starting materials and oligomeric byproducts that could act as nucleation sites for defects in the final resist film. The use of specific starting materials, such as the hydroxyl-containing aldehydes or ketones shown in Formulas VII to XIII, allows for precise tuning of the steric and electronic environment around the acid-sensitive center. This tuning capability ensures that the monomer remains stable during storage and coating but degrades rapidly upon exposure, providing the sharp dissolution threshold necessary for high-fidelity patterning.
How to Synthesize Aldopentose-Based Photoresist Monomers Efficiently
The synthesis of these high-performance monomers follows a logical two-step sequence that is amenable to standard organic synthesis infrastructure. The first step involves the esterification of a hydroxyl-containing aldehyde or ketone with an acrylic acid derivative, typically acryloyl chloride, to introduce the polymerizable vinyl group. This intermediate is then isolated and purified to ensure high quality before proceeding to the second step. The second step is the key acetalization reaction where the acrylic intermediate is condensed with aldopentose (such as arabinose or xylose) under acid catalysis. This step forms the critical hemiacetal linkage that defines the monomer's lithographic properties. The detailed standardized synthesis steps for this process are outlined in the guide below, providing a clear roadmap for laboratory and pilot-scale production.
- React a hydroxyl-containing aldehyde or ketone with an acrylic compound (such as acryloyl chloride) in the presence of a base like triethylamine to form an acrylic ester intermediate.
- Purify the intermediate via extraction and drying, ensuring removal of residual acid chlorides and salts before the next step.
- React the purified intermediate with aldopentose (e.g., arabinose) in toluene under reflux using an acid catalyst like p-toluenesulfonic acid to form the final photoresist monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this aldopentose-based synthesis route offers significant strategic advantages for procurement and supply chain management within the electronic chemicals sector. The primary benefit lies in the accessibility and cost-effectiveness of the raw materials. Aldopentoses, such as arabinose and xylose, are abundant carbohydrates that can be sourced from renewable biomass, reducing reliance on petrochemical feedstocks and potentially lowering the carbon footprint of the manufacturing process. The co-reactants, including simple hydroxy-ketones and acryloyl chloride, are commodity chemicals available from multiple global suppliers, mitigating the risk of supply chain disruptions associated with specialized or proprietary intermediates. This abundance of starting materials ensures a stable supply chain and facilitates cost reduction in electronic chemical manufacturing by avoiding expensive, custom-synthesized precursors often required for high-end photoresists.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts or complex organometallic reagents, relying instead on common organic acids like p-toluenesulfonic acid. This simplification of the catalytic system reduces both the direct cost of reagents and the downstream costs associated with metal removal and waste treatment. The high yields reported in the patent examples, consistently exceeding 80%, indicate an efficient process with minimal material loss, further driving down the cost per kilogram of the final monomer. Additionally, the use of standard solvents like dichloromethane and toluene allows for easy recovery and recycling, contributing to overall process economics and environmental compliance.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent product quality, which is critical for maintaining yield in semiconductor fabrication lines. Because the synthesis does not depend on sensitive air- or moisture-sensitive catalysts that require specialized handling, the manufacturing process is more resilient to operational variances. The ability to produce a range of monomer variants (Formulas II-VI) from a common set of starting materials provides flexibility to adjust production based on specific customer demands without retooling entire production lines. This adaptability reduces lead time for high-purity photoresist resin monomers, allowing suppliers to respond quickly to market fluctuations and urgent customer requirements.
- Scalability and Environmental Compliance: The reaction conditions, primarily involving reflux in organic solvents, are easily scalable from gram to ton quantities using standard reactor vessels. The absence of hazardous heavy metals simplifies the waste stream, making disposal and treatment more straightforward and compliant with increasingly stringent environmental regulations. The potential for using bio-based aldopentose aligns with corporate sustainability goals, offering a 'green' credential that is increasingly valued by downstream electronics manufacturers. This scalability ensures that the commercial scale-up of complex photoresist monomers can be achieved without prohibitive capital expenditure on specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aldopentose-based technology in photoresist formulations. These answers are derived directly from the technical disclosures and beneficial effects described in the patent documentation, providing clarity on performance and applicability. Understanding these details is essential for technical teams evaluating the feasibility of integrating these monomers into existing resist platforms.
Q: What is the primary advantage of using aldopentose in photoresist monomers?
A: The incorporation of aldopentose introduces a hemiacetal or ketal structure into the polymer main chain. Upon exposure and acid generation, this linkage cleaves, breaking the polymer backbone into small segments. This significantly enhances solubility in alkaline developers compared to traditional linear polymers that only undergo side-chain deprotection, leading to superior resolution and reduced edge roughness.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the synthesis utilizes standard organic reactions such as esterification and acetalization. The reagents, including aldopentose, hydroxy-ketones, and acryloyl chloride, are commercially available commodities. The process operates under conventional reflux conditions in solvents like toluene and dichloromethane, avoiding the need for exotic high-pressure or cryogenic equipment, which facilitates straightforward scale-up.
Q: How does this monomer improve wafer adhesion?
A: The molecular structure of the monomer contains polar epoxy-like structures and multiple oxygen atoms derived from the sugar backbone. These polar functional groups increase the surface energy and interaction with the wafer substrate, thereby improving adhesion. Additionally, the potential for cross-linking during polymerization forms a net structure that further enhances mechanical stability and corrosion resistance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoresist Resin Monomer Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced electronic materials, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise requirements of photoresist monomer synthesis, ensuring stringent purity specifications and rigorous QC labs that meet the exacting standards of the semiconductor industry. We understand that the transition to new monomer architectures requires a partner who can navigate the complexities of scale-up while maintaining the critical quality attributes necessary for high-resolution lithography. Our team of expert chemists is ready to optimize the aldopentose-based synthesis route to maximize yield and minimize impurities, ensuring a consistent supply of high-performance materials.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our capabilities can support your product development goals. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our efficient synthesis methods can reduce your overall material costs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify the suitability of our monomers for your specific photoresist applications. Let us be your trusted partner in advancing the next generation of lithographic materials.
